Designing Disorder from the Bottom Up
Published in Materials
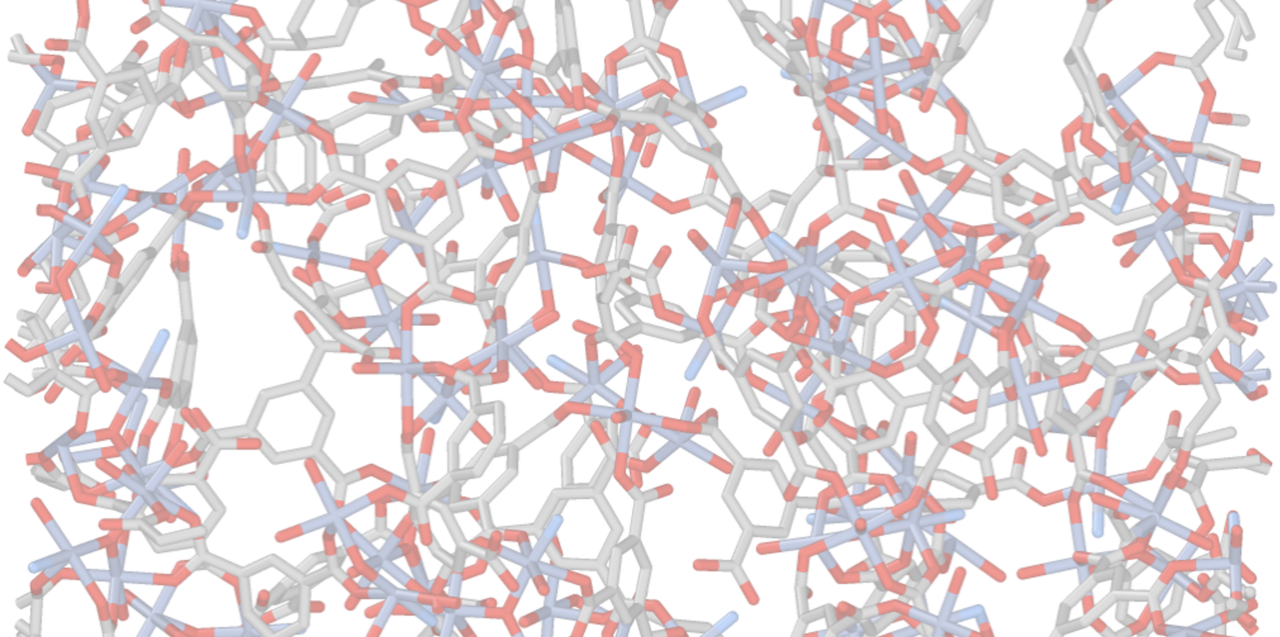
Amorphous materials are often seen as what remains after order is lost. That mindset has shaped how amorphous metal–organic frameworks (aMOFs) are typically produced: you first build a beautiful crystalline MOF, then destroy it.
Our work started with a simple question: What if we stopped treating amorphous MOFs as failed crystals and instead tried to construct them intentionally from the beginning?
That question appears simple, but it challenges a core instinct in materials chemistry. We are trained to value order. Crystallinity gives us sharp diffraction peaks, precise structures, and a reassuring sense that we understand the material. Amorphous materials are far less cooperative. They are more difficult to characterise, harder to classify, and often described by what they are not.
And yet, they are also interesting precisely for those reasons. Amorphous MOFs can be more processable, can form monoliths and gels, and can exhibit useful catalytic and transport properties. The issue is that they are still often approached indirectly. The common method is to make a crystalline MOF and then amorphise it using heat or pressure. This approach has been effective, but it also imposes a limitation: you always start from a crystal and, to some extent, inherit its logic. This raises a provocative question: Are we really designing amorphous materials or simply damaging crystalline ones?
We wanted a different starting point.
One of the big successes in crystalline MOF chemistry was the use of pre-formed building units—secondary building units or nanoclusters—to direct assembly. So we asked whether that same approach could be reversed. Could pre-formed nanoclusters be employed not to produce a better crystal, but to create a better amorphous material?
That became the central idea behind the paper.
What followed was a lesson in how counterintuitive amorphous synthesis can be. Many of the conditions that chemists use to improve crystallinity—heat, time, reversibility, careful self-correction—were exactly the things we needed to work against. To access amorphous frameworks directly, we found ourselves moving towards fast reactions, room temperature, high concentrations, and basic conditions. In other words, if crystalline synthesis is often about giving a system time to find the “right” answer, amorphous synthesis can be about preventing it from doing so.
We used pre-formed M3O nanoclusters as a starting point because they provide a valuable feature in amorphous materials: a well-defined local motif. This gave us a way to introduce some structural control without requiring long-range order. However, it also came with a trade-off. These clusters still needed to exchange ligands and reorganise into a network, and this extra reorganisation tends to bias the system towards more highly disordered frameworks. In our case, the nanocluster route consistently produced the shortest coherence lengths. Thus, the clusters were not merely rigid building blocks transferred intact into a network; they served as a means to shape disorder from the bottom up.
Another key aspect of the story involved chromium. Chromium is kinetically sluggish compared to iron: it exchanges ligands at a slower rate and is less capable of rearranging once incorporated. This made it a useful chemical lever. By introducing chromium, we could drive the system towards higher defect concentrations and increased disorder, because the framework had fewer opportunities to “heal” itself during formation. This was exciting because defects in amorphous materials are usually regarded as difficult to define, let alone control. Here, composition provided us with a practical means to influence them.
Perhaps the most conceptually interesting part came later, when we attempted to answer what seemed like a simple structural question: What are these amorphous M-BDC materials actually analogous to?
The obvious temptation was to map them onto known crystalline polymorphs. The M-BDC family is structurally diverse, with several plausible candidates. However, that view proved to be too simplistic. Analysis of local and medium-range structures revealed that, although the amorphous materials share motifs with crystalline frameworks, no single crystal structure can fully account for them. They are not merely blurred versions of MIL-101, MOF-235, or other known phases. Instead, they represent structurally distinct states that emerge from the synthesis pathway, kinetics, and composition.
For me, that was one of the most important take-home messages of the paper. Directly synthesised amorphous MOFs should not be automatically regarded as damaged crystals or incomplete versions of ordered frameworks. They can possess their own structural identity.
A number of key design principles also emerged from the work: use kinetic conditions to bias towards amorphisation, utilise nanoclusters to impose local structure while increasing disorder, and use composition to tune the defect landscape. None of these are universal rules for all systems. But collectively, they offer a starting point for understanding how disorder can be engineered, rather than merely tolerated.
That shift in perspective is truly the core focus of this paper. Yes, we present a synthetic strategy. Yes, we introduce practical design principles. But more broadly, we aim to transition amorphous framework chemistry away from a language of compromise and toward a language of design.
However, there is still a long way to go. We still need better methods to define amorphous phases, better ways to classify them, and better ways to connect structure to function. But this is exactly what makes this space exciting. Once disorder stops being seen as what is left over, it becomes a design space in its own right.
And that, ultimately, was the motivation behind the paper: not just to create new amorphous MOFs, but to demonstrate that disorder can be deliberately constructed from the bottom up.
Follow the Topic
-
Nature Communications

An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in