Exploratory evidence for differences in GABAergic regulation of auditory processing in Autism Spectrum Disorder
Published in Biomedical Research
Clinical trials that have tested pharmacological interventions in large numbers of autistic people have so far been unsuccessful 1. This is despite evidence from preclinical studies, genetics and post-mortem studies linking peripheral and central markers of multiple candidate neurochemical systems to brain function in autism 2. Whilst this has in part been explained by the heterogeneity of the autistic population, supporting evidence is largely indirect, coming from animal models and ‘correlational’ approaches which suggest a plausible link between brain chemistry and function. Correlations are not causal and the only way to establish that a neurosignalling system is involved in target neural process is to challenge it and observe a change in the mechanism. This is basis of preclinical study designs, but there has been limited translation to humans.
To bridge the translational gap in autism research, we have developed a direct experimental approach to examine neurochemical regulation of information processing across the different organizational levels of the human brain which is sensitive to individual differences in neurotypical and neurodivergent people. We call this a “Shiftability” paradigm 3. It is based on the concept that any initial neurodevelopmental divergence in conditions such as autism must act on processes, such as maturation of sensory systems, which happen early in life. Sensory processes may then constitute more tractable targets to investigate the differences in neurochemical regulation of the autistic and non-autistic brain than the complex social and behavioural characteristics which emerge later in postnatal life and are downstream of these more ‘foundational’ early-life processes.
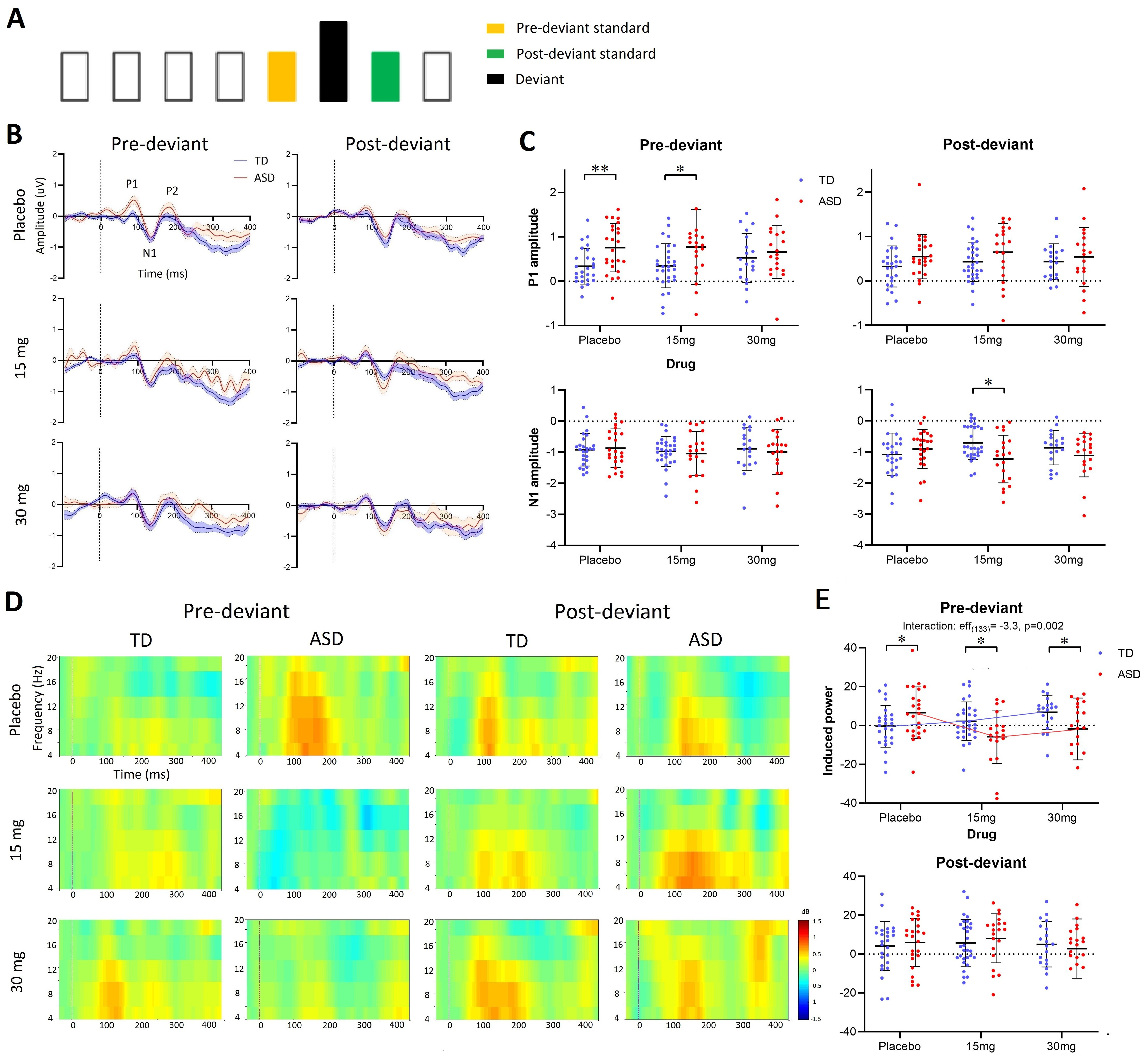
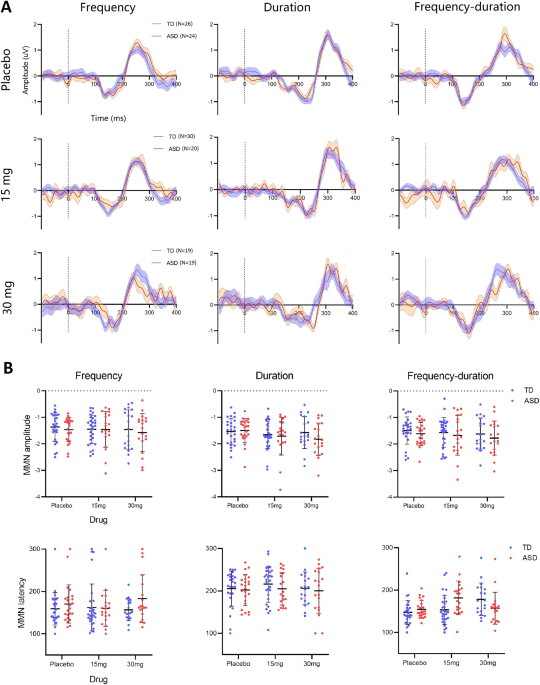
In our article “Exploratory evidence for differences in GABAergic regulation of auditory processing in Autism Spectrum Disorder” recently published by Translational Psychiatry, we tested the hypothesis that there are differences in the GABAergic control of sensory processing in autistic and non-autistic people. To do this, scalp EEG was used to record neurophysiological responses induced in an auditory oddball paradigm. In this paradigm, the neural response to a stream of regular repeating sounds (standards) is expected to be suppressed unless occasionally interrupted by sounds deviating in frequency, duration or frequency-duration combination (odd-balls). Our main finding was that the GABAB receptor agonist arbaclofen reversed weaker repetition suppression of spectral responses in autistic participants but disrupted suppression in non-autistic participants. Thus, GABAergic regulation of auditory processing is fundamentally different in autism. The extent of these GABA-dependent sensory differences across individuals in both groups was tightly linked to the extent of their autistic traits, as captured on the Autism Quotient, indicating that autistic behavioral complexity is likely downstream of more fundamental differences in information processing. We also delivered a means to measure biological responsiveness to drug challenge at the level of the individual which we hope will support precision medicine approaches in ASD, such as pretrial stratification. The approach used in our study is also useful because the neural computations underlying sensory processing are relatively well conserved between humans and other animals 4, facilitating back-translation for a deeper investigation of the cellular and molecular under-pinnings of sensory processing differences in autistic and non-autistic people.
