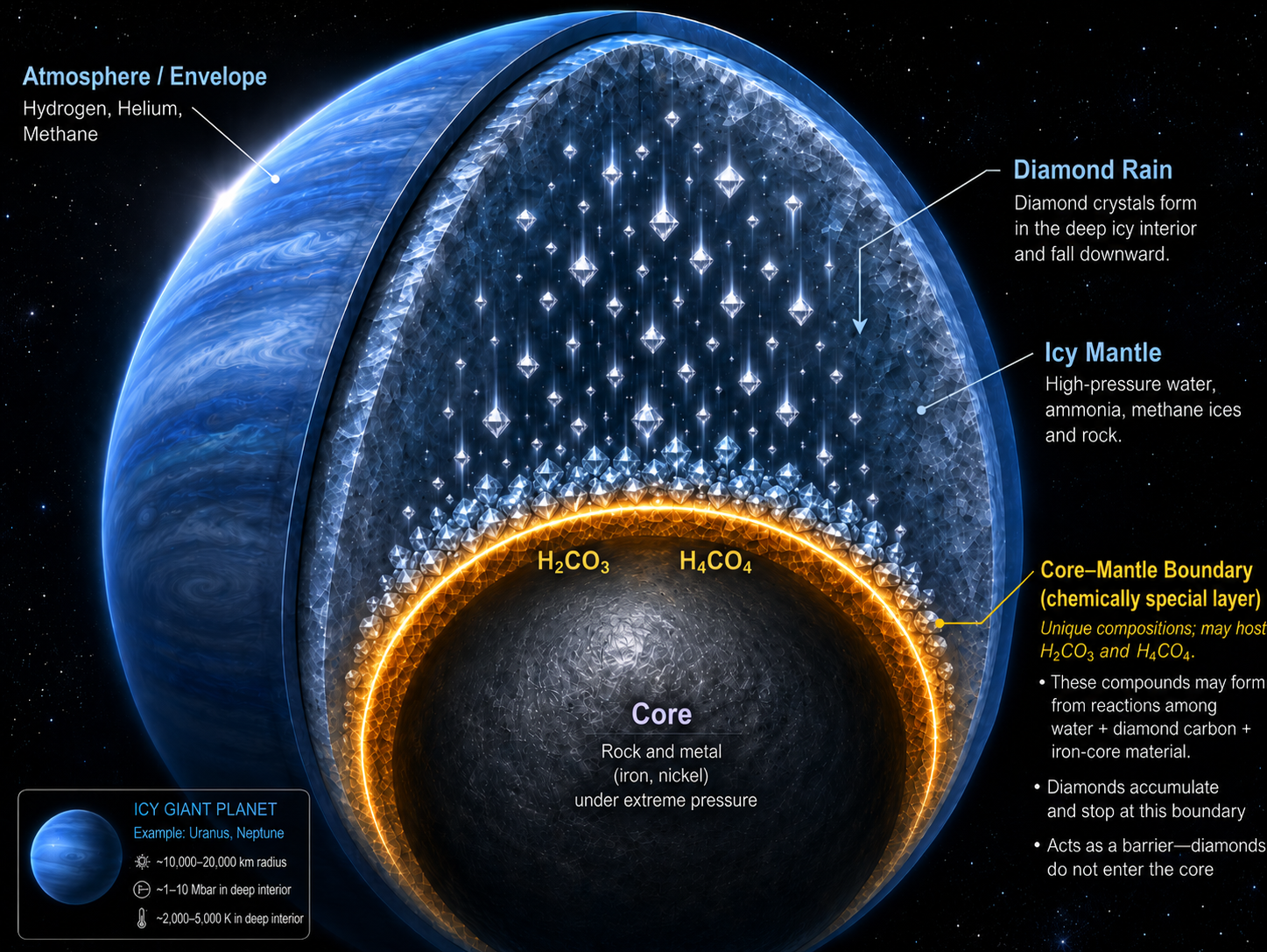
For decades, scientists have imagined the interiors of Uranus, Neptune, and similar exoplanets as places where carbon may literally fall as diamond rain. Under the immense pressures and temperatures inside these ice-rich worlds, hydrocarbons such as methane are thought to break apart. Hydrogen is released, while carbon condenses into diamond, sinking through the mantle toward the deep interior. This “diamond rain” hypothesis has become one of the most vivid pictures of carbon chemistry in giant planets.
A new theoretical study led by Dr. Qingyang Hu from Center for High Pressure Science and Technology Advanced Research (HPSTAR) suggests that the story may not end with diamonds reaching the core. Instead, when diamond-like carbon approaches the boundary between an icy mantle and an iron-bearing core, it may react again with ice and iron. The result is not a permanent layer of diamond atop the core–mantle boundary, but a new class of mobile, electrically conductive carbon-bearing ices: superionic carbonic acids. This research article was recently published by Science Advances.
“We study the chemistry of carbon-oxygen-hydrogen system at pressures at most 5 million times of Earth’s atmosphere and thousands of degrees of centigrade. These conditions were only found in the deep interiors of Neptune-like exoplanets,” said Dr. Jun Deng, the first author of the work. “We conduct extensive simulation on the chemical reactions of water ice, carbon dioxide and minerals. The results show that they will eventually form stable carbonic acid phases, including H₂CO₃ and H₄CO₄.” At sufficiently high pressures and temperatures, these compounds do not behave like ordinary solids. Their oxygen atoms form a relatively stable framework, while hydrogen and carbon can move through interconnected voids in the structure. This produces a “doubly superionic” state, in which both hydrogen and carbon become diffusive within a solid-like oxygen lattice.
