Adults on Pre-Exposure Prophylaxis (tenofovir-emtricitabine) Have Faster Clearance of Anti-HIV Monoclonal Antibody VRC01
Published in Biomedical Research
Why we did this
Globally, 1.3 million people were newly diagnosed with HIV-1 in 2022. Although testing, treatment, pre-exposure prophylaxis (PrEP) have slowed the spread of HIV-1,1 many people remain at-risk. Passive immunization with broadly neutralizing monoclonal antibodies (mAbs) provides a novel approach as an additional potential HIV-1 prevention modality, alone or in combination with other existing prevention modalities. In the US, there were about 1.2 million persons with an indication for PrEP in 2020, of which 25% have received a prescription.2 Although the likelihood of an interaction between these two classes of drugs may be limited due to the distinct pathways regulating their metabolism, there have been reports of mAb-small molecule drug interactions.3 Therefore, it is important to investigate whether VRC01 pharmacokinetics (PK) in oral PrEP ([tenofovir disoproxil fumarate (TDF)-emtricitabine (FTC)], TDF-FTC) users differs from that in non-PrEP users, and if present, explore potential mechanisms of such interactions between PrEP and VRC01.
What we did
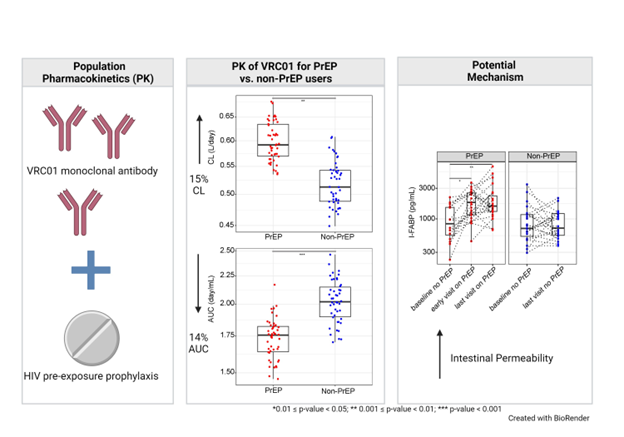
We used a cohort of 24 oral PrEP users (of TDF-FTC), and 24 non-PrEP users drawn from an efficacy trial of VRC01 in which participants were provided access to free oral PrEP, to compare PK features of VRC01 between PrEP and non-PrEP users. We measured participant serum concentrations of VRC01 after each of ten 8-weekly VRC01 infusions ~ every 4 weeks and applied population pharmacokinetics (popPK) models to analyze VRC01 concentrations over-time from both PrEP and non-PrEP users. We used the Targeted Maximum Likelihood Estimation (TMLE) approach to compare VRC01 PK features between PrEP and non-PrEP users. We estimated five individual-level PK features: clearance, volume of the peripherical compartment, distribution half-life, elimination half-life, and steady state dose-normalized area under the curve after adjusting for: age, body weight, race, baseline behavioral risk score,4 and creatinine clearance, and two inflammatory markers (IFN-γ and IL-10). We further explore potential molecular and physiologic mechanisms of the findings including an exploration of pro-inflammatory markers, liver and kidney function, and intestinal permeability.
What we found
We found two VRC01 PK features were significantly different between PrEP and non-PrEP users. Specifically, the mean clearance rate of VRC01 was 15% greater, and the mean area under the curve of VRC01 serum concentrations was 14% lower in PrEP users than in non-PrEP users.
These differences were not associated with serum inflammatory markers, liver, or kidney functions. We did; however, observe a significant increase in I-FABP levels, a marker of intestinal permeability, over time, among PrEP users, but not among non-PrEP users. I-FABP levels measured at about 4 weeks after PrEP use also positively correlated with the subsequent clearance rate of VRC01. We did not find evidence of an association with intestinal permeability via LBP, a marker of larger size than I-FABP, suggesting a selective leakage mechanism.
