When Liquid Grabs a Solid the Collar!
Published in Physics
Explore the Research
Simulation of a liquid drop on a soft substrate
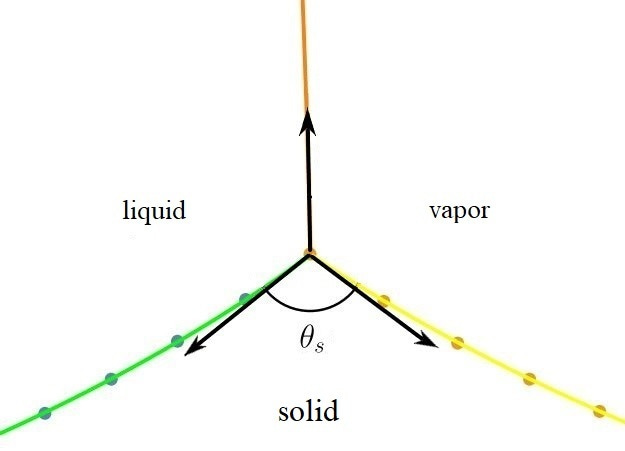
The gentle landing of dewdrops on leaves may seem harmless at first glance, but it hides a complex relationship between the liquid and solid at their point of contact. While solids are inherently rigid, they must also exhibit some flexibility to accommodate the forces exerted by the liquid. The interaction between liquid and solid is not just a simple encounter; it's dynamic and filled with tension, governed by intricate physical principles. In essence, the contact between liquid and solid is a tense affair!
Our story begins in the fall of 2022 when Yalda (the first author) and I were searching for a topic for her master's thesis. When Amir the 2nd (the third author) learned about our topic, he shared his insights from teaching fluid mechanics. He pointed out that while we often discuss the horizontal balance of forces at the solid-liquid-gas contact point leading to Young's law, we typically treat the vertical balance as a constraint, assuming the solid is completely rigid. Amir suggested that we focus on the vertical balance as a thesis problem by modeling the solid and simulating its deformation. And that's where our journey truly began!
During our literature survey, we discovered Lester's paper from the 1960s, along with subsequent studies that explored the behavior and deformation of solids by solving elasticity equations affected by the load of a droplet. A fundamental issue arose from the logarithmic singularity in the expression for the vertical displacement of the solid at the contact point. To address this singularity, researchers typically introduced a cut-off limit, which implied that the liquid was in contact with the solid over a small surface layer rather than at a single point.
A significant advancement came with the 2011 paper by Jerison et al., which proposed a natural solution to this singularity by adding a term that accounts for the bending of the solid at the contact point to the elasticity equations. This addition effectively managed the logarithmic singularity, similar to the manual introduction of a cut-off limit, but in a more integrated manner.
When we began modeling the phenomenon of wetting, our first step was to incorporate the solid into our simulation. We opted for the well-established mass-spring network system. However, we soon realized that a pure mass-spring network alone could not adequately model solids with arbitrary compressibility, which is influenced by the Poisson ratio. A network of masses connected by central (radial) springs leads to a fixed Poisson ratio. To achieve the desired Poisson ratio, we chose a method that adds an energy term proportional to the change in volume or surface area of each unit cell in the network.
Next, we introduced the liquid into the system. The liquid surface, which takes the form of a sphere in the absence of gravity, was represented as a chain of points on the liquid-vapor boundary. For each interface—solid-liquid, solid-gas, and liquid-gas—we introduced energy terms proportional to the surface tension in the system.
We set up a numerical simulation that treats the problem as an energy minimization challenge, a necessary condition for equilibrium. This approach allowed us to simulate how the droplet interacts with the material at a granular level. By examining various parameters—such as the stiffness (Young's modulus) and deformation characteristics (Poisson's ratio) of the solid substrate, the surface tension of the liquid, and geometric parameters like the contact radius and substrate thickness—we gained insights into how these factors influence the overall system.
Our method enabled us to determine the displacement of the solid and the precise points of contact with the liquid. This allowed us to analyze key characteristics, such as contact angles and effective surface tension, providing deeper insights into the equilibrium conditions at the interface.
Through our simulation, we uncovered an intriguing relationship between the liquid drop and the substrate. We found that when the soft material is less rigid, our predictive models hold up well. However, as we increased the stiffness of the solid, the results began to diverge from established theories near the contact points where the droplet meets the surface.
This raises an important question: does the assumption of zero tangential traction in these contact areas truly apply in all scenarios? Our findings suggest otherwise, indicating that there is noticeable tangential traction at these contact points, challenging long-held beliefs in the field.
One of the highlights of our research was calculating the effective surface tension at the contact points in relation to the Young’s modulus of the solid substrate. We discovered that as the stiffness of the solid increases, the effective surface tension also rises linearly. Harder solids tend to maintain a flatter surface profile, necessitating a significant increase in surface tension to balance the vertical component of the tension introduced by the liquid. Notably, for incompressible solids, the effective surface tension at the contact points exhibits a linear relationship with the solid’s Young’s modulus, highlighting a direct correlation between material stiffness and interfacial tension. This insight could inform future designs of soft materials across various applications, from soft robotics to protective coatings. Understanding these interactions better can lead to innovations in how we design materials that need to interact with liquids, enhancing everything from industrial processes to everyday products.
Our innovative approach to the wetting problem allows us to simultaneously capture the coupled behaviors and profiles of both solids and liquids. However, we recognize that we have only scratched the surface of exploring many parameters in depth. Our primary goal was to validate whether this approach works and to provide essential elements and sample codes, which are available through links in the paper.
While our simulations offer valuable theoretical insights, experimental studies would be crucial in bridging the gap between our simulated predictions and real-world observations. Such experiments could validate our findings and refine the models, fostering a more comprehensive understanding of liquid-solid interactions. We’re eager to spark discussions around these findings and would love to hear your thoughts! If you’re interested in the details or want to chat about the implications of our work, feel free to reach out. Let’s connect and dive deeper into this fascinating topic together!



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
Hi @De Coninck Joel , please check out the paper; you might find it useful!