A tale of Precision Medicine and the importance of repeat genomic testing
Published in Cancer

Contributors: Niamh Coleman, Vivek Subbiah, Funda Meric-Bernstam
Acquired resistance to molecular targeted therapy is a veritable challenge in the modern era of precision medicine. The ability to truly understand these mechanisms of resistance, by improving patient selection and allowing for the development of rationally designed next-line or combination treatment strategies, can only enhance outcomes for cancer patients. In NPJ Precision Oncology, we recently published a case that highlights the importance of repeat tissue biopsy post targeted therapy.1 Using next-generation sequencing (NGS) we identify an acquired mTOR resistance following targeted AKT inhibition, and describe a remarkable response to further targeted therapy in the context of an early phase clinical trial. To our knowledge based on literature review, this is the first reported response to ATP-competitive mTOR inhibition in this setting and our case supports mTOR mutation as a novel mechanism of resistance.
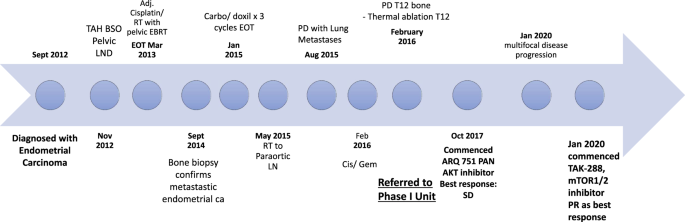
Following progression of disease on multiple lines of standard-of-care therapies, a 64-year-old patient with endometrial cancer was referred for consideration of a phase I trial.1 NGS analysis revealed an AKT1 E17K activating mutation. The patient was enrolled on a phase I study of an allosteric pan-AKT inhibitor ARQ751 (NCT02761694). The patient had stability of disease (4% reduction by RECIST criteria version 1.1)2 derived clinical benefit for twenty-seven months. On progression of disease the patient underwent repeat tumor sampling. Here, CT-guided lung biopsy confirmed adenocarcinoma consistent with the patient’s primary cancer, and NGS analysis confirmed the persistence of AKT1 E17K mutation, as well as mTOR A1459D - an activating mTOR mutation. Based on this data the patient was subsequently enrolled on a phase I study of the mTORC1/2 inhibitor sapanisertib (TAK-228) given 4mg daily with metformin 500mg twice daily (NCT03017833). To date - the patient has been on therapy for 14 months and has achieved a confirmed partial response (PR) by RECIST criteria version 1.1 2, response confirmed post-6 cycles of therapy (maximum reduction 30% from baseline) (Figure 1).1
AKT/PI3K/mTOR signaling is commonly disrupted in human cancers,3 and mTOR is a key figure in this pathway, influencing multiple processes which are directly involved in oncogenesis. Mutations in the mTOR gene are exceedingly rare, though recent pre-clinical work has shown that mTOR mutations may indeed be a mechanism of resistance to allosteric mTOR inhibitors, with a potential role for catalytic inhibitors.4 Our case, to the best of our knowledge, is the first documented clinical case of mTOR mutation as an acquired mechanism of resistance in the setting of AKT inhibition, with subsequent documented response to mTOR-based targeted therapy.
This case study is a true tale of precision medicine success: a patient with advanced incurable cancer, following multiple lines of treatment with cytotoxic chemotherapy, is referred for Phase 1 clinical trials. Here, she receives two novel early-phase genomic-driven clinical trials, and experiences durable clinical activity of targeted therapy - for almost four years. Repeat genomic testing on progression of disease allowed for the identification of a novel mechanism of resistance.
Our case highlights the importance of repeat genomic testing – without NGS, the true driver of this patient’s cancer would not have been identified and the patient would never have received tailored, targeting therapy which has dramatically increased her life expectancy and improved her quality of life. The patient continues to derive clinical benefit: she continues to work, has good control of her cancer-related symptoms and can live her life as she wishes – a true measure of targeted-therapy success. These rare cases underscore the importance of precision medicine and repeat tumor sampling over time – allowing for the ability to rapidly identify a patients’ oncogenic driver, drivers of resistance, and ultimately allowing us, as physicians, to precisely target oncogenic drivers of disease, in real-time. Liquid biopsy technology is also evolving at a rapid pace and can provide mutation information in addition to identifying resistance mechanisms. As we move forward in this era of targeted therapy, delving further into the complexities of pathways driving cancer, it is likely that real-time repeat genomic testing on disease progression will act as crucial pit-stops, key to ensuring that patients continue on their respective routes of targeted success.
References:
- Coleman N, Subbiah V, Pant S, et al. Emergence of mTOR mutation as an acquired resistance mechanism to AKT inhibition, and subsequent response to mTORC1/2 inhibition. npj Precis Oncol. 2021;5(1):99. doi:10.1038/s41698-021-00240-w
- Schwartz LH, Litière S, De Vries E, et al. RECIST 1.1 - Update and clarification: From the RECIST committee. Eur J Cancer. Published online 2016. doi:10.1016/j.ejca.2016.03.081
- Lawrence MS, Stojanov P, Mermel CH, et al. Discovery and saturation analysis of cancer genes across 21 tumour types. Nature. Published online 2014. doi:10.1038/nature12912
- Wagle N, Grabiner BC, Van Allen EM, et al. Response and Acquired Resistance to Everolimus in Anaplastic Thyroid Cancer. N Engl J Med. Published online 2014. doi:10.1056/nejmoa1403352
Follow the Topic
-
npj Precision Oncology
An international, peer-reviewed journal committed to publishing cutting-edge scientific research in all aspects of precision oncology from basic science to translational applications to clinical medicine.
Related Collections
With Collections, you can get published faster and increase your visibility.
Minimal Residual Disease and Circulating Tumor DNA Dynamics in Personalized Cancer Treatment
Publishing Model: Open Access
Deadline: Mar 12, 2027
Genomic Instability
Publishing Model: Open Access
Deadline: Jun 24, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in