
Explore the Research

3D bioprinting for reconstituting the cancer microenvironment - npj Precision Oncology
npj Precision Oncology - 3D bioprinting for reconstituting the cancer microenvironment
Cancer is a very complex and one of the most dreaded diseases. Although much progress have been made in diagnosis, chemotherapeutics, and with the recent advances in immunotherapies, there are still many cancer types, with very high mortality rates, which elude our understanding and current treatment options. The cost of treatment and hospital care of cancer patients also imposes a significant socioeconomic burden. The complexity of conducting clinical trials for new therapies along with high attrition rates also place tremendous demands on effective pre-clinical in vitro testing methods. However, results obtained from most in vitro or in vivo disease models so far, exhibit significantly less than the desired correlation with clinical results.
Due to the inherent complexity of cancer, in-vitro research on conventional two-dimensional (2D) culture techniques cannot faithfully recapitulate the dynamics of the disease. Apart from the mass of rapidly proliferating cancer cells, a tumor microenvironment comprises of leaky vasculature, stromal cells like fibroblasts, and several immune cells such as T cells and macrophages. Moreover, several biochemical and biophysical changes trigger signalling pathways regulating cancer pathogenesis, metastasis of tumor cells from a primary neoplasm to secondary regions.
Recapitulating such cellular and molecular processes and quantifying the effect of different therapeutic interventions in an in vitro model is bound to be challenging. Conventionally, biopolymer scaffolds, spheroid or organoid cultures, tumor-on-a-chip platforms, are some of the physiologically-relevant in vitro models. However, to enhance the performance of these models and to have better predictive power for clinical studies, the cellular heterogeneity and 3D architecture of a tumor microenvironment needs to be accurately mimicked. In this context, 3D bioprinting technology offers an obvious advantage over other fabrication techniques. Leveraging its ability to create high throughput models with excellent spatial control over biologics, complex as well as reproducible in vitro tumor models can be fabricated. Further, bioprinted tumor models can be incorporated with long term perfusable cultures that can mimic vascular tumor structures, which play essential role in tumor survival, metastasis and immune cell infiltration.
With this motivation, the present review led by Dr. Pallab Datta describes the different applications of bioprinting in fabricating such 3D cancer tissue models. For example, the freeform embedding of suspended hydrogels has potential for creating relevant neuroblastoma models. Extrusion or droplet based bioprinting modalities have also been used to establish breast, liver, pancreatic, cervical and ovarian cancer models. Bioprinted cancer models have also been fabricated to study differential response of cells at core and peripheral regions of a tumor under hypoxia. Similarly, some models have recapitulated drug resistance profiles and metastatic behaviour of cancer cells.
There have also been some advancements in studying immune-cancer interactions using in vitro tumor models as well (see Figure 1). We indeed collaborate with Dr. Derya Unutmaz (Jackson Laboratory) in order to explore immune-cancer interactions in a physiologically-relevant cancer model fabricated using advanced bioprinting technologies. Limited number of suitable bioinks, lack of high throughput generation of organoids, and difficulty in performing 3D functional assays in robust platforms are some of the challenges that needs to be addressed.
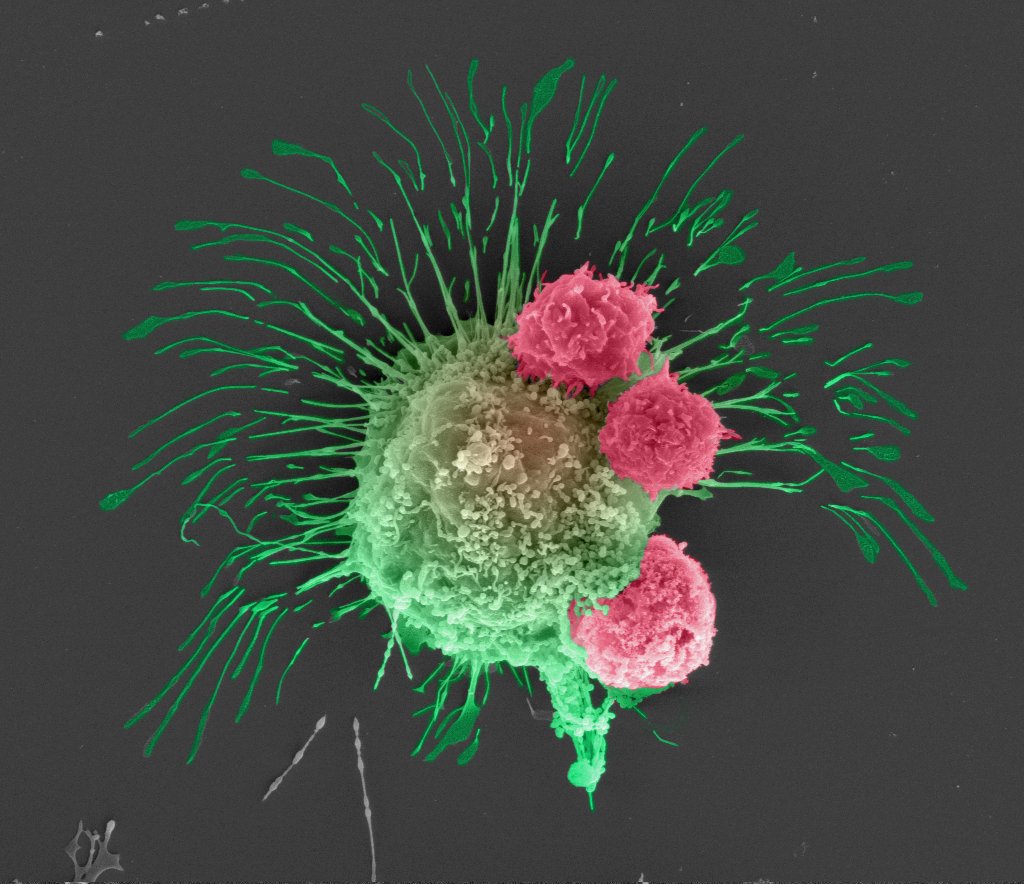
Figure 1: Scanning electron microscopy image of breast cancer cell (green) being attacked by killer T cells (pink, engineered by Unutmaz Lab at The Jackson Laboratory) to recognize it. Image by Madhuri Dey, Penn State University Ph.D. candidate, courtesy of the Ozbolat Lab.
Overall, we are hopeful and enthusiastic about the the pace at which exciting bioprinting technologies are evolving, mass scale production of in vitro constructs with high in vivo predictive performances will soon become a reality resulting in much more reliable, predictive, cost effective and rapid development of novel cancer therapeutics.
Follow the Topic
-
npj Precision Oncology
An international, peer-reviewed journal committed to publishing cutting-edge scientific research in all aspects of precision oncology from basic science to translational applications to clinical medicine.
Related Collections
With Collections, you can get published faster and increase your visibility.
Minimal Residual Disease and Circulating Tumor DNA Dynamics in Personalized Cancer Treatment
Publishing Model: Open Access
Deadline: Mar 12, 2027
Genomic Instability
Publishing Model: Open Access
Deadline: Jun 24, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in