Development of supramolecular anticoagulants with on-demand reversibility
Published in Pharmacy & Pharmacology
Explore the Research
Development of supramolecular anticoagulants with on-demand reversibility - Nature Biotechnology
A supramolecular drug design yields anticoagulants that are rapidly reversible.
Development of supramolecular anticoagulants with on-demand reversibility
Have you ever wanted to target a specific protein but the therapeutic window seemed too risky? Would inhibiting a specific protein’s function be the holy grail treatment, yet never attempted due to unforeseen risks? These concerns are valid and of high importance when conducting benefit versus risk analysis and in many cases, the risk is simply too high. But what if we told you that the risk could be mitigated by designing reversible drugs? If a dangerous event were to occur, a simple antidote could reverse the drug’s effect, eliminating any risk.
To develop a reversible inhibitor, we chose a well-studied protein with a narrow therapeutic window and a high risk of adverse effects: thrombin, a key serine protease in the blood coagulation pathway. Thrombin regulates the transformation of fibrinogen to fibrin – an important protein in blood clot formation. Whilst many approved drugs target thrombin by binding to one or multiple of its binding sites (e.g. Argatroban, Lepirudin, etc.), they require close monitoring to ensure correct dosage as both under and over-dosing can be life threatening. It is estimated that 15% of all emergency hospital visits for adverse effects are caused by anti-coagulant related bleeding.1
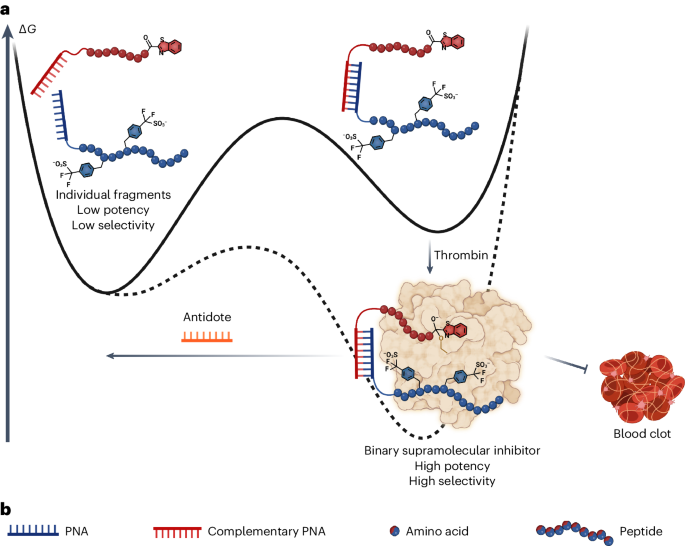
Our collaborators on the project, from the laboratory of Prof. Richard Payne (University of Sydney) and from the laboratory of Prof. Pedro Pereira (University of Porto) have dedicated much of their careers to studying small anti-coagulant proteins found in blood sucking insects. These proteins, which are post-translationally sulphated, display picomolar inhibition of thrombin by bridging multiple binding sites. Inspired by these small proteins, we decided to split the two binding fragments into two distinct molecules that bind to the active site and exosite II of thrombin. These individual fragments were attached to complementary peptide nucleic acid (PNA) strands which would hybridise together to form the active inhibitor. To our delight, when tested in a fluorogenic inhibition assay, the combined inhibitor worked wonderfully. Both fragments alone showed minimal inhibition, but when simply pipetted into the same well, they showed a Ki of 74 pM. Further work was performed to vary the PNA length and to conduct structure activity relationships (SAR) studies, all of which corroborated our previous results.
