Dual color pH probes made from silica and polystyrene nanoparticles and their performance in cell studies
Published in Chemistry
Generally, there is a high demand for reliable, versatile fluorescent pH sensors – especially for in vitro studies as pH is an important indicator of cell function.1,2 pH and changes in pH can be indicative of inflammation and certain diseases. Therefore, not only sensor dyes, yet also sensor particles are needed that are brighter than molecular systems, small enough to penetrate cells, non-toxic, and allow for the close-up optical monitoring of pH. A simple and straightforward method for the read out of such cell studies is fluorescence microscopy, which is minimally invasive and suitable for in situ measurements.3
When developing such a sensor system, the pH range to be detected by the sensor dye and the intrinsic physicochemical properties of the carrier particles must be considered. The latter includes particle size, shape, and crystallinity as well as particle charge and surface functionalities that can be utilized for a subsequent surface modification. The particles can act as a matrix for the sensor molecules embedded inside the particle core or can be, alternatively, surface functionalized with analyte-responsive dyes, which must bear reactive groups for the covalent attachment to functional groups at the particle surface. Also, a combination of both strategies is feasible, enabling the usage of different spectrally distinguishable sensor dyes and thus, either the signaling of different analytes or broad range pH sensing as desired in our case. Moreover, many fluorescent nanosensors use a so-called self-referencing or ratiometric concept to account for fluctuations in the excitation light intensity. This implies the combination of a spectrally distinguishable sensor dye and an analyte-inert reference dye, excitable at the same wavelength, and can be easily realized with particle systems.
Choosing and tailoring the intrinsic physicochemical properties of the carrier particles should be based on application-specific considerations like biocompatibility, ease and low cost of preparation, and colloidal stability and performance in the respective environment determined by the desired application. In addition, for particles to be core stained with sensor dyes, the particle matrix must be permeable by the target analyte. The choice of a specific material can be justified by many factors. Based upon these options, the actual research is done raising questions like: “Does it work the way we predicted? If not, why and what may have to be modified?”. If the research is going into the right direction, there is commonly no need to consider other materials although this could yield even better results than the chosen approach. For a working system that has its assets and flaws like everything in science, another route with other possible materials is often only pursued to create new scientific impact.

In our work, we aimed for comparing two frequently used carrier materials for our pH nanosensor, namely silica and polystyrene, and assessed their pros and cons to set the ground for further developments regarding bioimaging applications. For optimum comparability, we ensured that both sensor particles showed closely matching physicochemical and optical properties. Special emphasis was dedicated to the surface of the carrier particles and final sensor particles, as the particle surface largely controls the interaction with the environment, potential toxicity issues, and particle fate.5
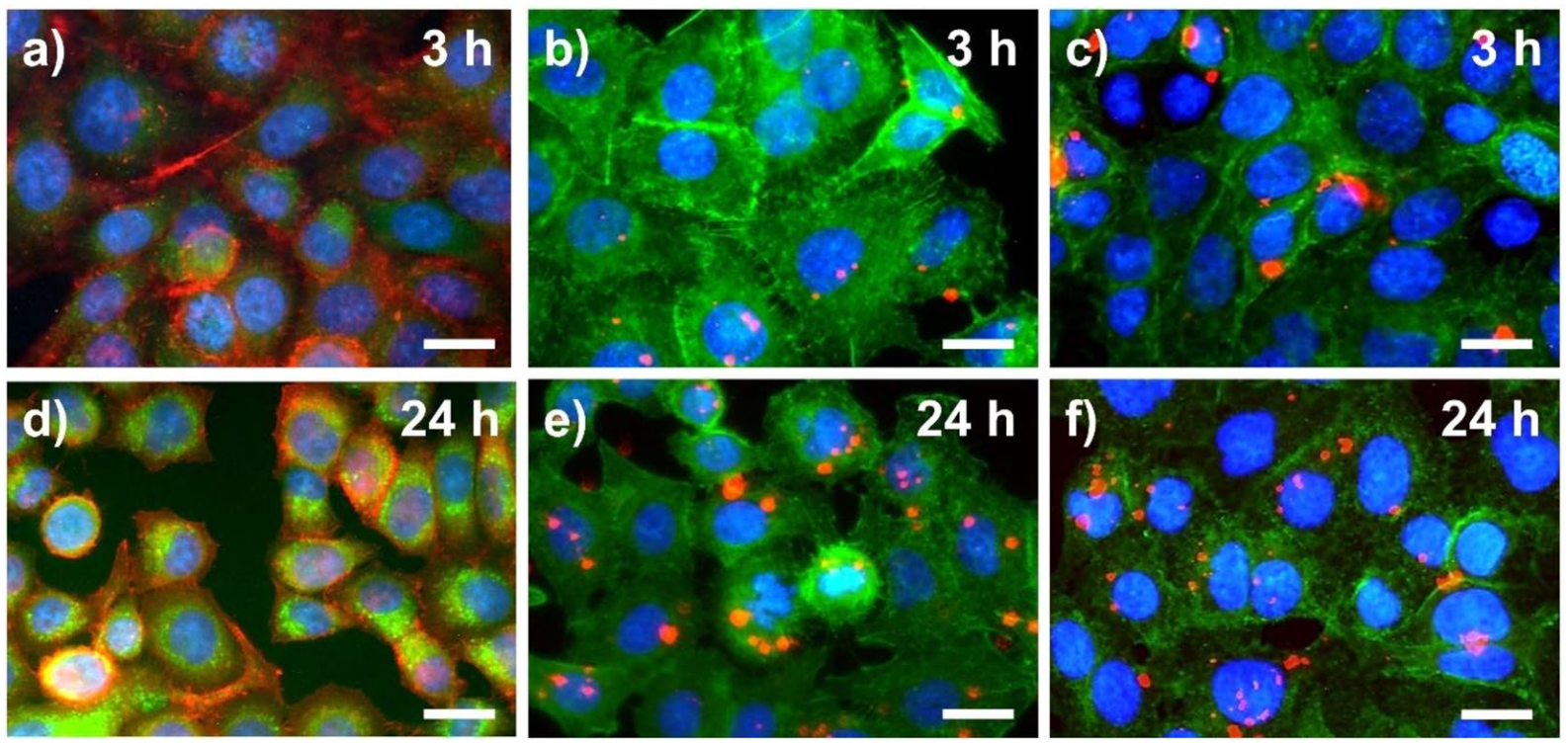
