Behind the learning curve: bringing AI-assisted fluid management into real-world oncologic surgery
Published in Computational Sciences and Pharmacy & Pharmacology
In perioperative medicine, artificial intelligence is often discussed in terms of innovation, predictive performance and future potential. But in daily clinical practice, an equally important question is what happens when these tools actually enter the operating room. How are they used? How quickly do clinicians become familiar with them? And does their value change over time as confidence grows?
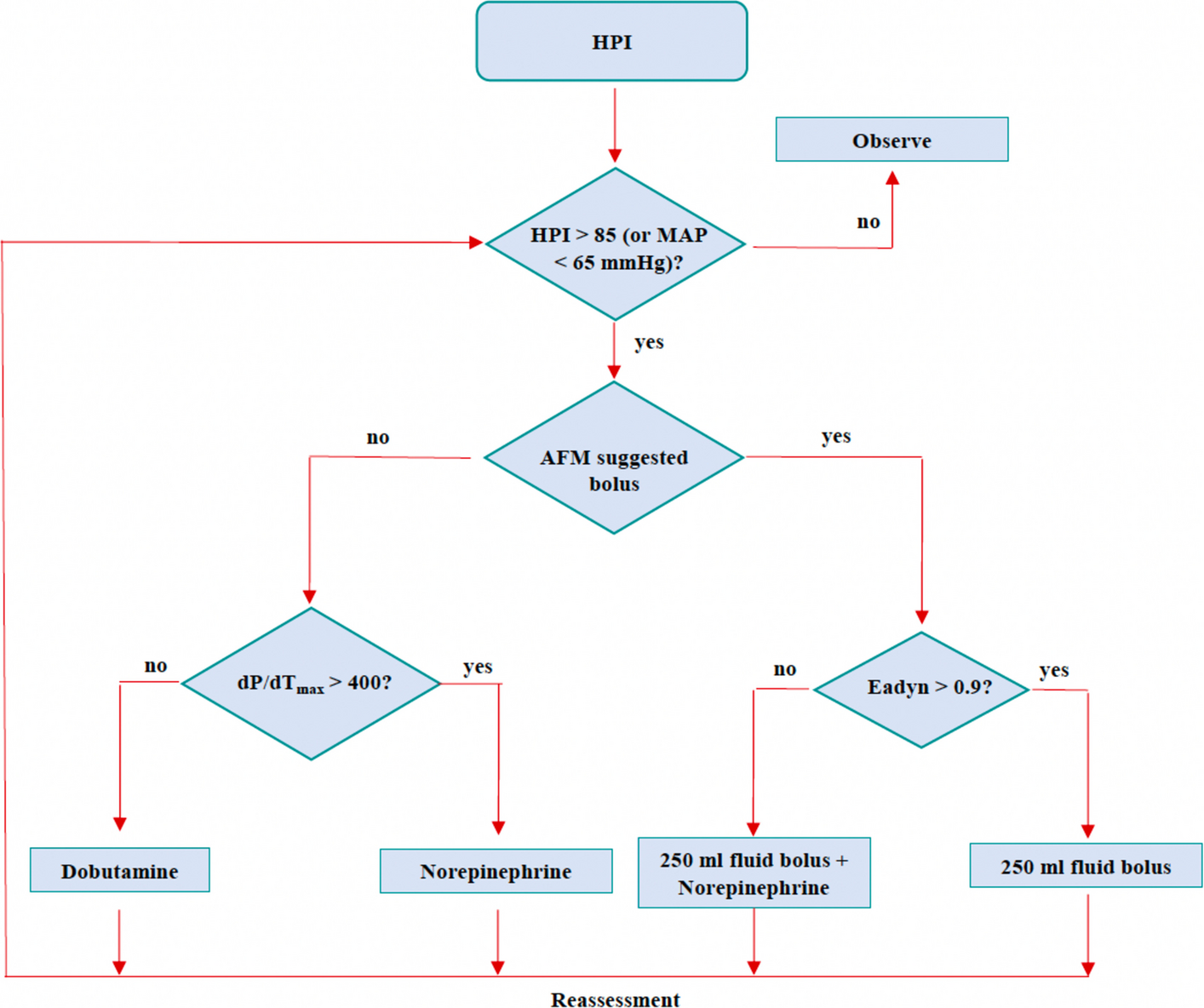
These were the questions behind our study on Assisted Fluid Management (AFM), an artificial intelligence–based decision support system designed to guide intraoperative fluid challenges using real-time stroke volume monitoring. We were interested not only in the technology itself, but in its implementation in routine care.
This question felt especially relevant in major abdominal oncologic surgery, where fluid therapy is a continuous balancing act. Too little fluid may compromise tissue perfusion, while too much may contribute to postoperative complications. In this setting, clinicians are constantly making time-sensitive decisions based on physiology, surgical conditions and patient-specific factors. A decision-support system may help structure these choices, but only if it is integrated into real clinical workflow in a meaningful way.
Our study was a retrospective observational analysis conducted in a high-volume tertiary referral center for abdominal oncologic surgery. We included adult patients monitored with AFM between February 2024 and March 2025 and compared two consecutive implementation periods. Rather than focusing on postoperative outcomes, we chose to examine the implementation process itself. We analyzed each fluid challenge at the bolus level, looking at whether it was initiated by the clinician or suggested by AFM, and whether it generated an effective stroke volume response.
