Antheridium development in fern gametophytes
Published in Protocols & Methods, Plant Science, and Anatomy & Physiology
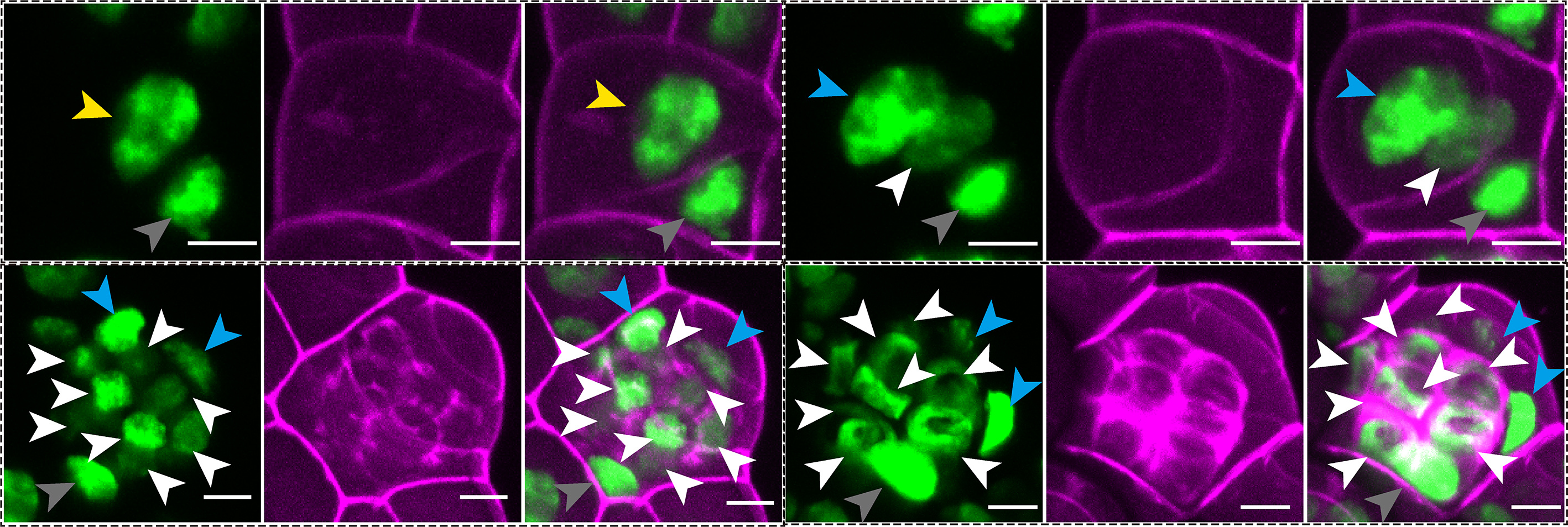
Ferns, which do not produce flowers or seeds, reproduce by spores. Unlike seed plants, ferns use a different strategy for sexual reproduction, producing motile sperm during the gametophyte phase that rely on water for fertilization.
In this study, we examine the cellular basis of the dynamic processes by which ferns make sperm-producing organs. Using non-invasive time-lapse imaging of a GFP reporter that labels every nucleus, we followed how male reproductive structures, called antheridia, initiate and develop at single-cell resolution in the model fern Ceratopteris richardii. Rather than relying on static snapshots, this approach, combined with three-dimensional computational analysis, allowed us to reconstruct detailed lineage maps, visualized as family trees of cells, showing how individual cells divide and contribute to the formation of sperm-producing antheridia. These lineage maps show that antheridium development follows a highly organized sequence of events. It begins with two rounds of asymmetric cell divisions, in which the two daughter cells differ in identity and morphology. These early steps establish two distinct cell populations with different cell fates and behaviors. One group, the spermatogenous cell lineage, is responsible for producing sperm. These cells divide continuously and in a largely synchronized manner, producing a cluster of cells within the antheridium that eventually differentiate into motile, flagellated sperm. Upon maturation, the antheridium ruptures and releases these sperm for fertilization. The other group forms the sterile lineage. These cells divide only a limited number of times and instead build the supporting structure of the antheridium. They form the outer layers of an antheridium, referred to as cap and ring cells, which surround the spermatogenous cells.
Beyond reconstructing how a complex, 3D antheridium forms from a single cell, we also examined how this process is regulated. Previous studies showed that a pheromone called antheridiogen promotes antheridium formation, while another plant hormone, abscisic acid (ABA), has the opposite effect. However, how these signals influence cell behavior during antheridium initiation and development was not clear. By combining time-lapse live imaging with quantitative analysis under different treatments, we were able to directly observe how these hormones affect cell division and cell fate in real time. We found that antheridiogen induces the formation of new antheridia, while ABA suppresses the initiation of new antheridia and strongly reduces cell proliferation within developing antheridia, especially in the spermatogenous lineage. These results provide a cellular explanation for how these two signals act in opposition to regulate male reproductive development. Taken together, our work shows how a combination of precise cell division patterns and hormone signaling coordinates the formation of a functional reproductive organ. More broadly, these findings provide insight into how plants control cell fate and cell proliferation in response to internal and external signals, and how these mechanisms may have evolved across different plant lineages.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
DNA repair and human disease
Publishing Model: Hybrid
Deadline: Oct 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in