Atomic-Site Control Unlocks Durable Acidic OER in Ru–Co₃O₄
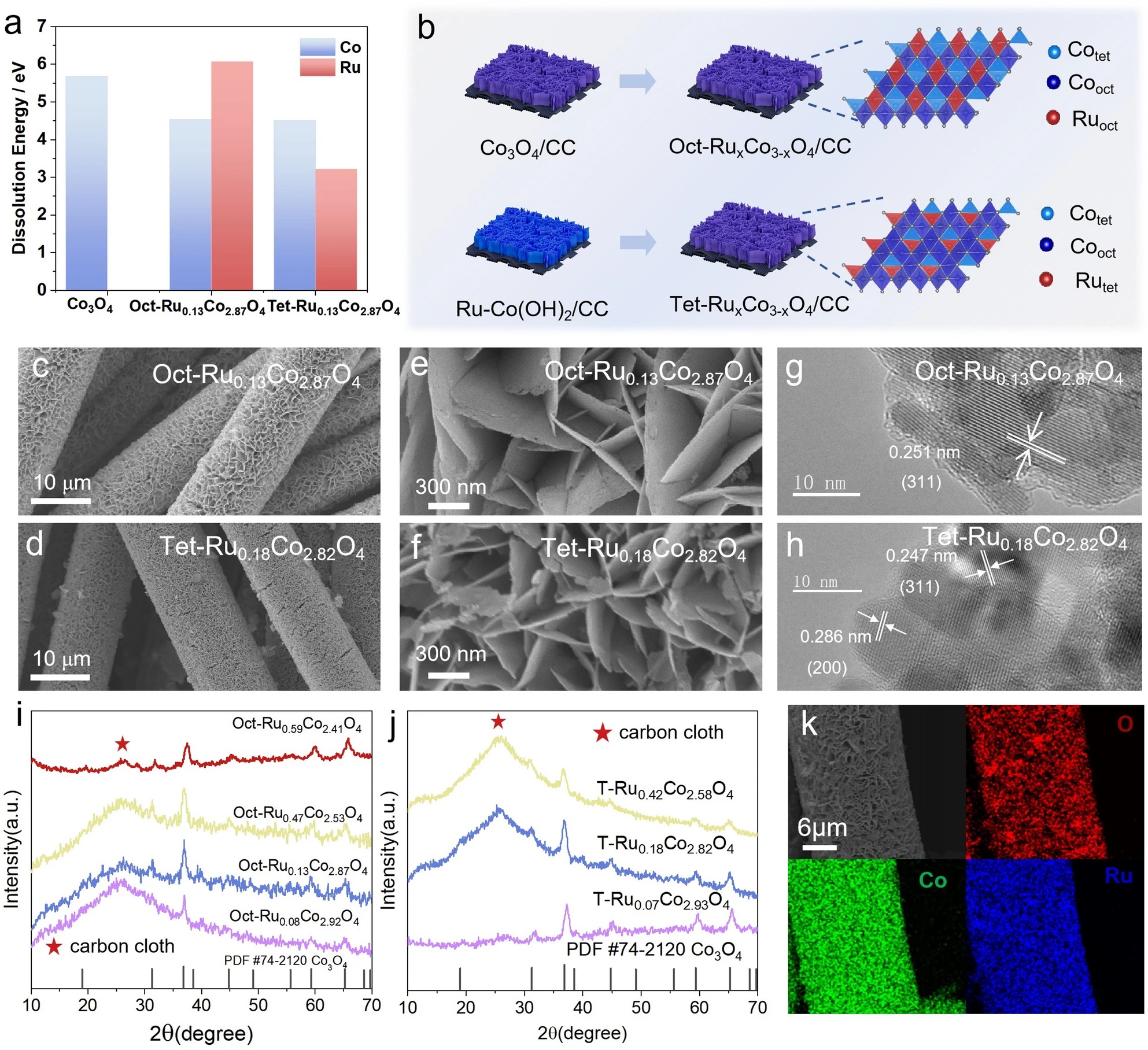
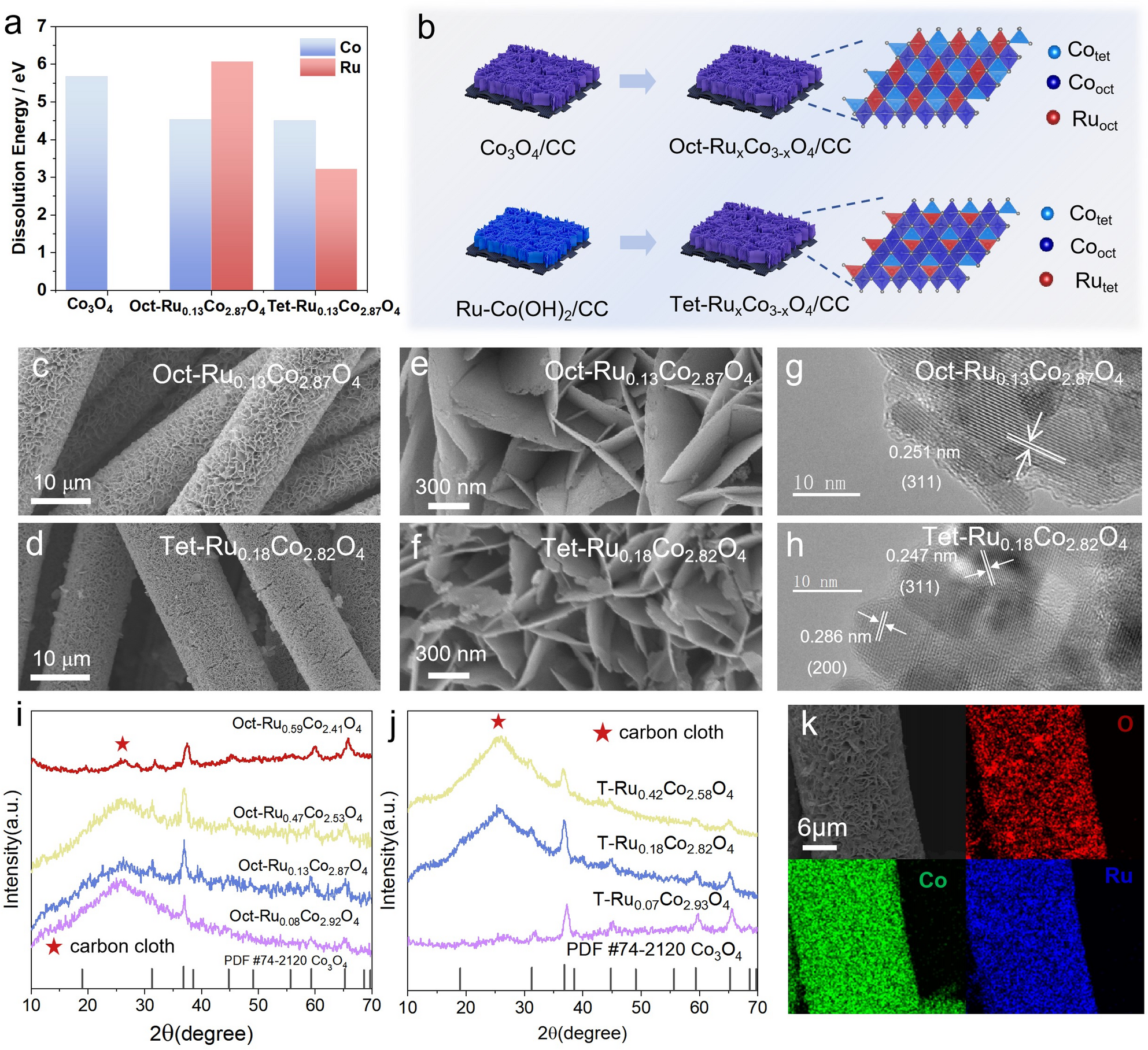
In this work, we demonstrate that crystallographic site control of ruthenium in spinel Co₃O₄ is decisive for efficient and durable acidic oxygen evolution. Selective octahedral Ru substitution forms a cooperative Ruₒct–O–Coₒct active motif that activates the oxide-path mechanism, suppresses cobalt over-oxidation, and enables stable OER performance with only minimal Ru content. The optimized Oct-Ru₀.₁₃Co₂.₈₇O₄ catalyst therefore delivers low overpotential and long-term stability, highlighting site-specific engineering as a practical strategy for low-precious-metal PEM electrolysis anodes.