Why it matters
Given that oral PrEP has been licensed in several countries as an HIV-1 prevention tool, additional studies of mAbs for HIV prevention may include PrEP users. Understanding differential mAb PK measurements among PrEP users could ensure optimal dosing. The long duration of I-FABP upregulation may also be important to assess if the increased clearance rate of VRC01 also affects other populations taking anti-retrovirals, such as people living with HIV, who participate in treatment interruption trials using monoclonals.
Given the low to moderate influence on VRC01 clearance and area under the curve between PrEP and non-PrEP users, it is unlikely that dose adjustment would be needed for VRC01. However, it may be needed if verified for other HIV-1 mAbs entering the prophylaxis pipeline. Similarly, because the PK of VRC01resembles the PK of other IgG-based monoclonals, our investigation suggests that the PK of prophylactic or therapeutic antibodies for other diseases in PrEP users should be investigated to determine if optimal doses of immunotherapies are affected by PrEP.
If you want to know more, you can read the full text here: Adults on pre-exposure prophylaxis (tenofovir-emtricitabine) have faster clearance of anti-HIV monoclonal antibody VRC01 | Nature Communications
References
- Joint United Nations Programme on HIV/AIDS. IN DANGER: UNAIDS Global AIDS Update 2022. Licence: CC BY-NC-SA 3.0 IGO. 2022. https://www.unaids.org/sites/default/files/media_asset/2022-global-aids-update-summary_en.pdf
- Centers for Disease Control and Prevention. PrEP Coverage. Accessed May 30, 2023. https://www.cdc.gov/hiv/statistics/overview/in-us/prep-coverage.html
- Ferri N, Bellosta S, Baldessin L, Boccia D, Racagni G, Corsini A. Pharmacokinetics interactions of monoclonal antibodies. Pharmacological Research. 2016/09/01/ 2016;111:592-599. doi:https://doi.org/10.1016/j.phrs.2016.07.015
- Seaton KE, Yunda Huang P, Karuna S, et al. Pharmacokinetic serum concentrations of VRC01 correlate with prevention of HIV-1 acquisition. EBioMedicine. 2023;93:104590. doi:https://doi.org/10.1016/j.ebiom.2023.104590
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
There are a few points that we were not able to discuss in the paper due to space limitations, but may be interesting to discuss in this forum. Because the AMP Study only had access to blood, we were unable to explore any mechanisms linking PreP with intestinal permeability, as identified by systemic I-FABP.
However, several studies have assessed the effects of tenofovir in the rectal epithelium. In colorectal epithelial cell lines, tenofovir transiently reduced epithelial monolayer integrity 1. In human rectal explants, 1% tenofovir gel caused epithelial fracture without a decrease in viability1. Rectal biopsies from individuals who received 1% tenofovir gel applied once a day for 7 days had broad changes including less transcription of anti-inflammatory mediators, as well as epithelial cell differentiation and proliferation.2. Lastly, a study of duodenal and rectal biopsies collected before and after 2-month use of oral tenofovir-emtricitabine demonstrated that initiation of PreP upregulates an intestinal type I/III interferon signature in epithelial cells, which is not detected systemically3. They reported no changes in the mRNA expression of IFABP in the intestinal mucosa, but it would be interesting to assess whether the localized inflammation mediates the permeability changes that lead to IFABP detection in blood.
The current study by Huang Y et al reports the unexpected finding that the anti-HIV antibody VRC01 is cleared more quickly from circulation in people taking oral Truvada® (tenofovir/emtricitabine) preexposure prophylaxis (PrEP) than in non-PrEP users. Interestingly, the VRC01 clearance rate positively correlated with serum levels of intestinal fatty acid binding protein (I-FABP), indirectly suggesting some damage to the intestinal mucosa by oral PrEP. Because tenofovir and emtricitabine are also mainstay components of combination antiretroviral treatment (cART), these data could be important for people living with HIV (PWH) as well. cART has dramatically extended the lifespan of PWH, but they still experience higher morbidity than the general population.1 The reasons for this elevated morbidity remain largely unclear, but it is possible that antiretroviral drugs contribute to the etiology of HIV-associated co-morbidities.
