Boosting durability of Fe–N–C electrocatalysts for PEM fuel cells via dicyandiamide modification
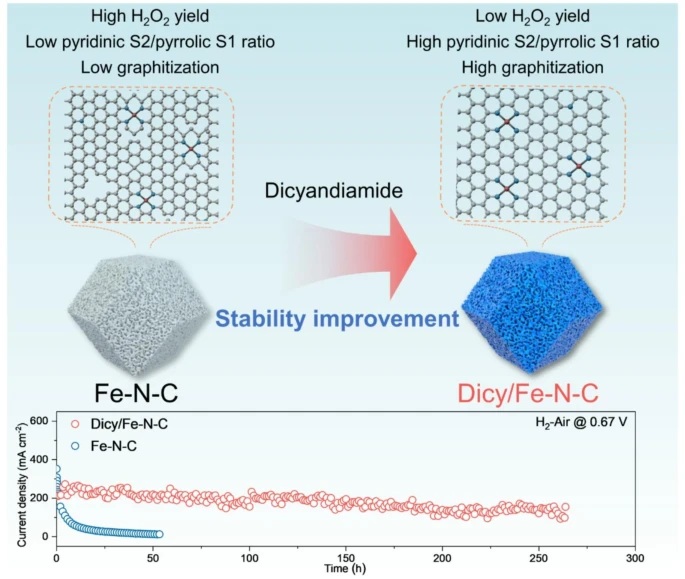
In this work, we report a dicyandiamide (DCDA)-modified Fe–N–C electrocatalyst (Dicy/Fe–N–C) that effectively addresses the long-standing durability challenges of platinum-group-metal-free catalysts in proton exchange membrane fuel cells (PEMFCs). By introducing DCDA during pyrolysis, we simultaneously regulate the coordination environment of Fe active sites and reinforce the carbon support structure. As a result, the optimized catalyst exhibits enhanced resistance to demetallation, carbon corrosion, and peroxide-induced degradation, while maintaining competitive oxygen reduction reaction (ORR) activity.
Key Insights
-
Dicyandiamide promotes formation of stable active sites
We find that DCDA modification effectively increases the proportion of pyridinic FeN₄ sites, which possess higher energy barriers against demetallation and radical attack, thereby improving intrinsic catalyst stability. -
Graphitized carbon support enhances durability
The incorporation of DCDA leads to a more graphitized carbon framework, which improves resistance to electrochemical corrosion and helps preserve active site integrity during long-term operation. -
Suppression of H₂O₂ formation mitigates degradation pathways
The Dicy/Fe–N–C catalyst favors a four-electron ORR pathway, resulting in reduced hydrogen peroxide generation and minimizing the formation of harmful reactive oxygen species. -
Excellent durability demonstrated under both RDE and PEMFC conditions
We observe minimal performance decay after extended cycling, with stable operation up to 264 h in PEMFC tests and limited power density loss after 30,000 cycles, confirming the robustness of the catalyst under realistic conditions. -
Performance meets and surpasses DOE targets
The Dicy/Fe–N–C-based membrane electrode assembly delivers a current density of 50.4 mA cm⁻² at 0.9 V, exceeding the U.S. DOE 2025 target for PGM-free catalysts.
Significance of This Work
This study demonstrates that a simple dicyandiamide modification strategy can simultaneously optimize active site structure and carbon support properties in Fe–N–C catalysts. By mitigating key degradation mechanisms—including Fe site demetallation, carbon corrosion, and peroxide attack—we provide a viable pathway toward durable and scalable platinum-free electrocatalysts. We anticipate that this strategy will accelerate the practical deployment of PEM fuel cells in sustainable energy systems.
Authors & Affiliations
Xu Lin†, Zhankuan Lu†, Luojie Zhao†, Mengting Han, Yuting Wang, Shiqing Huang,
Hao Ling*, Yan Huang*, Jimmy Yun, Dapeng Cao*
† These authors contributed equally.
Affiliations:
State Key Laboratory of Organic-Inorganic Composites, Beijing University of Chemical Technology, Beijing, China
College of Chemical Engineering, Xiangtan University, Xiangtan, China
Qingdao International Academician Park Research Institute, Qingdao, China
School of Chemical Engineering, The University of New South Wales, Sydney, Australia
Corresponding Authors:
Hao Ling (haoling@xtu.edu.cn)
Yan Huang (huangyan@buct.edu.cn)
Dapeng Cao (caodp@buct.edu.cn)
How to Cite This Article
Lin, X.; Lu, Z.; Zhao, L.; Han, M.; Wang, Y.; Huang, S.; Ling, H.; Huang, Y.; Yun, J.; Cao, D. (2026).
Boosting the durability of Fe-N-C electrocatalysts for PEM fuel cells by dicyandiamide modification strategy.
Catal, 2, 12. https://doi.org/10.1007/s44422-026-00023-z
Follow the Topic
-
Catal
Catal is an open access journal covering full spectrum of catalysis critical advances. From biocatalysts to heterogeneous catalysts, it integrates fundamental and applied sciences. Catal offers a primary platform for researchers and practitioners in the field.
Related Collections
With Collections, you can get published faster and increase your visibility.
National Catalysis Conference 2025
The 22nd National Catalysis Conference, held in December 2025 in Xiamen, China, stands as the largest national academic event in the field of catalysis, bringing together leading scientists from academia and industry to discuss frontier research and emerging technologies across catalytic science.
In collaboration with several internationally recognized researchers from Xiamen University - the main organizer of the conference - Catal will publish a Special Issue featuring selected contributions from the meeting. This joint effort ensures high academic standards and showcases the scientific breadth and excellence presented at the conference.
Potential topics include, but are not limited to:
• Catalytic materials
• Thermocatalysis
• Electrocatalysis
• Biomass and CO₂ conversion
• Industrial catalysis
• Theoretical and computational catalysis
Publishing Model: Open Access
Deadline: Dec 31, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in