Key Insights
- Site-selective Ru doping in spinel Co₃O₄ enables direct comparison between octahedral (Co³⁺) and tetrahedral (Co²⁺) substitution.
- Octahedral Ru incorporation creates a synergistic Ruₒct–O–Coₒct dual-metal active center, enabling the oxide-path mechanism with a reduced reaction barrier.
- The optimized Oct-Ru₀.₁₃Co₂.₈₇O₄ achieves a low overpotential of 240 mV at 10 mA cm⁻² and remains stable for over 240 h in acidic electrolyte.
- Tetrahedral Ru substitution disrupts orbital overlap, leading to higher overpotential and rapid degradation.
- Electron transfer within the Ru–O–Co framework suppresses cobalt over-oxidation and dissolution, accounting for the enhanced durability.
Why It Matters
- Identifies octahedral Co³⁺ sites as the intrinsic active centers for acidic OER in spinel Co₃O₄.
- Demonstrates that high activity and durability can be achieved with only ~4 at% Ru, substantially reducing noble-metal demand.
- Establishes crystallographic site engineering as a general, scalable design principle for next-generation PEM water-electrolysis anodes.
Authors, Corresponding Authors, and Affiliations
Authors: Yumeng Wang, Yaling Zhang, Shiyao Chen, Yameng Fan, Jian Peng, Tofik Ahmed Shifa, Alberto Vomiero, Yaping Li*, Fengmei Wang*, and Xiaoming Sun* (corresponding authors).
Corresponding Authors (Emails):
- Yaping Li — liyp@mail.buct.edu.cn
- Fengmei Wang — wangfm@buct.edu.cn
- Xiaoming Sun — sunxm@mail.buct.edu.cn
Affiliations: State Key Laboratory of Chemical Resource Engineering, College of Chemistry, Beijing University of Chemical Technology, Beijing, China; School of Science, RMIT University, Melbourne, Australia; Eastern Institute for Advanced Study, Eastern Institute of Technology, Ningbo, China; Department of Molecular Sciences and Nanosystems, Ca’ Foscari University of Venice, Venice, Italy.
How to Cite
Wang, Y., Zhang, Y., Chen, S. et al. Site-selective Ru doping in spinel Co3O4 unlocks dual-site synergy for acidic water electrolysis. Catal 2, 4 (2026). https://doi.org/10.1007/s44422-026-00018-w
Follow the Topic
-
Catal
Catal is an open access journal covering full spectrum of catalysis critical advances. From biocatalysts to heterogeneous catalysts, it integrates fundamental and applied sciences. Catal offers a primary platform for researchers and practitioners in the field.
Related Collections
With Collections, you can get published faster and increase your visibility.
National Catalysis Conference 2025
The 22nd National Catalysis Conference, held in December 2025 in Xiamen, China, stands as the largest national academic event in the field of catalysis, bringing together leading scientists from academia and industry to discuss frontier research and emerging technologies across catalytic science.
In collaboration with several internationally recognized researchers from Xiamen University - the main organizer of the conference - Catal will publish a Special Issue featuring selected contributions from the meeting. This joint effort ensures high academic standards and showcases the scientific breadth and excellence presented at the conference.
Potential topics include, but are not limited to:
• Catalytic materials
• Thermocatalysis
• Electrocatalysis
• Biomass and CO₂ conversion
• Industrial catalysis
• Theoretical and computational catalysis
Publishing Model: Open Access
Deadline: Dec 31, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in