The implication is especially important for diamond rain. Earlier models commonly assumed that carbon formed from methane decomposition would sink as diamond into the deep interior. The new work proposes a further chemical step: at the boundary of mantle and core, the diamond-derived carbon can react with water from the mantle and iron from the core, forming “superionic” carbonic acids together with iron hydride. In this scenario, diamond rain does not simply accumulate at the core. It is chemically transformed before, or as, it reaches the boundary.
This idea changes the role of carbon in icy planets. Rather than being locked into unreactive diamond, carbon may re-enter the planet’s dynamic interior as a mobile superionic species. “Carbon evolves through several chemical identities: methane or other reduced hydrocarbons in the mantle, diamond during gravitational settling, and finally oxidized carbonic-acid phases near the core–mantle boundary,” continued by Qingyang, “The rain therefore stops above the core, not because the carbon disappears, but because it is converted into a new, diffusive form.”
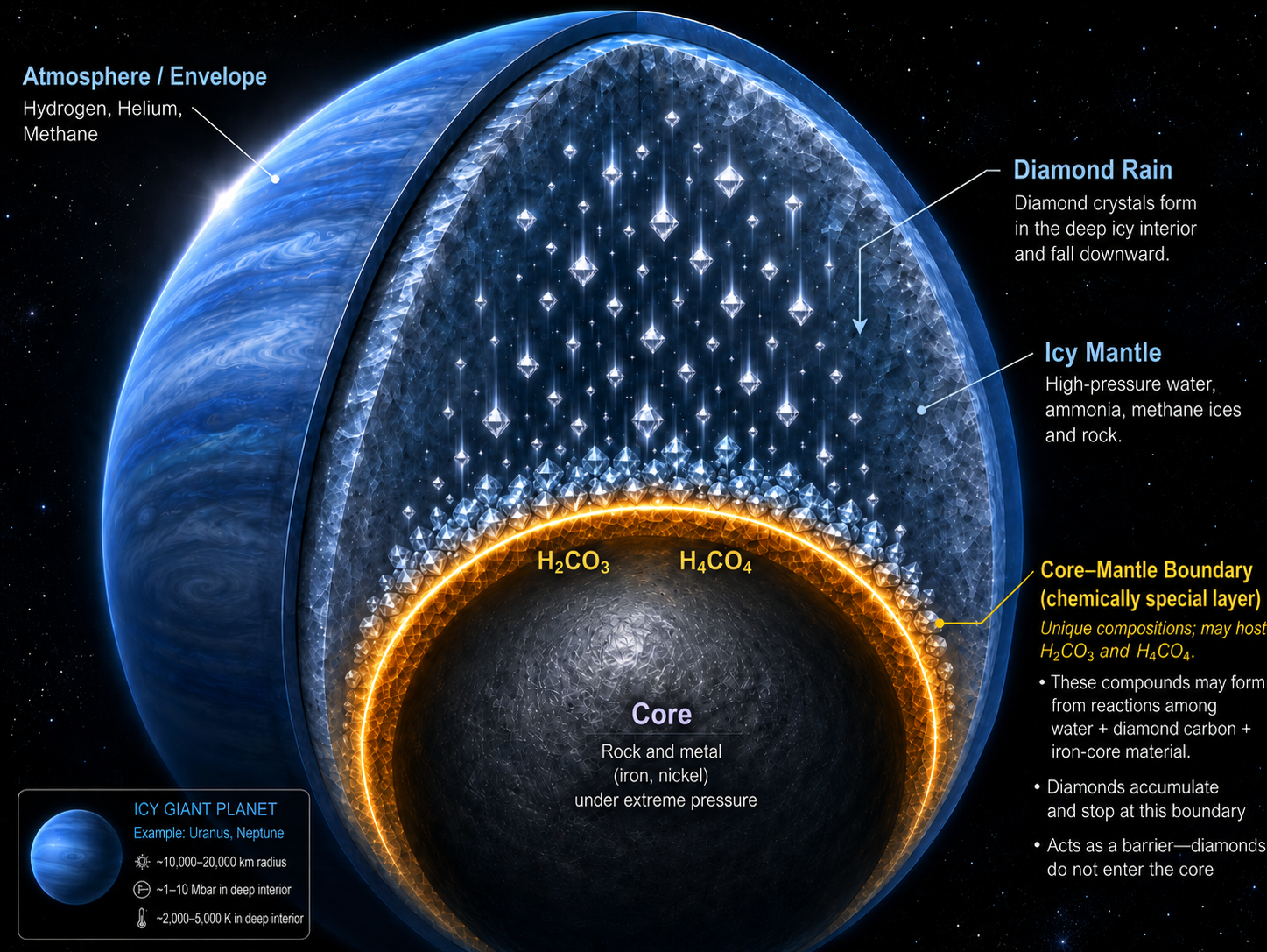
The study does not eliminate diamond rain. Instead, it gives the hypothesis a deeper and more dynamic ending. Diamond may still form from methane and sink through icy mantles. But near the core mantle boundary, the falling carbon may be chemically remobilized by iron and water, becoming part of a superionic transport system. In this view, diamond rain is not a final carbon sink. It is an intermediate stage in a larger planetary carbon cycle, one that may influence interior convection, volatile exchange, and magnetic-field generation in ice giants and water-rich exoplanets.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in