Beyond the "Love Hormone": How Oxytocin Modulates the Juvenile Brain Differently in Males and Females
Published in Neuroscience
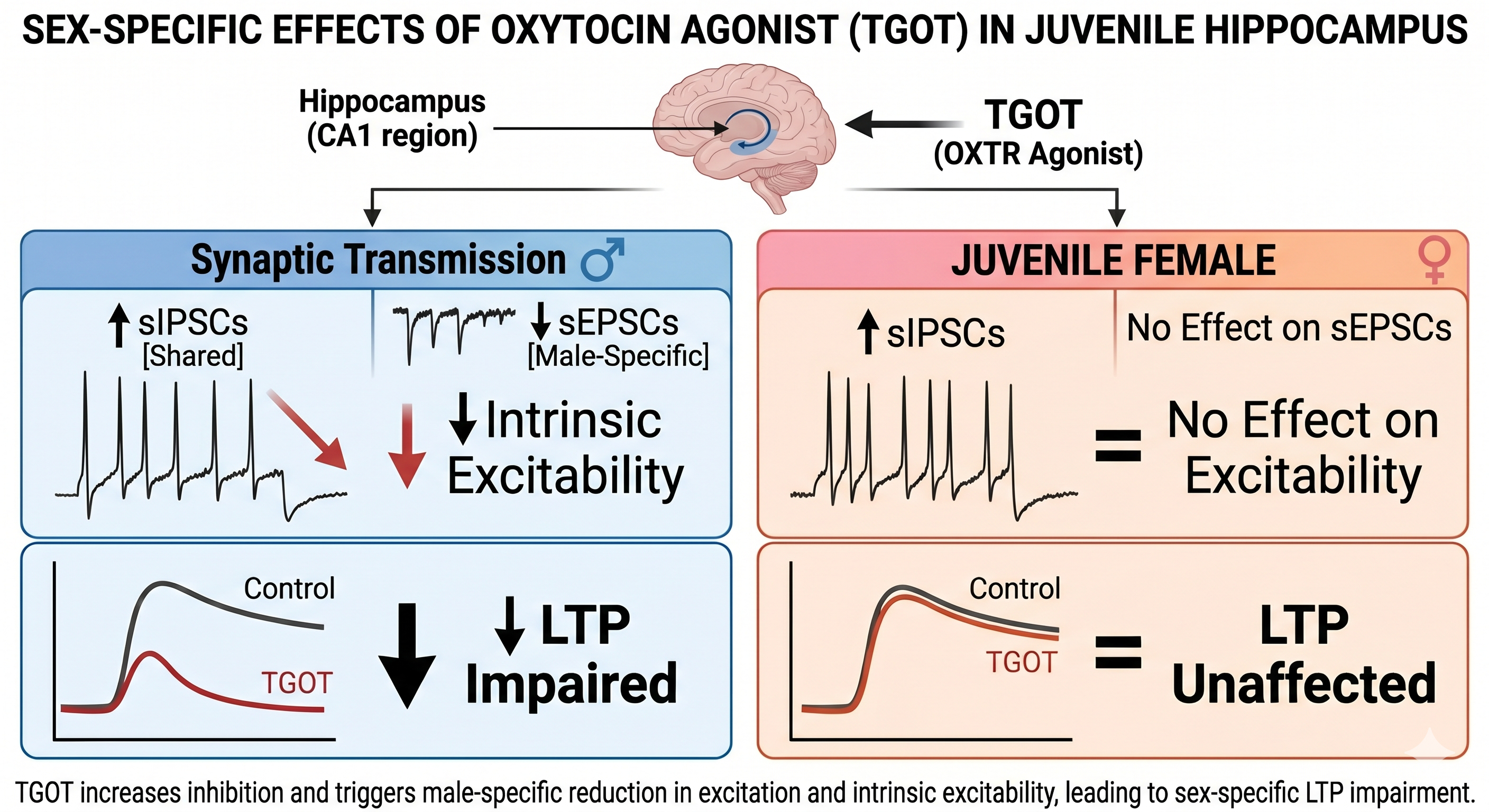
When most people hear "oxytocin," they think of the "love hormone." But looking past the catchy nicknames, this neuropeptide is crucial for shaping the raw electrical machinery of learning and memory. In our latest paper, we discovered that oxytocin does some heavy lifting in the developing brain—and its effects diverge completely depending on biological sex.
To isolate oxytocin's effects without accidentally triggering similar pathways (like vasopressin), we used TGOT, a highly specific synthetic agonist. We focused on the juvenile rat hippocampus—a critical window for brain plasticity—and utilized whole-cell patch-clamp recordings and electrophysiology to see how the neurons responded.
The data told two completely different stories:
- In males: TGOT essentially hit the brakes. It significantly reduced intrinsic excitability, meaning individual neurons were less eager to fire. It decreased spontaneous excitatory currents (sEPSCs) and, crucially, impaired long-term potentiation (LTP)—the fundamental process that strengthens synaptic connections for memory formation in the Schaffer collateral–CA1 pathway.
- In females: Baseline excitability, excitatory currents, and LTP were completely unaffected by the agonist.
Interestingly, TGOT did increase spontaneous inhibitory currents (sIPSCs) in both sexes, but the downstream dampening of plasticity and overall excitability was exclusively seen in the juvenile males.
Why it matters: The juvenile period is a vulnerable neurodevelopmental window, and many psychiatric or developmental conditions have significant sex biases. If the baseline machinery of how the brain wires itself—and how it responds to modulators like oxytocin—is fundamentally different between sexes, their vulnerabilities to these disorders are different, too.
Our findings are a clear reminder: age and biological sex are critical variables, and we can no longer afford to treat male and female brains as identical black boxes in neuroscience. You can read the full study: Oxytocin agonist TGOT differentially modulates synaptic plasticity and intrinsic excitability in a sex-specific manner in the juvenile rat hippocampus.https://www.nature.com/articles/s42003-026-10088-0
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Mechanistic insights into human host and microbiome interactions
Publishing Model: Open Access
Deadline: May 31, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in