Evolution of Gene Therapy products : promising non-viral vector prospects from innovation patents
Published in Bioengineering & Biotechnology and Genetics & Genomics
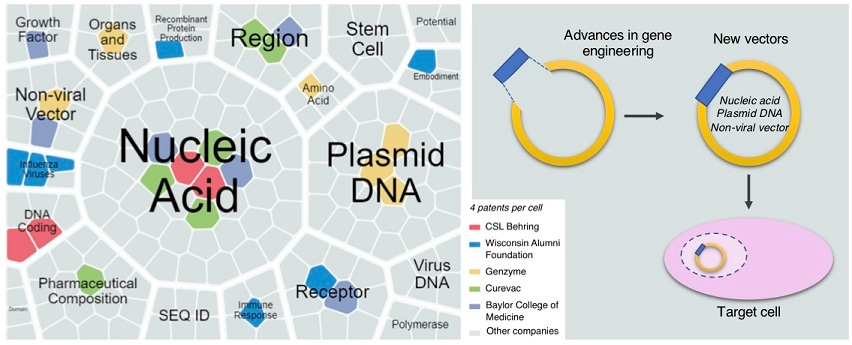
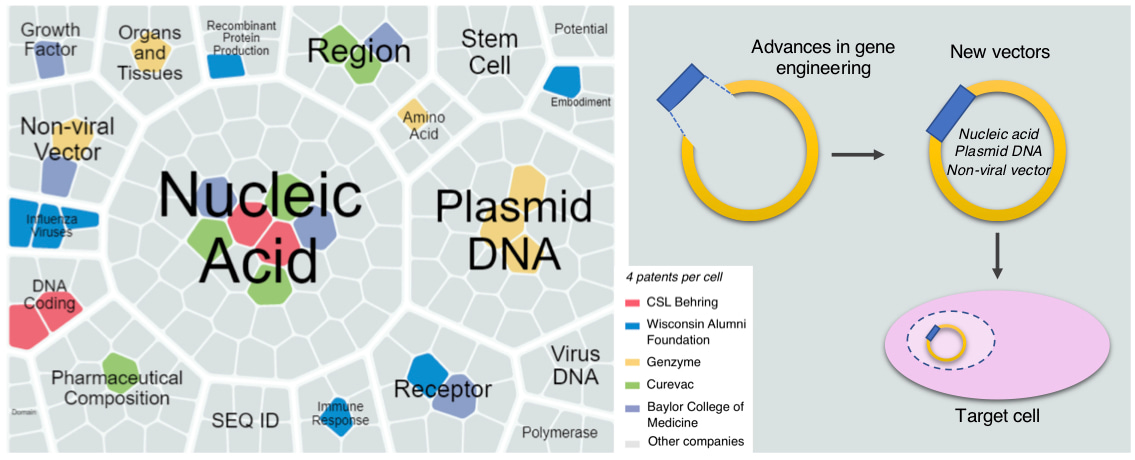
A number of technical advances have been made to introduce exogenous DNA more effectively into host cells, and in parallel, many efforts have been made to improve and refine various expression vector systems used in gene therapy. To identify key trends in the discovery and development of new “gene therapy drugs,” we analyzed data available from the Integrity database (Clarivate Analytics, USA), which provided a scenario of current developing trends, focusing on emerging technology of nonviral vectors. Non-viral vectors are considered a safer alternative to viral vectors and appear to reignite the interest of clinicians and pharmaceutical industries in the field of gene therapy.
