A metabolite, not the parent compound: Mitorubin restores mitochondrial function in cardiac aging
Published in Biomedical Research
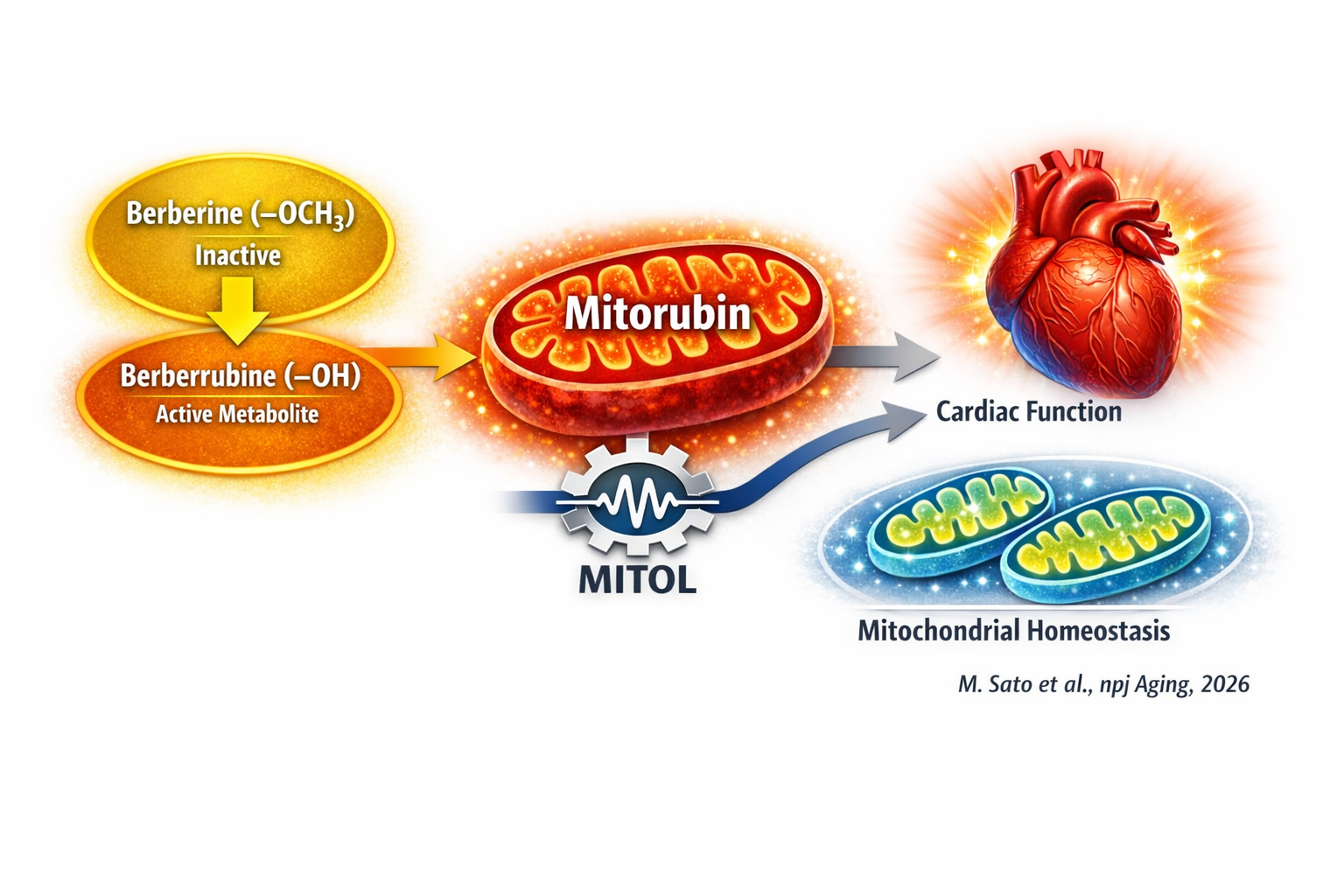
To address this question, we set out to identify compounds that restore mitochondrial function by targeting MITOL, a key regulator of mitochondrial homeostasis that declines with age.
We screened natural products and bioactive compounds, expecting known candidates. Instead, we found something unexpected. Berberine itself showed only modest effects on MITOL expression. In contrast, its major metabolite, berberrubine, significantly induced MITOL and increased mitochondrial protein expression. This shift—from parent compound to metabolite—was both surprising and conceptually important.
A major obstacle was the extremely poor water solubility of berberrubine. To overcome this, we collaborated with chemists to develop water-soluble derivatives. We collectively named this class of compounds “Mitorubin,” reflecting their mitochondria-activating properties.
Mitorubin consistently enhanced mitochondrial content and function. In cultured cells, it increased mitochondrial DNA content, upregulated mitochondrial proteins, and enhanced respiratory capacity. Notably, it induced a mild increase in mitochondrial ROS without compromising membrane potential, consistent with a mitohormesis-like adaptive response rather than overt stress.
We then asked whether these effects translate into physiological benefits in vivo. In aged mice, oral administration of Mitorubin markedly attenuated cardiac dysfunction. It improved systolic and diastolic function, suppressed cardiac hypertrophy, and alleviated pulmonary congestion. These benefits were accompanied by enhanced mitochondrial gene expression, increased respiratory capacity, and activation of autophagy-related pathways.
An intriguing observation emerged from lifespan analyses. Mitorubin did not extend lifespan in normally aged mice but significantly prolonged survival under high-fat diet conditions. This context-dependent effect suggests that Mitorubin enhances mitochondrial resilience particularly under metabolic stress.
Conceptually, our findings redefine how we think about pharmacological targeting of mitochondrial function. Rather than acting on a single downstream pathway, Mitorubin enhances mitochondrial adaptability, potentially through mitohormesis-like mechanisms.
Several questions remain open. The upstream mechanism by which Mitorubin induces MITOL is not yet fully understood, and identifying its direct molecular target is an ongoing effort. In addition, its potential applications may extend beyond the heart, including skeletal muscle and neurodegenerative diseases.
For further information, please refer to our publication: Sato, M., Tanabu, D., Torigoe, D. et al. Mitorubin, berberrubine-based compounds that improve mitochondrial function, exhibit cardioprotective effects against age-related cardiac dysfunction. npj Aging (2026). https://doi.org/10.1038/s41514-026-00366-w
Follow the Topic
-
npj Aging
The mission of this journal is to provide the community with a platform to publish new high-profile insights into all aspects of aging.
Related Collections
With Collections, you can get published faster and increase your visibility.
Mitochondria at the Heart of Aging: Structure, Function and Failure
Publishing Model: Open Access
Deadline: Jul 11, 2026
Healthy eating and dietary intervention to promote longevity and prevent age-related diseases
Publishing Model: Open Access
Deadline: Jun 09, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in