From Obstacle to Opportunity: Targeting TGFβ in Pancreatic Cancer
Published in Bioengineering & Biotechnology, Cancer, and Immunology
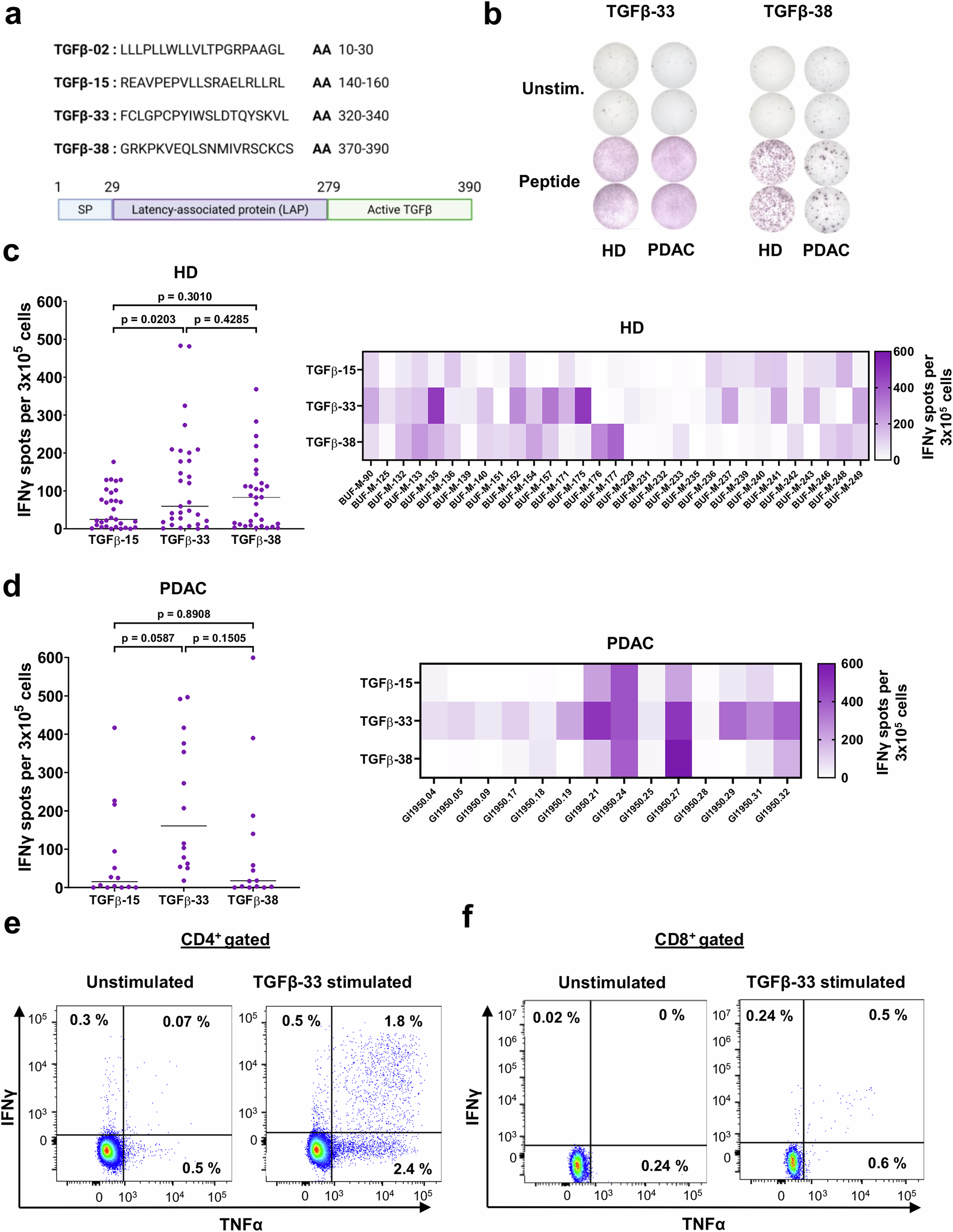
Despite the success in several malignancies, most patients with pancreatic ductal adenocarcinoma do not benefit from immune checkpoint inhibitors (ICIs). A major reason for this is the tumor microenvironment, which actively suppresses immune responses and prevents effective T cell activity. Among the key players in this suppressive environment is transforming growth factor beta (TGFβ). This cytokine is a double-edged sword: while it plays essential roles in normal tissue homeostasis, it promotes fibrosis, immune evasion, and metastasis in established tumors. Because of this, TGFβ has long been considered an attractive therapeutic target. However, directly blocking TGFβ has proven challenging, with issues related to toxicity and limited clinical efficacy. Instead of trying to inhibit TGFβ itself, our work builds on a different idea: what if we could train the immune system to recognize and eliminate the cells that produce it? This concept forms the basis of immune-modulatory vaccines (IMVs), which aim to activate anti-regulatory T cells; pro-inflammatory T cells that target cells based on their expression of immunosuppressive molecules within the tumor microenvironment. Previous studies have demonstrated that T cells can recognize epitopes derived from TGFβ, and importantly, that immunity against one such epitope, TGFβ-15, is associated with improved survival in patients with pancreatic cancer treated with immunotherapy and radiotherapy. However, preclinical studies suggested that targeting multiple epitopes might be more effective than focusing on a single one.
