First-in-man transplantation of a liver after 3 days of ex situ preservation
Published in Bioengineering & Biotechnology
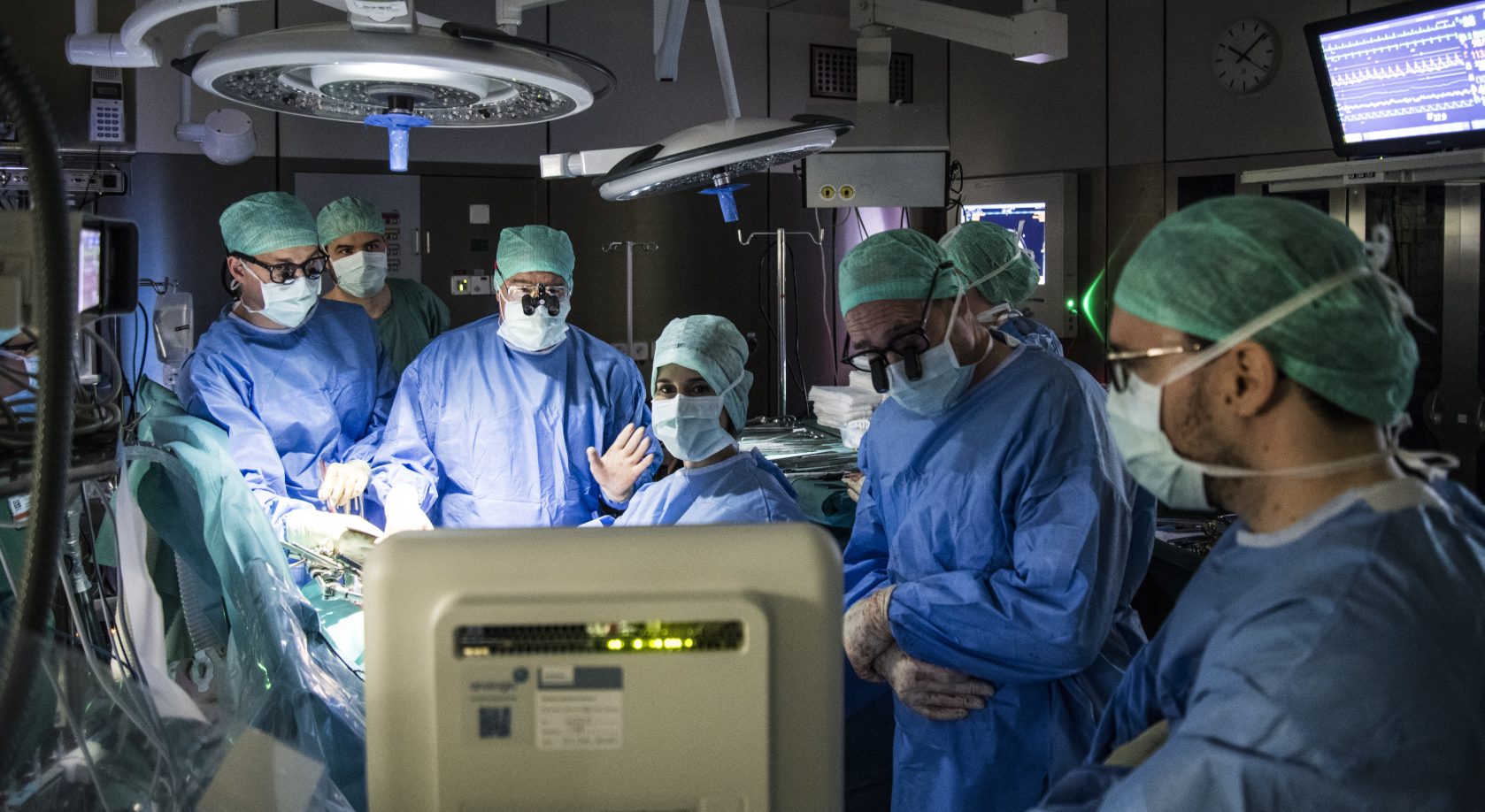
The success of liver transplantation, which routinely restores normal quality of life to patients suffering from dismal conditions due to end stage liver disease, has created a high demand for donor livers that exceeds supply. This has resulted in ever-growing waitlists, and many are never able to receive a donor graft. Therefore, the community has been exploring novel avenues to provide access to more organs.
Our group developed a novel perfusion technology that significantly advanced the viable time of ex situ preservation of whole and partial human livers from a day to a whole week and beyond1,2. In addition to extending the storage time, our perfusion system enables detailed organ assessment over multiple days with the possibility of therapeutic intervention. The increased time and ability to address underlying issues could allow for the recovery or repair of poor-quality donor organs that would be rejected for transplantation. The presented work formed the first step towards the proof-of-principle, i.e., transplantation after ex situ perfusion after multiple days.
