An unexpectedly simple route to hydrogen
Published in Chemistry, Earth & Environment, and Sustainability
When we started this project, the idea that simple metal ions could function as catalysts for hydrogen evolution had not even crossed our minds. In fact, this work began as a control experiment for a study using coordination complexes with designed ligands. What we observed, however, was something entirely unexpected.
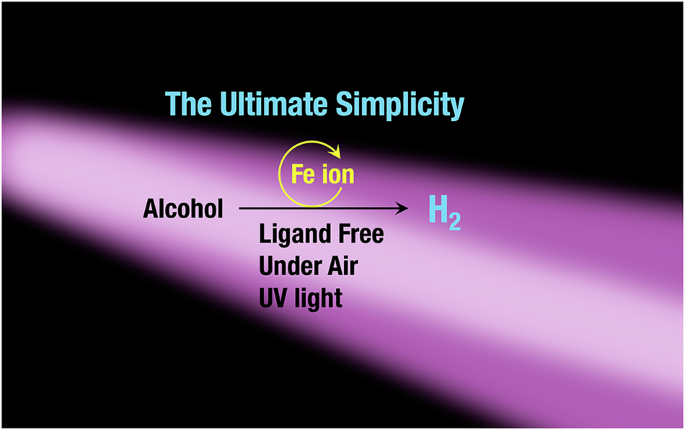
One day, a student in my group came to me with GC data, saying that hydrogen had been detected from an alcohol using only iron(III) chloride under light irradiation. At first, I could hardly believe it. Iron salts are not typically considered as photocatalysts for hydrogen evolution. But once we confirmed the reproducibility of the data, it became clear that something unusual was happening. I remember literally jumping in surprise.
This finding was a classic case of serendipity. A control experiment—often treated as routine—turned out to be the key to uncovering a new type of reactivity. Importantly, the system we discovered does not fit neatly into conventional categories. It is neither a well-defined molecular complex catalyst with elaborate ligands, nor a heterogeneous solid catalyst. Instead, it is an extremely simple, ligand-free system based on iron ions in solution. This simplicity, while exciting, also makes mechanistic understanding particularly challenging, as this type of system does not fall within established categories of catalysis and lacks clear precedents.
Indeed, elucidating the mechanism of this reaction has proven to be one of the most difficult aspects of the study. Without prior examples to guide our interpretation, many of the standard approaches in catalysis do not directly apply. The reaction appears to involve photoinduced processes, but a full picture remains elusive. We believe that this system may represent a new regime of catalytic chemistry, one that requires us to rethink how we conceptualize catalysis.
What makes this discovery particularly compelling is the nature of iron itself. Iron is one of the most abundant elements on Earth. It is inexpensive, widely available, and environmentally benign compared to many precious or rare metals commonly used in catalysis. The fact that such a simple iron salt can promote hydrogen evolution from alcohols under light irradiation raises an intriguing possibility: if such processes can occur under these minimal conditions, could similar reactions already be happening naturally, in environments around us?
This idea may sound speculative, but it highlights an important point. Chemistry does not only occur in carefully designed laboratory systems. If a reaction is sufficiently simple and relies on ubiquitous components—light, oxygen, alcohols, and iron ions—it invites us to consider its broader relevance beyond the laboratory.
On a personal level, I have always been drawn to simple catalytic systems. There is something fundamentally appealing about achieving complex transformations with minimal components. In modern catalysis, there is often a tendency toward increasing complexity—more sophisticated ligands, more controlled environments, and more intricate designs. While these approaches are powerful, our work suggests that simplicity itself can open new directions.
When I first became convinced that this reaction was real and reproducible, I felt that it could represent a significant turning point. Perhaps even a once-in-a-generation discovery. When I shared these results with fellow researchers, however, not everyone immediately appreciated their significance or found them compelling. I came to feel that truly unconventional breakthroughs arising from discontinuous ideas can be difficult to grasp, even for researchers who value originality. Of course, such statements must be made with caution, and much work remains to fully understand and develop this system. However, the experimental fact remains: hydrogen can be generated from alcohols using one of the simplest and most abundant metal ions under light.
If this finding can be further developed and generalized, it may have implications not only for catalysis but also for how we think about sustainable energy and resource cycles. The possibility of generating hydrogen using earth-abundant, low-cost materials under mild conditions is an attractive vision.
Looking ahead, our immediate goal is to better understand the mechanism of this reaction. We hope that a combination of experimental and theoretical studies will shed light on the key intermediates and pathways involved. At the same time, we are exploring ways to improve the efficiency and expand the scope of the system.
This work reminds us that important discoveries can still emerge from the simplest experiments. Sometimes, by paying close attention to unexpected results, we can uncover entirely new directions in chemistry.
Follow the Topic
-
Communications Chemistry

An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
What are SDG Topics?
An introduction to Sustainable Development Goals (SDGs) Topics and their role in highlighting sustainable development research.
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Sustainable waste management through polymer upcycling
Publishing Model: Open Access
Deadline: May 30, 2026
Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in