This raised an important question for us: do pancreatic cancer patients naturally harbor T cells specific to multiple TGFβ-derived epitopes, and if so, do these have clinical relevance? To explore this, we examined immune responses to several TGFβ-derived peptides in both healthy donors and patients. As the experiments progressed, one observation that stood out to us was that T cells recognizing multiple epitopes, particularly TGFβ-33 and TGFβ-38, were not only present but also readily activated. Interestingly, TGFβ-33 appeared especially immunogenic in the patient cohort. We then asked whether these immune responses were merely detectable or actually clinically relevant. Using samples from pancreatic cancer patients treated with checkpoint inhibitors and radiotherapy, we observed that individuals with stronger baseline TGFβ-33-specific T cell responses experienced better clinical outcomes. Even more strikingly, patients with responses to more than one TGFβ-derived epitope had the longest overall and progression-free survival. Seeing this pattern emerge was particularly encouraging, as it suggested that T cells specific to multiple TGFβ-derived epitopes are associated with a better response to immunotherapy, thereby supporting a TGFβ multi-epitope vaccine in combination with ICIs in this patient cohort.
Naturally, the next question was therefore: could such multi-epitope responses be harnessed therapeutically? To address this, we designed a single mRNA construct encoding multiple TGFβ-derived epitopes and tested whether it could activate T cells with multiple specificities. In addition to experiments using peptide-pulsed antigen-presenting cells (APCs), we found that APCs transfected with this construct could process and present all included epitopes, thereby simultaneously activating multiple T cell populations. This provided proof of concept for a multi-epitope vaccination strategy, either based on peptide combinations or mRNA-based delivery.
Another important aspect of our study was to understand the function of these TGFβ-specific T cells. We found that they recognize target cells in a TGFβ-dependent manner and display both pro-inflammatory and cytotoxic features, including the production of granzyme B and granulysin. This suggests that the clinical effect may, in part, arise from these T cells directly targeting TGFβ-expressing cancer cells and other immunosuppressive cells within the tumor microenvironment, thereby mediating both direct anti-tumor effects and indirect effects through modulation of the microenvironment.
