Behind the paper: Sulfated bile acid produced by a human gut commensal alleviates paediatric sepsis in mice
Published in Biomedical Research
The starting point of this work was not a grand theory, but rather a small and recurring pattern we noticed while processing fecal samples from pediatric patients.
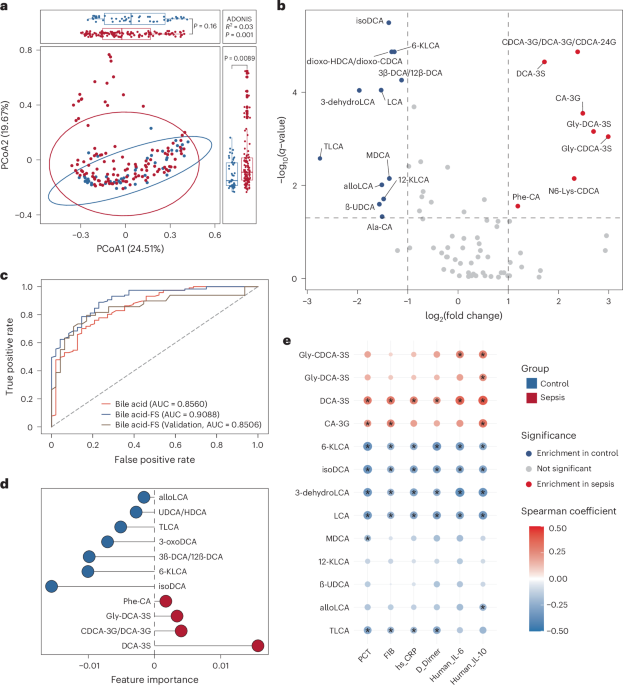
As we analyzed fecal samples from children with sepsis, we gradually observed that changes in the abundance of certain bacterial species seemed to consistently coincide with fluctuations in a specific class of bile acids. What drew our attention most was a sulfated bile acid, deoxycholic acid-3-sulfate (DCA-3S). Its levels changed remarkably in patients. However, when we searched the literature, we found surprisingly few reports describing the direct involvement of gut bacteria in bile acid sulfation. For a long time, bile acid sulfation had largely been regarded as a liver-dominated process. This led us to formulate a bold hypothesis: could gut bacteria themselves possess the ability to sulfate deoxycholic acid?
Once we had this hypothesis, the real challenge began. We screened potentially relevant bacterial strains from patient samples and attempted to validate, in vitro, the associations we had observed in our data analyses. Initially, our experiments were persistently complicated by background interference, making it difficult to determine whether the observed signals were truly reliable. Later, we reintroduced isotope-labeling strategies, which finally allowed us to track the metabolic process more clearly and gradually resolve the previously ambiguous signals. Throughout this process, we continuously cross-referenced our experimental findings with the earlier clinical multi-omics data and repeatedly verified the consistency between them. As the results from both sides became increasingly aligned, we began to realize that this was unlikely to be a coincidental association.
That moment was tremendously exciting for us, because it provided the first in vitro evidence supporting our hypothesis: bacteria were indeed capable of sulfating deoxycholic acid. Yet success in vitro did not necessarily mean the same process occurred in vivo. The subsequent animal experiments prompted us to ask a deeper question: where exactly was this modification primarily taking place? To address this, we introduced antibiotic-treated mouse models and combined them with metabolite analyses across different tissues, aiming to understand the process at a systemic level. During this stage, we again repeatedly compared the animal data with our clinical multi-omics findings. As evidence accumulated, it became increasingly clear that this bile acid modification might be less connected with host metabolism, but was closely associated with the gut microbiota. In particular, after systematically comparing hepatic and intestinal contributions, we gradually arrived at a clearer understanding: the production of DCA-3S does not primarily depend on the liver, but is more likely derived from the sulfation of DCA by intestinal microbiota.
To answer this question more definitively, we ultimately turned to germ-free mouse experiments and carried out key validation studies in a collaborating laboratory. At this stage, we wanted to directly determine whether specific bacterial strains were truly involved in the generation of DCA-3S. To achieve this, we designed a series of corresponding experimental groups, including blank controls, administration of isotope-labeled DCA alone, colonization with E. raffinosus alone, and co-administration of isotope-labeled DCA together with E. raffinosus. Through these carefully controlled comparisons, we were able to directly observe the relationship between the bacterial strain and the metabolite within a simplified experimental system. As the results became clearer, we ultimately confirmed that the corresponding DCA-3S product could be stably detected only when both E. raffinosus and isotope-labeled DCA were present simultaneously. These findings further supported our conclusion that specific gut commensal bacteria are indeed capable of participating in the sulfation of DCA and producing DCA-3S.
Looking back, perhaps the most important aspect of this study was not merely the final demonstration that specific gut commensal bacteria can generate DCA-3S and exert protective effects in sepsis. More importantly, the study taught us that discoveries of this kind rarely emerge from a single experiment. Instead, they require continuous iteration between data analysis and experimental validation. In many cases, clinical multi-omics data first provided the initial clues, which were then progressively tested through in vitro experiments, animal models, and germ-free systems. At the same time, new observations arising from experiments repeatedly pushed us to reinterpret the earlier datasets. Ultimately, the conclusions were established gradually through this constant back-and-forth process.
At the same time, this work also made us more aware of how difficult it can be to challenge a long-accepted “textbook” understanding. What we have demonstrated so far is only that DCA can undergo sulfation by the gut microbiota. Whether other bile acids may undergo similar modifications, whether more complex cooperative interactions exist among different microbial communities, and what physiological significance these modifications may hold in disease contexts all remain important questions worthy of further investigation.
Follow the Topic
-
Nature Microbiology
An online-only monthly journal interested in all aspects of microorganisms, be it their evolution, physiology and cell biology; their interactions with each other, with a host or with an environment; or their societal significance.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in