Atrial Fibrillation in Rodents: Time to Raise the Standards?
Published in General & Internal Medicine, Zoology & Veterinary Science, and Anatomy & Physiology
Atrial fibrillation (AF) is a major medical challenge, affecting millions worldwide and significantly increasing the risk of stroke, heart failure, and other cardiovascular complications. While large animal models and human studies provide invaluable insights, rodent models have become increasingly popular in AF research due to their genetic manipulability, cost-effectiveness, and the capacity of clinically relevant stimuli to promote AF susceptibility. However, there are critical limitations that hinder the reliability and reproducibility of rodent AF studies, and our recently published paper in Lab Animal seeks to address some of these issues.
The Challenges in Rodent AF Research
Three key limitations characterize most AF studies in rodents:
- The Role of Anesthesia – Many studies are conducted under anesthesia, but the specific type and dosage are often insufficiently reported. Anesthetic agents have profound electrophysiological effects that can influence arrhythmia susceptibility and properties, yet these factors are frequently overlooked.
- Signal Resolution and Rhythm Classification – Atrial recordings often suffer from poor signal resolution, making it difficult to distinguish between regular and irregular rhythms. Consequently, researchers may misclassify arrhythmic events, leading to inaccurate conclusions.
- Variability in Burst Pacing Protocols – The methods used to induce AF vary widely, with many studies providing limited details on stimulation parameters. This lack of standardization hampers data comparability and reproducibility.
Our Contribution: A Novel Approach to Rodent AF Research
Over the past decade, our team has developed an implanted device with specialized atrial electrodes, allowing us to study AF in conscious rodents. This system offers several distinct advantages:
- Eliminates the confounding effects of anesthesia, enabling a more accurate representation of intrinsic electrophysiological properties.
- Achieves high-resolution atrial recordings, allowing clear differentiation between regular and irregular rhythms.
- Standardizes burst pacing protocols, ensuring reproducibility and consistency across experiments.
Key Findings: The Complex Influence of Anesthesia
In our study, we compared AF susceptibility and arrhythmic properties in anaesthetized rodents versus those under two commonly used anesthetics. Our results reveal a complex and nuanced picture:
- Anesthesia significantly alters AF susceptibility and arrhythmic dynamics, making data interpretation challenging.
- Sex differences play a crucial role, with males and females responding differently to anesthetic agents.
- Isoflurane, the most widely used anesthetic in rodent AF research, exerts profound effects, transforming irregular atrial rhythms into regular supraventricular rhythms in male rats. (This can be detected however, only with appropriate signal resolution of the atrial signal, as we could routinely obtain with our methodology)
Implications for Future Research
Our findings underscore the need for a paradigm shift in rodent AF research. We demonstrate that high-quality, reproducible AF research can be conducted in unaesthetized rodents, bypassing the confounding effects of anesthesia. We hope that our approach will inspire others to develop similar systems and refine methodologies for studying AF in rodent models (including mice). Alternatively, for studies that must rely on anesthesia, it is imperative to rigorously characterize and report the electrophysiological effects of anesthetic agents to improve data interpretation.
Acknowledgments
This study was made possible through the dedication of our team, and I would like to extend my sincere gratitude to all contributors. In particular, I wish to thank Michael Murninkas and Or Levi, two exceptionally talented MD-PhD students whose expertise and commitment were instrumental in driving this research forward.
We believe that by addressing these fundamental challenges, we can enhance the reliability of rodent AF models and ultimately contribute to a deeper understanding of this complex arrhythmia.
Follow the Topic
-
Lab Animal
In vivo studies with model organisms are critical for uncovering the complex interactions between genes, cells, organisms and their environment. As a Springer Nature journal, this journal is dedicated to publishing the latest innovations in in vivo science & technology.
Your space to connect: The Nitric oxide signalling in cardiovascular health and disease Hub
A new Communities’ space to connect, collaborate, and explore research on Cardiovascular Physiology, Clinical Medicine, and Diseases!
Continue reading announcement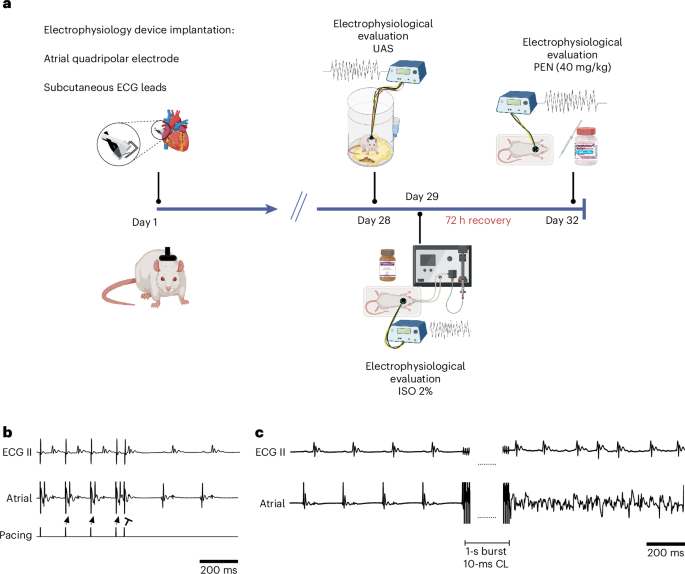

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
📢Publication alert: new paper from our lab in Europace📢
We are pleased to share our latest work examining the effects of semaglutide and colchicine on atrial remodeling in a post-myocardial infarction model with reduced ejection fraction. In this study, we demonstrate that both agents attenuate key components of the atrial fibrillation substrate, with distinct effects on electrical, structural, and inflammatory remodeling pathways. These findings further support the concept that targeting atrial cardiomyopathy, rather than the arrhythmia itself, may represent an effective therapeutic strategy.
A central aspect of this work is the use of our implantable atrial pacing and recording system in freely moving rats. This platform allows longitudinal, high-resolution electrophysiological assessment in the unanesthetized state, enabling repeated measurements of atrial function over time. The ability to track dynamic changes in AF substrate within the same animal provided a level of precision and physiological relevance that would be difficult to achieve in rodents with conventional approaches.
We believe that this technological capability was critical for enabling these findings and several additional findings from our recent publications. More broadly, after years of development and refinement, this platform represents a step toward a new experimental paradigm for studying atrial fibrillation in rodents, with clear advantages in the reliability, longitudinal resolution, and physiological relevance of in vivo electrophysiological data.
Full paper:
https://lnkd.in/dRarwcfA