In our current publication3, we present the results of the first-in-human transplantation of a discarded injured liver, which was preserved, assessed and treated during 3 days in our novel device. The liver was rejected by all centers for transplant because of the presence of a tumor of unknown dignity (benign or malignant) and a microbial contamination caused by chronic abscesses in the abdominal cavity of the donor. Our system provided the required time to establish the nature of the tumor, to combat the infection with a course of antibiotics, and, thus, to assess in detail the quality of the liver graft. Therefore, we placed the graft on our device providing time for the pathologists to determine that the tumor was benign and to clear the organ of any sign of residual infection. After 3 days of perfusion, we decided to proceed with transplantation as the liver met the previously determined viability criteria1.
The recipient was suffering from recurrent liver cancer and cirrhosis, with only a miniscule chance to receive a donor liver in time (the expected waiting time for patients with liver cancer is 1 year in Switzerland). On its 4th day outside of the body, the donor liver was transplanted in May 2021 to the 62-year-old patient. For the first time in medical history, a solid organ was transplanted after being multiple days outside of the human body.
We were amazed by the course within the first days after transplantation: reperfusion injury was minimal with very low release of liver enzymes (AST, ALT), stable hemodynamics, the absence of rejection and necrosis. Full liver function returned within 4 days post-transplantation. Interestingly, the liver exhibited pronounced proliferation following transplantation, and a markedly increased volume after week 6. Now, one year after transplantation, the patient is cured from his cancer and is enjoying a normal quality of life.
The successful first-in-human case is the result of a highly interdisciplinary research endeavor, that was carried out by a team of surgeons, bio-chemists and engineers working together on a daily basis. This team was assembled and supported through the generous support of the Wyss Zurich Translational Center – without a doubt, this work would not have been possible without the vision and generosity of Hansjörg Wyss. The key to success was the persistent dedication of the team to create the most ‘physiological’ preservation machine to date to treat (figure below) and rescue injured donor organs. Throughout the machine development, we recreated critical functions of the body as closely as we could, as exemplified by automatic glucose control using insulin and glucagon, flow control using vasodilators and vasoconstrictors, by pulsatile arterial flow, by a dialysis system controlling for hematocrit, and by physiological oxygen saturation in the portal flow. Moreover, feedback control loops enabled autonomous operation of the device to minimize the possibility of human error. Our hypothesis is that the combination of these features allowed us to push the state of the art of machine perfusion beyond 24 hours and up to 10 days.
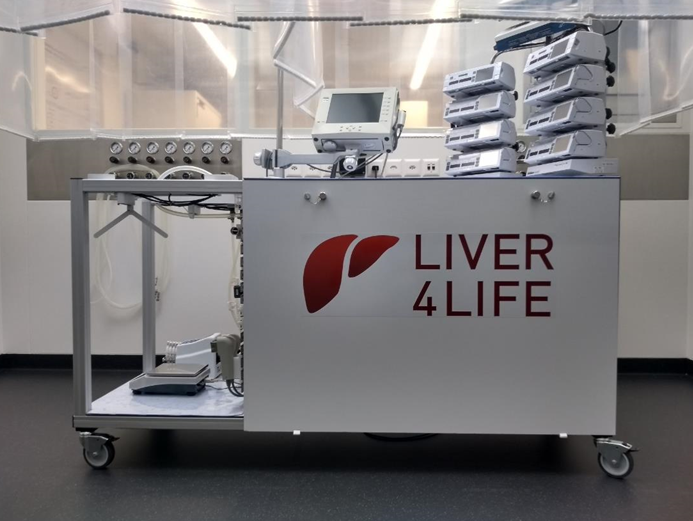
This first-in-human case shows that donor liver grafts can be preserved in an artificial environment for multiple days, which opens up new horizons and treatment possibilities. Here, we capitalized on the extended preservation time to treat infection, assess tumor dignity and validate graft viability, providing the required confidence to proceed with the transplant. This initial case needs to be reproduced with further transplantations and assessed at longer follow-up times. Nonetheless, we present here the first evidence that long-term ex situ preservation is a promising therapeutic approach with countless treatment opportunities, e.g., for tissue repair, immunomodulation, and even ex situ regeneration.
- Eshmuminov, D., Clavien, P.-A. et al. An integrated perfusion machine preserves injured human livers for 1 week. Nat. Biotechnol. (2020). doi:10.1038/s41587-019-0374-x
- Mueller, M., Hefti, M., Clavien, P.-A . et al. Long-term Normothermic Machine Preservation of Partial Livers: First Experience With 21 Human Hemi-livers. Ann. Surg. 274, 836–842 (2021).
- Clavien P.-A., Dutkowski P. et al. Transplantation of a human liver following 3 days of ex situ normothermic preservation. Nature Biotechnology (2022) doi: 10.1038/s41587-022-01354-7
Follow the Topic
-
Nature Biotechnology
A monthly journal covering the science and business of biotechnology, with new concepts in technology/methodology of relevance to the biological, biomedical, agricultural and environmental sciences.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in