Sensory atypicalities are among the earliest manifestations of ASD 5. These persist lifelong and can be distressing, with 60 to 96% of children with ASD exhibiting some degree of divergent responses to sensory stimuli 6-8. Our findings consolidate a role for GABA differences in sensory processing differences in ASD; they provide a mechanistically informed intervention target; and generate an individual measure of target engagement. More generally, we hope our work will encourage a paradigm shift in neuropsychiatry by demonstrating that the gap between preclinical neuroscience in animals and the human can be effectively bridged to advance understanding of complex human conditions.
- McCracken JT, Anagnostou E, Arango C, Dawson G, Farchione T, Mantua Vet al.Drug development for Autism Spectrum Disorder (ASD): Progress, challenges, and future directions. European Neuropsychopharmacology 2021; 48: 3-31.
- Willsey HR, Willsey AJ, Wang B, State MW. Genomics, convergent neuroscience and progress in understanding autism spectrum disorder. Nature Reviews Neuroscience2022; 23(6): 323-341.
- Whelan TP, Daly E, Puts NA, Malievskaia E, Murphy DG, McAlonan GM. Bridging the translational neuroscience gap: Development of the shiftability paradigm and an exemplar protocol to capture psilocybin-elicited shift in neurobiological mechanisms in autism. medRxiv2023: 2023.2005. 2025.23290521.
- Robertson CE, Baron-Cohen S. Sensory perception in autism. Nature Reviews Neuroscience2017; 18(11): 671-684.
- Ciarrusta J, Dimitrova R, Batalle D, O’Muircheartaigh J, Cordero-Grande L, Price Aet al.Emerging functional connectivity differences in newborn infants vulnerable to autism spectrum disorders. Translational psychiatry 2020; 10(1): 1-10.
- Bogdashina O. Sensory perceptual issues in autism and asperger syndrome: different sensory experiences-different perceptual worlds. Jessica Kingsley Publishers2016.
- Marco EJ, Hinkley LB, Hill SS, Nagarajan SS. Sensory processing in autism: a review of neurophysiologic findings. Pediatric research2011; 69(8): 48-54.
- Schauder KB, Bennetto L. Toward an interdisciplinary understanding of sensory dysfunction in autism spectrum disorder: an integration of the neural and symptom literatures. Frontiers in neuroscience2016; 10: 268.
Follow the Topic
-
Translational Psychiatry
This journal focuses on papers that directly study psychiatric disorders and bring new discovery into clinical practice.
Related Collections
With Collections, you can get published faster and increase your visibility.
From mechanism to intervention: translational psychiatry of childhood maltreatment
Publishing Model: Open Access
Deadline: Jun 30, 2026
Moving towards mechanism, causality and novel therapeutic interventions in translational psychiatry: focus on the microbiome-gut-brain axis
Publishing Model: Open Access
Deadline: Nov 15, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in