Behind the Degradation of Si-based Li-Ion Batteries – A Story about Solid Electrolyte Interface Reformation
Published in Materials

Silicon (Si)-based anodes represent one of the most promising approaches towards high-energy Lithium (Li)-ion batteries for renewable energy, e-mobility, or mobile electronic devices. However, high-density silicon composite anodes show large volume changes of about 300% upon charging/discharging. The volume rearrangement leads to mechanically induced cracking and gradual degradation of the active silicon material.
In recent years several strategies have been developed to tackle the challenges with respect to the volume expansion of Si and associated issues leading to capacity fading. Nevertheless, upscaling to an industrial setting remains challenging due to the lack of a complete understanding of the underlying structural and chemical degradation mechanism in particularly at the active material interface.
Si-based anode materials conventionally show a porous matrix, graphite particles as well as the Si-based active material embedded in the carbon binder domain (CBD). The cycling stability is triggered by the interaction of the active material with the fluorine-based electrolyte. A so-called solid interlayer between the active material and the CBD is formed. This interlayer, formed within a single lithiation step, is also referred to the solid electrolyte interface (SEI).
Experimental proof is still lacking on both the structural and chemical evolution at the Si interface upon cycling for a full-cell configuration. However, such an understanding is crucial to further improve the anode material architecture especially suitable for industrial up-scaling.
An industrial relevant anode covers different length scales ranging from the mm-range, covering the whole electrode, to the anode microstructure and ultimately to the nm-range incorporating e.g. the immediate vicinity of the Si-domains or the single Si-particles.
The Team including the Materials Center Leoben Forschung GmbH, Carl Zeiss Microscopy GmbH, Varta Innovation and the University of Warwick, show for industrial relevant Si-based anodes the critical role of the interface formation up to high cycling numbers, which is strongly different to graphite-based anodes.
A correlated workflow incorporating X-ray microscopy, field-emission scanning electron microscopy tomography, elemental imaging and deep learning-based microstructure quantification is utilized, suitable to witness the structural and chemical progression of the silicon and SEI reformation upon cycling. The experimental results illustrate that the initially formed nm-sized SEI layer evolves into a micron-sized silicon electrolyte composite (SEC) like structure at prolonged cycles.
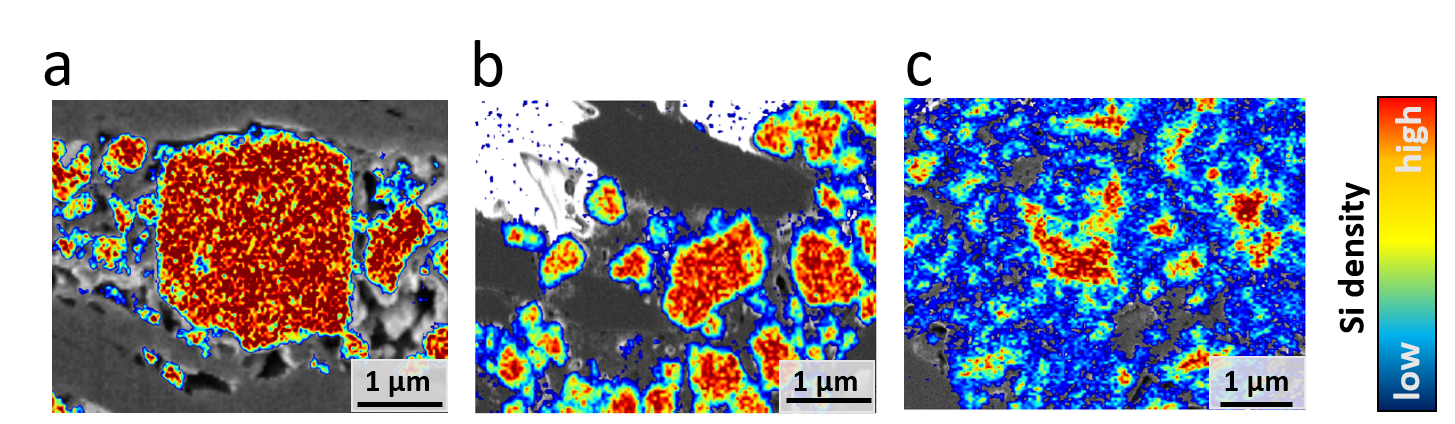
The findings are linked with experimental-informed electrochemical modelling. The modelling endorses an underutilization of the active material due to the reformation of the SEI resulting in the SEC growth and affecting the capacity. Further, a chemo-mechanical model is used to analyze the stability of the SEI/silicon reaction front and to investigate the effects of material properties on the stability that can affect the capacity loss.
We show that the SEC formation has direct consequences on Si-degradation, Li-trapping and transport as well as can lead to the emergence and growth of stress concentration and damage due its instable nature.
As discussed in biological materials1 the differences in the Young’s moduli between different material layers help stabilizing the system with stopping cracks from forming or regulating the build-up of new layers. That is, the Young´s moduli ratio, between materials present in the proximity of the reaction interface, may become a crucial parameter to consider when trying to enhance the anode performance.
We argue that the key for improved Si-based anodes is a proper design of the active material and its structure e.g. utilizing Si-alloys or Si-compounds, including the consideration of underlaying material parameters e.g. the Young’s modulus etc. .
A well-designed active material will help to control the interface kinetics during lithiation, stabilize the SEI formation and diminish its reformation as well as will limit the extensive spatial SEC growth.
Reference:
1. Fratzl, P. & Weinkamer, R. Nature’s hierarchical materials. Prog. Mater. Sci. 52, 1263–1334 (2007).
A link to the manuscript can be found here:
Follow the Topic
-
Communications Materials

A selective open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of materials science.
Related Collections
With Collections, you can get published faster and increase your visibility.
Materials for quantum sensing and computing
Publishing Model: Open Access
Deadline: Jul 09, 2026
Triboelectric nanogenerators for energy harvesting
Publishing Model: Open Access
Deadline: Jun 30, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in