
Over the past couple decades, scientists have made huge strides in understanding a new crystal-growth pathway called crystallization by particle attachment (CPA). The idea of CPA is that instead of building a material one atom at a time, entire crystals can snap-together from distinct particles that fuse to create those beautiful hierarchal nanostructures called ‘mesocrystals’, comprised of many sub-crystal domains. It’s a paradigm shift from atom-by-atom assembly, and it’s been an exciting area of research.
But now that scientists can create these structures, we need to know how they behave. What is their chemical stability? How does it compare to their counterpart grown conventionally. For example, during dissolution do they dissolve like conventional materials or do they dissolve via some different pathway, and how does this affect the dissolution rate? That was what we set out to answer in our study.
We decided to tackle the problem by using state-of-the-art imaging methods – in situ liquid phase transmission electron microscopy – to watch hematite mesocrystals crystals dissolve in real time in solution. And what we found is that they DON’T dissolve like conventional materials. Instead, their dissolution pathway seems to mirror their assembly pathway. Small particles dissolve away and leave behind a skeletal framework that gradually breaks apart in ‘chunks’. In analogy to CPA, we call this reverse process dissolution by particle detachment (DPD). Below is a typical example of how those daughter hematite particles break apart and detach from the mother structure (Figure 1).
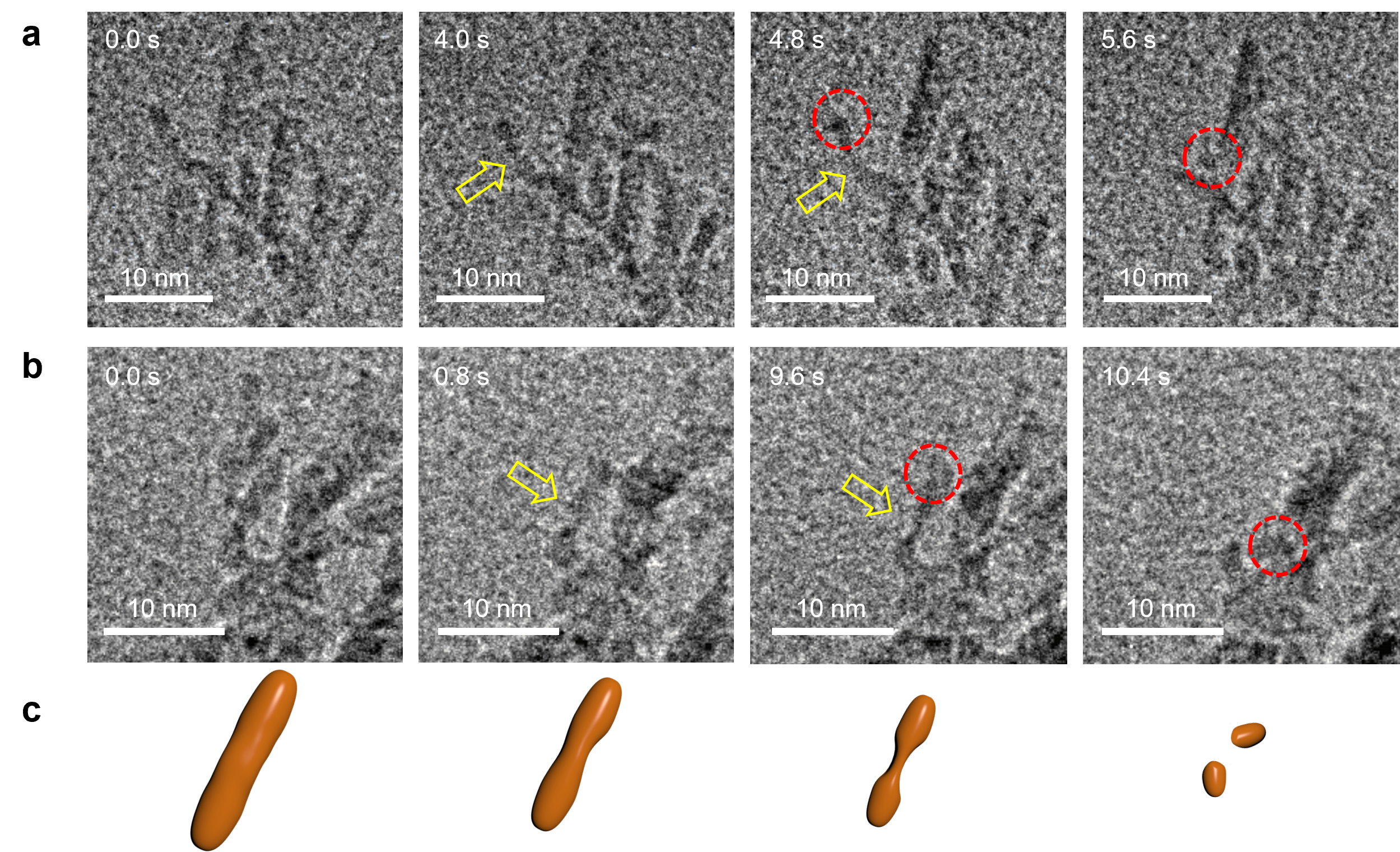 Figure 1. Necking and particle detachment during spHm mesocrystal dissolution. a, b Selected time-lapse TEM images showing the necking process (yellow arrows) and eventual particle detachment (dashed red circles). c Schematic of necking and particle detachment process (for details, see the paper).
