Horizontal propagation of EMT, stemness, and metastasis by transferring the MAP17 protein through secreted vesicles
Published in Cancer
MAP17 (PDZK1IP1, DD96, SPAP) [1-3] is a small (approximately 17 kDa), nonglycosylated protein usually localized to the plasma membrane and associated with areas of cell-cell contact [4]. Although its expression in non-tumoral cells is restricted to specific epithelial cell populations in kidney [4], more than 50% of advanced tumors exhibit significant expression of MAP17 [1, 3, 5, 6]. High MAP17 levels appear in most human carcinomas and in other non-epithelial neoplasias, such as glioblastomas or lymphomas [4, 7]. This protein is also associated with tumor progression, since it regulates cellular transformation and malignancy [1, 8, 9], correlating its expression with an increase in cell de-differentiation [8-10]. It has also been recently shown that the expression of MAP17 is not restricted to cancer, appearing upregulated in chronic inflammatory diseases and induces the transcription of genes related to inflammatory response, such as HLAs, IL-6 and NFAT2 [6, 11].
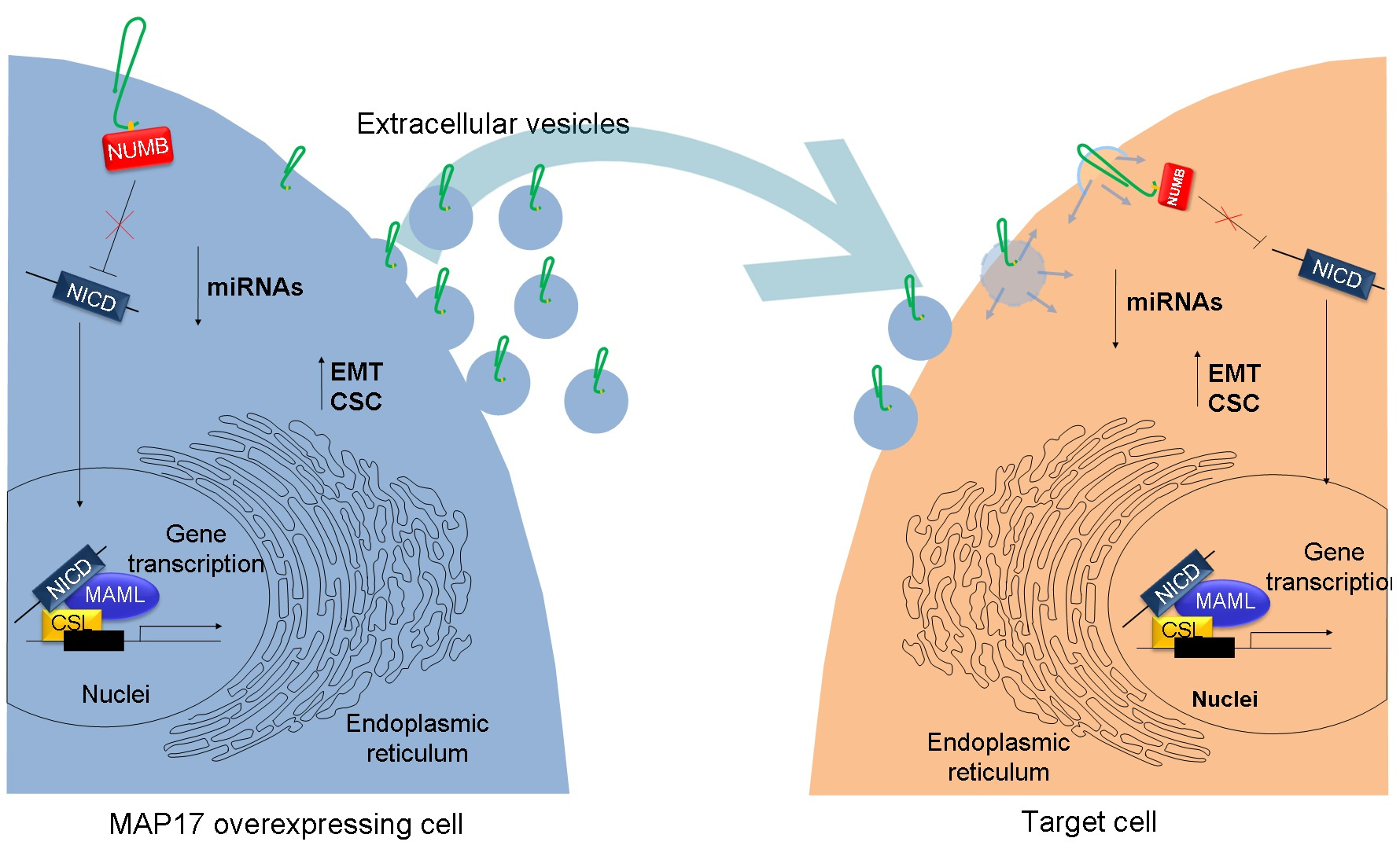
Although MAP17 has no enzymatic or transcriptional activity, it exerts its described roles through the regulation of several cell signaling pathways. MAP17 overexpression activates NOTCH pathway by sequestrating NUMB, a known inhibitor of NOTCH, allowing a higher activation of the pathway [12, 13].