For the design of our pH nanosensor, we chose a naphthalimide dye, the green luminescence of which can be turned ON or OFF by a pH-controlled photoinduced electron transfer (PET). Protonation of the nitrogen atoms of the piperazine group of the naphthalimide sensor molecule at acidic pH values ≤ 5 results in a bright green fluorescence, indicative of an acidic environment of the sensor particles, e.g., in cells as found in the lysosomes. The morpholine group of pH probe 3 allows for the direct targeting of lysosomes. Subsequently, the pH-responsive dye molecules were attached to the surface of both types of carrier particles, which had been stained before with a red fluorescent reference dye (see Figure 1). For the intended performance comparison, we made sure that the density of the COOH groups on the nanoparticle surface used for the attachment of the sensor molecules and the amount of surface bound fluorophore molecules closely matched. This important aspect is often overlooked in the literature, although surface properties of nanoparticles are of considerable importance for the interaction with their surroundings and possible applications.5
Studies with A549 cells (adenocarcinomic human alveolar basal epithelial cells) confirmed the cellular uptake of both types of sensor particles. Cellular uptake was slightly more efficient for the silica sensor particles. The silica particles also showed a better long-term and pH stability than the polystyrene nanoparticles that degraded over time. This degradation would not have been obvious, if only the properties of the freshly prepared sensor particles had been explored, especially since the sensing performance of both particle systems is comparable. This highlights the importance of comparative studies like the one shown here and the need of follow-up experiments and stability studies to recognize and avoid possible safety and health issues in future applications of nanosensors that can stem from particle degradation and the decomposition products. In the future, we will, e.g., assess the suitability of sensor particles of different size and chemical composition together with other dyes and/or sensor molecules for cellular imaging.
In summary, with this article, we highlight the importance of sometimes taking a step back, looking at the wider picture – and learn from comparing materials and procedures. This could help to identify the best fit-for-purpose solution for a scientific challenge or specific application.
1 Steinegger, A., Wolfbeis, O. S. & Borisov, S. M. Optical Sensing and Imaging of pH Values: Spectroscopies, Materials, and Applications. Chem Rev 120, 12357-12489, doi:10.1021/acs.chemrev.0c00451 (2020).
2 Wencel, D., Abel, T. & McDonagh, C. Optical chemical pH sensors. Anal Chem 86, 15-29, doi:10.1021/ac4035168 (2014).
3 Holzinger, M., Le Goff, A. & Cosnier, S. Nanomaterials for biosensing applications: a review. Front. Chem. 2, 63, doi:10.3389/fchem.2014.00063 (2014).
4 Srivastava, P. et al. Dual color pH probes made from silica and polystyrene nanoparticles and their performance in cell studies. Scientific Reports 13, 1321, doi:10.1038/s41598-023-28203-0 (2023).
5 Geißler, D., Nirmalananthan-Budau, N., Scholtz, L., Tavernaro, I. & Resch-Genger, U. Analyzing the surface of functional nanomaterials—how to quantify the total and derivatizable number of functional groups and ligands. Microchimica Acta 188, 321, doi:10.1007/s00604-021-04960-5 (2021).
Follow the Topic
-
Scientific Reports
An open access journal publishing original research from across all areas of the natural sciences, psychology, medicine and engineering.
Related Collections
With Collections, you can get published faster and increase your visibility.
Computational biology and mathematical modelling of biological systems
Publishing Model: Open Access
Deadline: Jul 18, 2026
Water pollution and advanced treatment processes
Publishing Model: Hybrid
Deadline: May 31, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in