That tenofovir/emtricitabine may cause some undesired changes in the gut is supported by our finding that oral Truvada PrEP induces type I/III interferon (IFN-I/III) pathway activation in the duodenal and rectal mucosa.2 Chronic IFN stimulation is known to have some deleterious effects on health.3 In the current and in our prior study, the drugs’ impact was relatively subtle. However, after long periods of treatment, these effects could incrementally contribute to the observed increase of co-morbidities in PWH.
Our prior study may also hold a clue regarding a possible mechanism for faster antibody clearance during oral PrEP use. A correlate of increased IFN-I/III pathway activation in the intestinal mucosa was an increase in the frequency of microfold or M cells in the columnar epithelium.2 M cells are immunologically hyperactive enterocytes, which constitutively express high levels of interferon-stimulated genes (ISGs). They are also primary transporter cells of macromolecules across the intestinal epithelium, which includes shuttling of antigens and antiviral antibodies from and to the intestinal lumen.4,5 The close relationship between M cells and humoral immunity is highlighted by the pairing of nearly every M cell with a B lymphocyte in its basolateral groove. Thus, it stands to reason to hypothesize that a higher number of M cells, as occurs with oral PrEP use, leads to increased shuttling of VRC01 antibody towards the lumen, thereby enhancing its clearance rate.
How this hypothesis ties in with the systemic increase of I-FABP in the current study remains to be determined. We have intestinal biopsies stored from our oral PrEP trial in Seattle (ACTU-3500, NCT02621242), with the duodenal tissues likely being the only ones in the world taken from such a cohort. According to the Human Protein Atlas, I-FABP is much more abundant in the small intestine than in the colorectum. As a first step, these specimens could be studied for I-FABP expression and distribution in oral PrEP users in comparison to their own pre-PrEP biopsies.
Two additional articles are of interest in light of the current report. Murata K et al showed that tenofovir suppresses the anti-inflammatory cytokine interleukin 10 (IL-10) and induces the pro-inflammatory cytokine interleukin 12 (IL-12) in peripheral blood monocytes, but only upon stimulation with bacterial lipopolysaccharide.6 This result might help explain why tenofovir’s (or oral PrEP’s) in vivo effect seems most pronounced in the gastrointestinal tract, where LPS levels are high.
The second article pertains to the mechanism of how these drugs might initiate the reported immunological changes. Rajurkar M et al studied 3TC (lamivudine), which is a drug of the same class as tenofovir and emtricitabine (nucleoside/nucleotide analogue reverse transcriptase inhibitor [NRTI]). Lamivudine inhibited reverse transcription of repeat RNA species such as long interspersed nuclear element-1 (LINE-1) retrotransposons and human endogenous retroviruses (HERV) in human colorectal cancer cells. By inhibiting reverse transcription of these RNAs, lamivudine caused accumulation of DNA:RNA hybrids, which are immunogenic and linked to the stimulation of interferon-response genes.7
Synthesizing the findings of these two studies, I speculate that LPS may activate endogenous retroelements in enterocytes, and NRTI drugs such as tenofovir and 3TC may inhibit subsequent reverse transcription of the resulting repeat RNA species. Finally, innate immune recognition of the DNA:RNA hybrids stemming from incomplete reverse transcription initiates off-target immunological effects, such as intestinal ISG activation, cytokine production, M cell proliferation, and increased immunoglobulin leakiness.
The views and opinions expressed herein are solely those of the author and do not necessarily reflect those of the other authors of the cited publications.
Florian Hladik, PhD MD Department of Obstetrics and Gynecology University of Washington Seattle, United States
Citations