Based on the successful results, additional assays were performed in Sydney (activated partial thromboplastin time (aPTT) assay and thrombinoscope assay) and in Portugal (protease selectivity assay), which showed selective and potent inhibition of thrombin. At this stage, we contacted the laboratory of Prof. Shaun Jackson at the Heart Research Institute (HRI) in Australia to perform in vivo studies. To optimise the dose for subsequent in vivo experiments, an ex-vivo aPTT assay was used. The mice were dosed with the inhibitor and their blood was acquired and subjected to an aPTT assay. Our combined inhibitor (2.5 mg/kg = 0.315 µmol/kg) showed similar effects to Argatroban (1 mg/kg = 1.95 μmol/kg), a clinically approved small molecule inhibitor, at ~6 times lower dose. From this, we decided to perform an intravital needle injury model,2 which involves administering the mice with compound, performing a small needle injury and monitoring platelet thrombus formation and fibrin generation using fluorescent anti-fibrin and anti-GPIbβ antibodies. To our pleasure, we saw both inhibition of fibrin and decrease in thrombus volume.
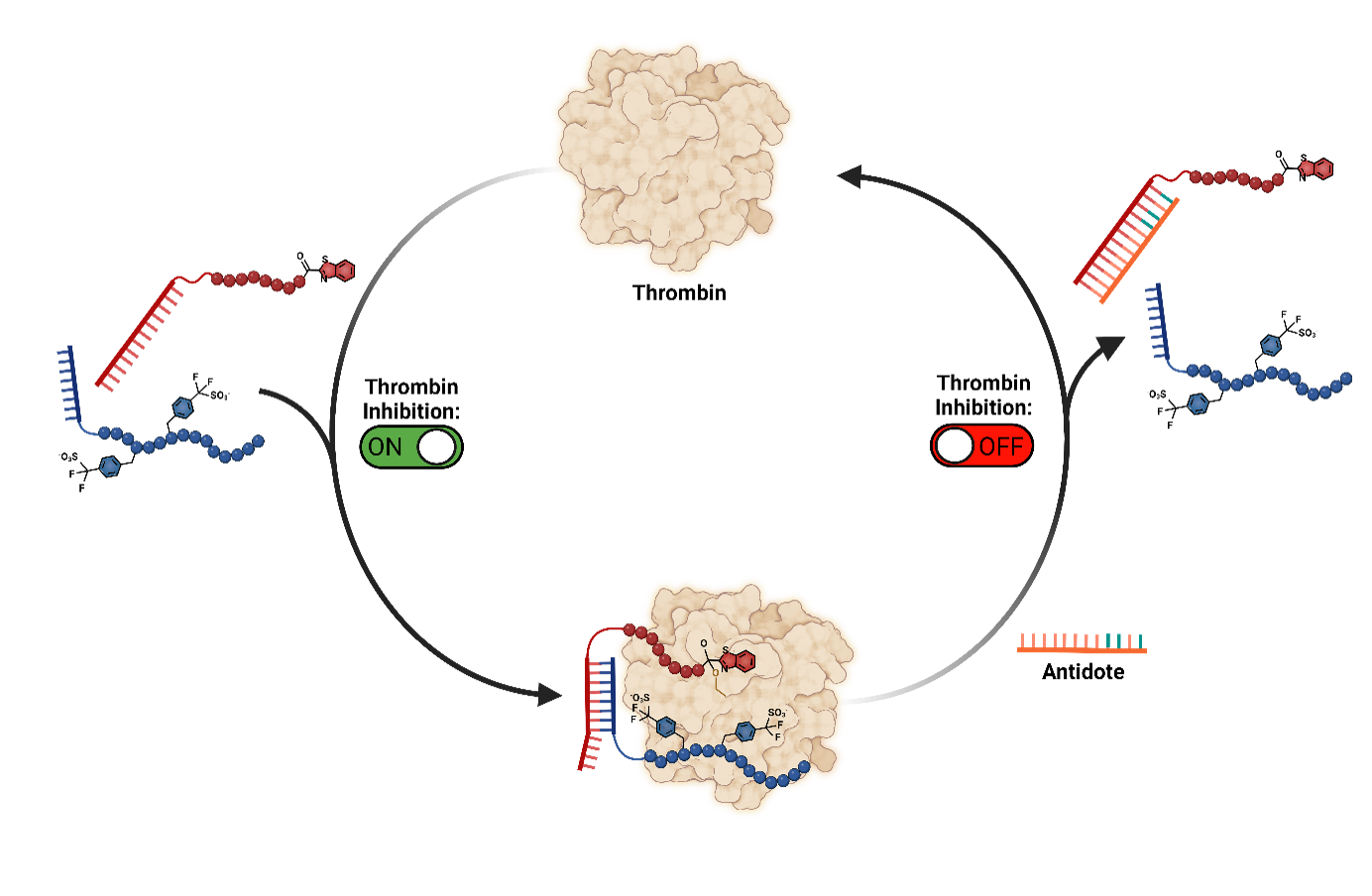
Figure 1: General Scheme for our supramolecular reversible drug. The two PNA-linked peptide fragments can assemble to inhibit thrombin. The inhibition can be rapidly reversed by the addition of a simple 12-mer antidote containing diaminopurine nucleobases. The antidote addition rapidly restores thrombin’s activity.
