Depolymerizable ROMP polymers discovered through ring strain calculations
Published in Chemistry

During the Covid lockdown in March 2020, we were staying at home and trying to find valuable things to do. The options include doing computations, reading papers, writing review papers, writing proposals, and maybe just having fun. Our research group was established in January 2019, still pretty new, so we were not like the established labs that can take advantage of the stay-at-home time to write the research papers. One day I asked my student Devavrat to calculate the ring strain energy of a monomer M1 that we used in a study that we published in January 2020.[1] 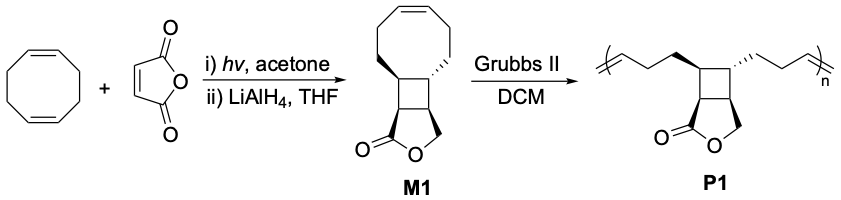 When he told me that the ring strain energy was below 5 kcal/mol, I was very surprised and I could not believe him. Devavrat repeated the calculation several times, and it always gave us this low value. I repeated the calculation myself and the value was still below 5 kcal/mol, which is 2-3 kcal/mol lower than that of a regular cyclooctene. What shocked us was that connecting to the highly-strained 4-membered ring does not make the 8-membered ring more strained, but instead, significantly reduces the ring strain energy. Notably, this ring strain energy is even lower than that of cyclopentene, the ROMP polymer of which can undergo depolymerization through ring-closing metathesis (RCM) in a presence of a ruthenium catalyst. This indicates that the polymer we used in our earlier study would be able to undergo depolymerization. I discussed this idea with Devavrat right away, and he agreed to try it out. The next day, he told me he tried out the experiments, and it worked!
When he told me that the ring strain energy was below 5 kcal/mol, I was very surprised and I could not believe him. Devavrat repeated the calculation several times, and it always gave us this low value. I repeated the calculation myself and the value was still below 5 kcal/mol, which is 2-3 kcal/mol lower than that of a regular cyclooctene. What shocked us was that connecting to the highly-strained 4-membered ring does not make the 8-membered ring more strained, but instead, significantly reduces the ring strain energy. Notably, this ring strain energy is even lower than that of cyclopentene, the ROMP polymer of which can undergo depolymerization through ring-closing metathesis (RCM) in a presence of a ruthenium catalyst. This indicates that the polymer we used in our earlier study would be able to undergo depolymerization. I discussed this idea with Devavrat right away, and he agreed to try it out. The next day, he told me he tried out the experiments, and it worked!
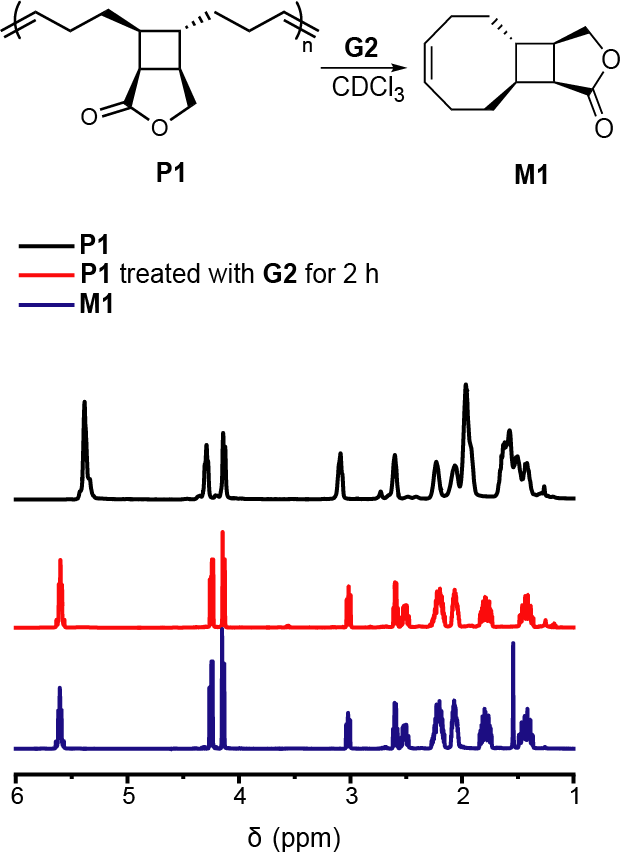
We were so excited about the results! It was an exciting period of time. The functional groups on cyclooctene can be easily tuned; we found that we were able to tune the glass transition temperature of the polymer from -30 ºC to 100 ºC, meaning that both rubber and plastic materials can be made through the cyclobutane-fused cyclooctene monomer. Compared to existing systems that are based on polyesters, polyamides, and polycarbonate, our system provides chemically recyclable polymers with a hydrocarbon backbone, which have greater thermal and hydrolytic stability. Our mechanical testing shows that it is feasible to make both rubber and plastics from this type of polymer. The monomers can be prepared with abundant material through a single-step reaction, which makes this system particularly attractive for transformational and manufacturing purposes.
[1] Hsu, T.-G. et al. A polymer with “locked” degradability: superior backbone stability and accessible degradability enabled by mechanophore installation. J. Am. Chem. Soc. 142, 2100–2104 (2020).
Follow the Topic
-
Nature Chemistry

A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in