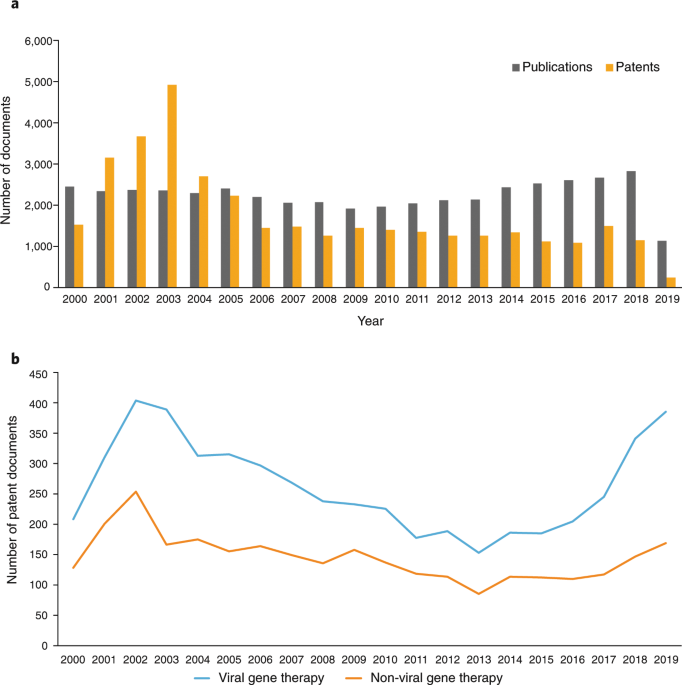
Viral vectors have played an important role in gene therapy. However, they also have several limitations, such as: i) risk of insertional mutagenesis, ii) risk of an immune response and increased inflammation, and iii) viral DNA is prone to silencing, and last but not least iv) the substantially high costs of viral production, which hinders more widespread clinical applications. With the growing interest in the application of nonviral vectors also for gene/drug delivery, diagnosis, and imaging, we analyzed the emerging patent landscape with a focus on non-viral vectors.
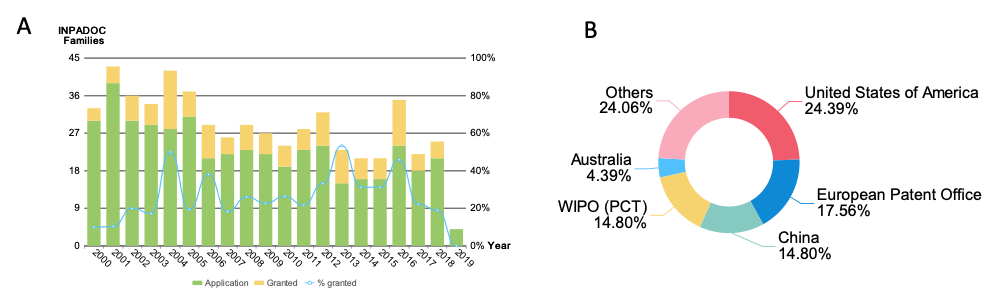
Non-viral vectors are highly desirable as they are easier to characterize and produce on a large scale. In addition, they enable greater reproducibility of large-scale production, harbor greater transgenic capacity and generate less biosecurity concerns. However, non-viral and non-integrative episomes are still relatively less efficient systems compared to viral systems and transgene expression is still often transient. In order to provide a patent landscape and to identify technologies that utilize non-viral vectors, search queries using Derwent Innovation and PatSnap platform were performed to assess the annual patenting trend in the technology field for non-viral vectors.
To overcome the challenges inherent in non-viral vectors, the stability and potency of these vectors need to be improved. These may include actions aimed at improving transfection efficiency, vector establishment, and vector maintenance in the host cell nucleus, as well as decreasing plasmid loss, cell toxicity, and production costs. Combined, these developments can be truly transformative, making non-viral DNA vectors highly favorable gene therapy tools.
Our work has identified cutting-edge inventions, major players, and top organizations working with non-integrating vectors. The team used big data, patent and clinical data mining to identify technological trends for the gene therapy field. We envision that these analyses will help guide future developments for gene therapy.
This work brought together investigators from across the globe in a joint effort to use new databases and methods to better understand the trends of the gene therapy field in respect to nonviral vectors. Dimas Covas, coordinator of the Center for Cell-based Therapy, affiliated with the University of São Paulo in Brazil, lent his extensive experience in cell therapy. Aglaia Athanassiadou, Virginia Picanço-Castro and Marxa Figueiredo contributed their extensive experience on nonviral vectors for gene therapy. Cristiano Pereira and Geciane Porto brought their expertise in economics and business administration to the analyses. Each contribution was fundamental to achieving a new way to identify technological trends in this field. Our groups continue to work individually or in collaboration to generate and patent new vectors to help fill the needs of this re-emerging field of nonviral gene therapy.
Gene therapies are transforming the current treatment landscape for various diseases, and this study reveals the possibility of using nonviral vectors for advancing the field. New innovations will surely continue to emerge to generate systems using non-viral vectors, with the potential to be expanded and suitable for clinical use.
We encourage those who are interested to learn more to read our full article through the link below:
Follow the Topic
-
Nature Biotechnology

A monthly journal covering the science and business of biotechnology, with new concepts in technology/methodology of relevance to the biological, biomedical, agricultural and environmental sciences.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in