After validating our inhibitor, we moved on to reversing the drug. Initially, we started with a simple 8-mer PNA which would compete for hybridisation by replacing 3 adenine PNA bases for 3 diaminopurine PNA bases. The latter forms 3 hydrogen bonds with thymine rather than 2 in the case of adenine. Whilst we observed reversal of inhibition, it was only efficient at a concentration of 50 µM of antidote for the reversal of 15 nM of inhibitor – which equates to more than 3000 equivalents! Whilst we were disappointed, it is not totally surprising as the effective molarity of the two fragments around the protein is much higher than 15 nM. To improve the antidote to make it viable in vivo, we added a 4-mer PNA toehold to the active site inhibitor which enables the antidote to ‘unzip’ the PNA no matter the concentration. This time, we observed rapid reversal of inhibition with only 1 equivalent. The antidote was also trialled in the in vivo needle injury model and restored fibrin formation and thrombus volume to the pre-inhibitor state.
To conclude, we developed a reversible inhibitor of thrombin which showed potent inhibition of thrombus formation and could be rapidly reversed by antidote addition in both in vitro assays and in an in vivo model.
Reversing the effect of a drug is a sought-after property in many settings such as blood coagulation, immunotherapies, and CAR T-cell therapy. Whilst this technology was applied to thrombin, the strategy is general and could be applied to any protein where two binding fragments can be assembled. DNA encoded libraries are commonly used to discover pairs of fragments by dual display.3 This technology displays two building blocks in space using nucleobase hybridisation and would therefore lend itself to our technology.
1. Geller, A. I., Shehab, N., Lovegrove, M. C., Rose, K. O., Weidle, N. J., Goring, S. K. & Budnitz, D. S. Emergency Visits for Oral Anticoagulant Bleeding. Gen. Int. Med. 35, 371-373 (2020). https://doi.org:10.1007/s11606-019-05391-y
2. Agten, S. M., Watson, E. E., Ripoll-Rozada, J., Dowman, L. J., Wu, M. C. L., Alwis, I., Jackson, S. P., Pereira, P. J. B. & Payne, R. J. Potent Trivalent Inhibitors of Thrombin through Hybridization of Salivary Sulfopeptides from Hematophagous Arthropods. Chem. Int. Ed.60, 5348-5356 (2021). https://doi.org:https://doi.org/10.1002/anie.202015127
3. Wichert, M., Krall, N., Decurtins, W., Franzini, R., Pretto, F., Schneider, P., Neri, D., Scheuermann, J., Dual-display of small molecules enables the discovery of ligand pairs and facilitates affinity maturation. Nature Chem7, 241–249 (2015). https://doi.org/10.1038/nchem.2158
Follow the Topic
-
Nature Biotechnology

A monthly journal covering the science and business of biotechnology, with new concepts in technology/methodology of relevance to the biological, biomedical, agricultural and environmental sciences.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in