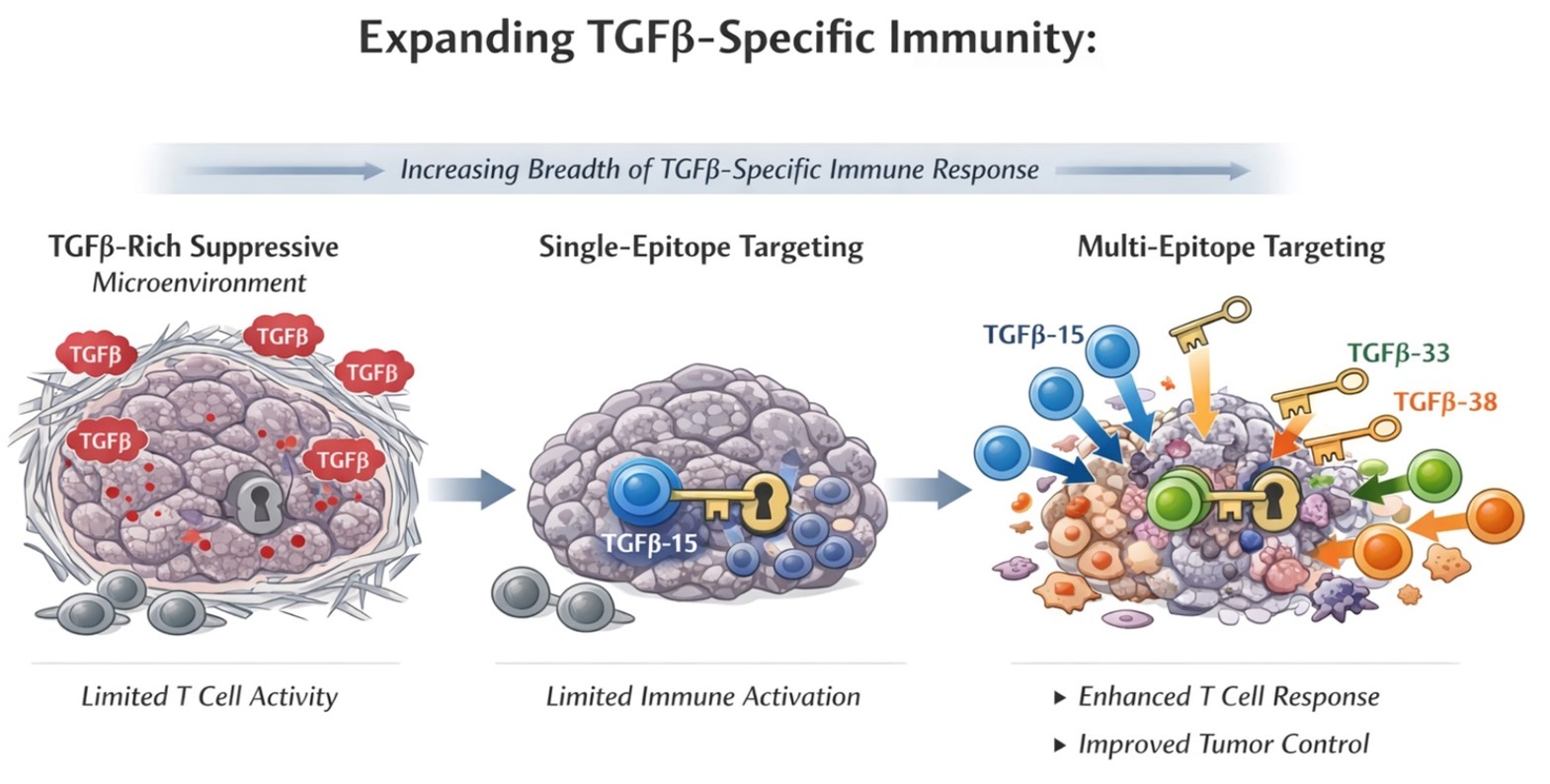
Taken together, our findings support a model in which naturally occurring TGFβ-specific T cells can be expanded and harnessed to counteract the immunosuppressive tumor microenvironment. Rather than blocking a single pathway, this approach directs the immune system toward the very cells that sustain suppression. As illustrated in the figure, this represents a shift from a TGFβ-“locked” tumor to one that can be opened by broad, multi-epitope T cell responses. By moving beyond a single “key” and instead engaging multiple TGFβ-derived epitopes, we expand and strengthen anti-tumor immunity.
Looking ahead, multi-epitope TGFβ-based IMVs could complement existing immunotherapies, such as ICIs, particularly in resistant cancers, such as pancreatic cancer. By broadening the immune response and alleviating suppression and fibrosis, this strategy may help convert “cold” tumors into immunotherapy-responsive ones, turning TGFβ from a barrier into a vulnerability, and ultimately, an opportunity for therapeutic intervention.
Follow the Topic
-
Signal Transduction and Targeted Therapy
This is an international, peer-reviewed, open-access journal publishing articles related to signal transduction in physiological and pathological processes, alongside signal transduction-targeted therapeutics in the form of biological agents and small molecular drugs used to treat human diseases.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in