During with tumor progression, MAP17 levels increase due to its promoter demethylation in cancer cells. The increased MAP17 levels in tumors induce NOTCH pathway activation regulating the expression or transcriptional repression of specific microRNAs producing multiple changes in gene expression. As a consequence, the epithelial mesenchymal transition, EMT, and stemness are induced, greatly increasing the metastatic potential of these cells both in vitro and in vivo. Additionally, MAP17 overexpression also increased extracellular vesicle secretion, and MAP17 itself was secreted as a cargo in these particles, inducing EMT and stemness phenotypes in the recipient cells. Therefore, MAP17 expression epigenetically enhances the horizontal propagation of EMT and metastatic phenotypes by transferring itself between subsets of neoplastic cells. Finally, elimination of MAP17 or exosomes from the microenvironment abrogates the induction of EMT/stemness transcription.
Altogether, our work provides a totally unknown functional circuit between the horizontal propagation, NOTCH pathway, miRNAs regulation, EMT-CSC pool and metastases and the oncogene MAP17, overexpresed in around a 50% of human breast tumors. This may provide, in the future, a new form to attack breast cancer metastasis. Furthermore, the mechanism might be broader since most tumor types, at advances stages, also present high levels of MAP17 [6,8].
- Guijarro, M. V., et al. MAP17 overexpression is a common characteristic of carcinomas. Carcinogenesis 2007; 28: 1646-1652
- Kocher, O., et al. Identification of a novel gene, selectively up-regulated in human carcinomas, using the differential display technique. Clinical Cancer Research 1995; 1: 1209-1215
- Wang, N., et al. Screening and Identification of Distant Metastasis-Related Differentially Expressed Genes in Human Squamous Cell Lung Carcinoma. The Anatomical Record: Advances in Integrative Anatomy and Evolutionary Biology 2012; 295: 748-757
- Kocher, O., et al.Identification and partial characterization of a novel membrane-associated protein (MAP17) up-regulated in human carcinomas and modulating cell replication and tumor growth. The American Journal of Pathology 1996; 149: 493-500
- Carnero, A. MAP17, a ROS-dependent oncogene. Frontiers in Oncology 2012; 2: 112
- García-Heredia, J. M., Carnero, A. Dr. Jekyll and Mr. Hyde: MAP17’s up-regulation, a crosspoint in cancer and inflammatory diseases. Molecular Cancer 2018; 17: 80
- Guijarro, M. V., et al. MAP17 inhibits Myc-induced apoptosis through PI3K/AKT pathway activation. Carcinogenesis 2007; 28: 2443-2450
- Carnero, A. MAP17 and the double-edged sword of ROS. Biochimica et Biophysica Acta (BBA) - Reviews on Cancer 2012; 1826: 44-52
- Perez, M., et al. MAP17 and SGLT1 Protein Expression Levels as Prognostic Markers for Cervical Tumor Patient Survival. PLOS ONE 2013; 8: e56169
- Guijarro, M. V., et al. p38[alpha] limits the contribution of MAP17 to cancer progression in breast tumors. Oncogene 2012; 31: 4447-4459
- Garcia-Heredia, J. M., Carnero, A. The cargo protein MAP17 (PDZK1IP1) regulates the immune microenvironment. Oncotarget 2017; 8: 98580-98597
- Garcia-Heredia, J. M., et al. The cargo protein MAP17 (PDZK1IP1) regulates the cancer stem cell pool activating the Notch pathway by abducting NUMB. Clinical Cancer Research 2017; 23: 3871-3883
- Wang, Z., et al. Emerging Role of Notch in Stem Cells and Cancer. Cancer letters 2009; 279: 8-12
Link: https://www.nature.com/articles/s41389-020-00280-0
Follow the Topic
-
Oncogenesis
A peer-reviewed open access online journal that publishes articles exploring mechanistic insight and molecular basis of cancer and related phenomena. It seeks to promote diverse and integrated areas of molecular biology, cell biology, oncology, and genetics.
Related Collections
With Collections, you can get published faster and increase your visibility.
Metabolic Reprogramming in Cancer
The field of cancer metabolism has expanded rapidly, revealing how metabolic reprogramming drives tumour progression, therapy resistance, and cellular adaptation. Beyond local interactions, accumulating evidence shows that circulating immune cells and blood‑borne factors—including metabolites, cytokines, and extracellular vesicles—actively influence tumour metabolic states. These systemic signals interact with tumour‑resident cells to shape metabolic plasticity, modulate survival pathways, and affect treatment responses.
Within the tumour microenvironment itself, metabolic interplay between cancer cells, immune cells, and stromal components remains a central determinant of tumour behaviour. Altered glycolytic flux, lactate‑driven microenvironmental changes, and cancer stem cell–associated metabolic adaptations contribute to tumour aggressiveness, immune evasion, and resistance mechanisms. Together, these local and systemic dimensions provide an integrated view of how metabolism underpins cancer progression.
This collection supports United Nations SDG 3: Good Health & Well-Being.
Topics of interest include:
- metabolic interactions within the tumour microenvironment
- cancer stem cell metabolism
- glycolytic pathways in tumour promotion
- tumour–immune cell metabolic interplay (local and systemic)
- interplay between circulating blood cells and tumour metabolism
- drug resistance and metabolic shifts
- protein expression in metabolic reprogramming
- role of transcription factors in metabolism
- signal transduction pathways affecting cancer metabolism
Publishing Model: Open Access
Deadline: Dec 31, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in