Figure 1. Necking and particle detachment during spHm mesocrystal dissolution. a, b Selected time-lapse TEM images showing the necking process (yellow arrows) and eventual particle detachment (dashed red circles). c Schematic of necking and particle detachment process (for details, see the paper).
One common question that arises in all in situ studies is whether the observation is influenced by the “probe” used to make the observations themselves. In our work, we apply an electron beam to probe the system. To address this question, we have carefully designed experiments from different angles. First, we played with electron beam at different dose, dose rate, and beam conditions to study their influence on the system. We conducted multiple in situ experiments to enable comparisons. Second, we designed experiments (Figure 2) to isolate the impact of radicals generated by the radiolysis of the solution, which have a well-defined chemical composition. This allowed us to separate the effects of these chemical radicals from the broader beam-related effects, which can involve multiple mechanisms. To achieve this, we selectively blocked most of the electron beam, using only a snapshot to study the influence of the radicals on the system. Lastly, we performed extensive ex situ experiments to compare the results with the in situ dissolution experiment. We found that the intermediate dissolution states are comparable.
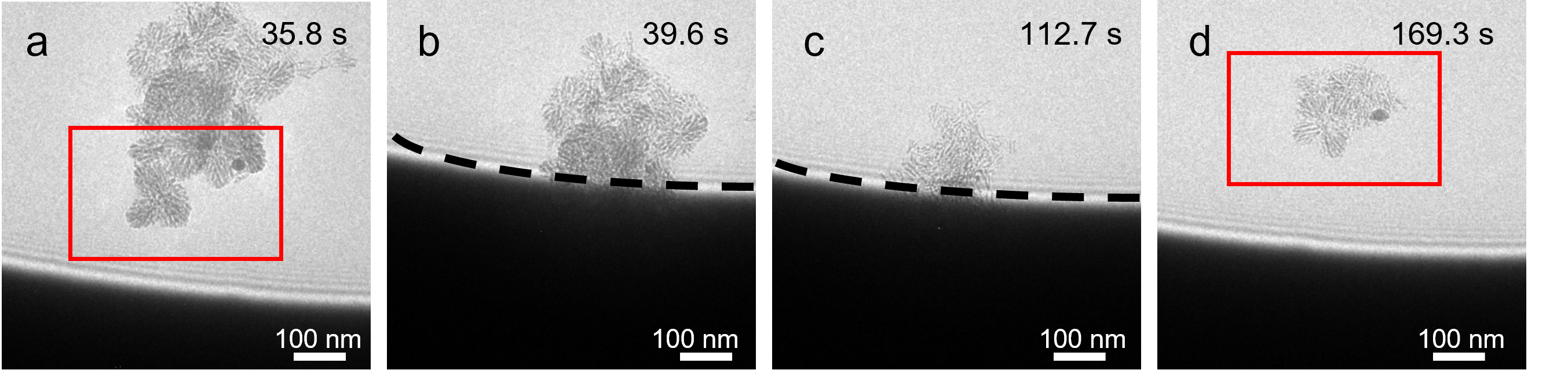
Figure 2. Hm dissolution in the electron-blocking area due to diffusive radicals. a,TEM image of aggregates of spHm. b and c, We moved the edge of beam only to irradiate the top part of the aggregates. d, after continuous imaging for a while, we shifted the beam to look at the Hm, which were in the electron-blocking area, as highlighted by the rectangle.
After confirming this, we wanted to understand WHY the particles disassemble like this. What causes these particles to detach The story comes from the earliest work on CPA by Lee Penn and Jill Banfield, who showed that if two crystals fuse imperfectly, they would trap defects called ‘dislocations’ at the interface. These dislocations would create significant local strain that might drive the particles to dissolve apart at the boundaries where they originally fused. But we couldn’t be sure. Perhaps other factors also come into play – like some quirks of the geometry? For example, there is a well-known phenomenon in materials science called the Gibbs-Thomson effect which predicts that curved surfaces can dissolve differently from uncurved surfaces. We wanted to understand whether this effect might also drive particle detachment. To understand further about the influence of strain and the Gibbs-Thomson effect on the dissolution by particle attachment, we decided to run additional modeling.
The modeling of particle dissolution was a special challenge because these mesocrystals have complex geometries. The Gibbs-Thomson equations are easily applied to predict enhanced-solubility around simple shapes like spheres and cylinders, but these are simple cases. For example, when a sphere dissolves, it typically stays a sphere. But when the neck between sub-crystals of mesocrystal starts to dissolve, the shape will locally change. The driving force for dissolution depends on exactly HOW that shape changes, and there are no analytic equations to describe this process. To deal with this problem, we developed a generalization of the Gibbs-Thomson equation that we could apply to arbitrary shapes. This let us describe the shape of a particle neck numerically, and then calculate the local effective solubility around different parts of the neck to predict how the shape should evolve. What we found, is that geometric solubility effects were insufficient to explain how these mesocrystals disassemble – we needed to factor in the interfacial strain between sub-crystals to predict their detachment. Those trapped dislocations at the boundaries between sub-crystals turn out to have a dramatic effect on the properties of the mesocrystals.
We are excited to see where this work leads. This new understanding of how mesocrystals dissolve should help us predict their behavior and find better applications in the future.
Follow the Topic
-
Nature Communications

An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in