What we found was, for us, the most interesting part of the story. Over time, clinician-initiated boluses decreased, while AFM-suggested fluid challenges increased. At the same time, the physiological effectiveness of AFM-suggested boluses improved, as did the overall effectiveness of fluid challenges. In total, the study included 59 patients and 404 fluid challenges, and the second implementation period showed a pattern consistent with increasing familiarity, confidence and more targeted use of the system.
This is why we believe the “learning curve” is central to the discussion around AI in anesthesiology. A tool like AFM is not simply switched on and instantly absorbed into practice. Its role evolves. Clinicians learn when to trust its suggestions, how to interpret them in context, and how to integrate them into their own judgment. In this sense, implementation is not a side issue; it is part of the technology’s real-world performance.
For us, this study also speaks to a broader issue in healthcare innovation. AI systems should not be evaluated only under ideal or early conditions. They should also be studied as they are adopted in practice, because maturity of use may shape results. Early implementation may reflect caution, novelty or incomplete integration, whereas later phases may better capture how a system truly functions in routine care.
At the same time, our findings should be interpreted within the limits of the study design. This was a retrospective, single-center observational analysis, and it was not designed to determine whether these implementation-related changes improve postoperative outcomes. That remains the next key question.
Still, what this work offers is a practical perspective on how AI enters clinical care: not as an abrupt transformation, but as a gradual process of adaptation between clinicians, workflow and technology. In perioperative medicine, that process may matter just as much as the algorithm itself.
Follow the Topic
-
Journal of Anesthesia, Analgesia and Critical Care
Journal of Anesthesia, Analgesia and Critical Care is an open access international peer reviewed journal dedicated to emerging trends and highly debated topics in anesthesia, analgesia and critical care.
Related Collections
With Collections, you can get published faster and increase your visibility.
Ultrasound-Guided Fascial Plane Blocks: Current Evidence and Clinical Practice
Journal of Anesthesia, Analgesia and Critical Care is calling for a submission on a Collection “Ultrasound-Guided Fascial Plane Blocks: Current Evidence and Clinical Practice”. This collection would explore the clinical science, technical advances, and translational applications of fascial plane blocks, which have become essential components of modern multimodal analgesia strategies.
The scope of this collection to focuses on well-established techniques such as the Transversus Abdominis Plane (TAP) block, Erector Spinae Plane (ESP) block, Quadratus Lumborum (QL) block, and Parasternal blocks, among others. These blocks are now widely adopted across various surgical specialties—abdominal, thoracic, orthopedic, cardiac—and supported by growing evidence for their safety, efficacy, and opioid-sparing benefits. We invite submission of articles on the following topics:
• Present high-quality original research and systematic reviews on block efficacy, anatomical mechanisms, and safety profiles.
• Highlight ultrasound guidance, catheter-based innovations, and anatomical variants.
• Address comparative studies and integration of these blocks into ERAS protocols.
• Examine their impact on chronic postsurgical pain, recovery times, and opioid minimization.
All submissions in this collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Jul 15, 2026
The Geriatric Patient Across the Perioperative and Critical Care Continuum: Frailty, Aging and Personalized Management
Journal of Anesthesia, Analgesia and Critical Care is calling for a submission on a Collection “The Geriatric Patient Across the Perioperative and Critical Care Continuum: Frailty, Aging and Personalized Management”. This collection will explore the growing challenges associated with the care of frail and geriatric patients undergoing surgery or requiring critical care, a population that is rapidly expanding worldwide and increasingly represented in both the perioperative and intensive care settings. Indeed, frailty, multimorbidity, polypharmacy, and diminished physiological reserve substantially influence perioperative risk, postoperative recovery, and long-term outcomes, requiring tailored multidisciplinary approaches to care.
The scope of this Collection focuses on advances in the assessment, risk stratification, and management of frailty across the perioperative and critical care continuum, with particular emphasis on strategies to optimize outcomes in older adults undergoing major surgery or experiencing critical illness.
We invite submissions addressing topics including (but not limited to):
• Frailty assessment tools and risk stratification in anesthesiology, perioperative medicine, and critical care.
• Prehabilitation, geriatric co-management models, and multidisciplinary perioperative pathways designed to improve outcomes in frail surgical patients.
• Perioperative anesthetic strategies, hemodynamic management, and organ protection in elderly and frail individuals.
• Postoperative complications and recovery, including prevention and treatment of delirium, cognitive dysfunction, functional decline, and long-term disability.
• Critical care management of the frail patient, including mechanical ventilation strategies, circulatory support, and decision-making in advanced age.
• Ethical considerations, shared decision-making, and patient-centered outcomes in the care of older adults at heightened perioperative risk, including the application of palliative care.
• Translational and implementation research aimed at integrating frailty-informed approaches into routine perioperative and ICU practice.
This Collection aims to provide clinicians and researchers with an updated and comprehensive overview of frailty-informed perioperative and critical care medicine, highlighting strategies to improve outcomes and quality of care for this vulnerable and increasingly prevalent patient population.
All submissions in this Collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Jan 08, 2027






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in