How do cortical networks generate repeating activity patterns?
Published in Neuroscience and Protocols & Methods
Our recent article grew out of an intersection of two research directions that happened to share the same office.
At the time, I was studying the dynamics of large spiking neural networks. My work focused on distance-dependent connectivity networks, based on the observation that neurons are more likely to connect to nearby neighbours than to distant ones. In computational models, this constraint produces rich dynamics: irregular activity, synchronised fluctuations, and waves of activity that propagate across the network.
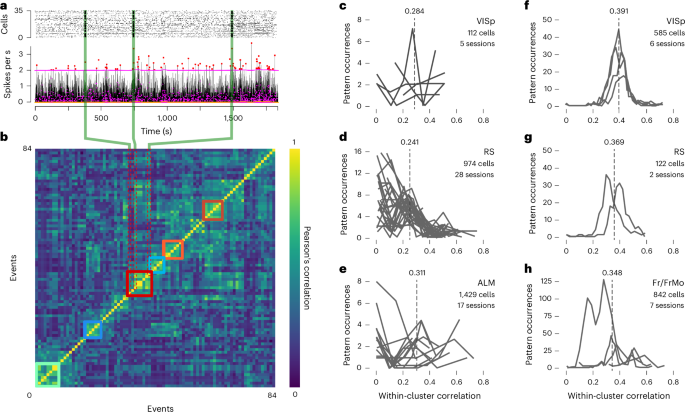
In the same lab, led by Alain Destexhe, Anton Filipchuk was approaching cortical dynamics from an experimental perspective. Using in vivo two-photon calcium imaging in mouse auditory cortex, he analysed how neurons organise into ensembles—groups of cells that repeatedly participate in similar activity events.
Although our approaches were very different—computational modelling on one side, experimental recordings on the other—we quickly realised we were fascinated by the same phenomenon: repeating patterns in cortical population activity.
From ensembles to “core neurons”
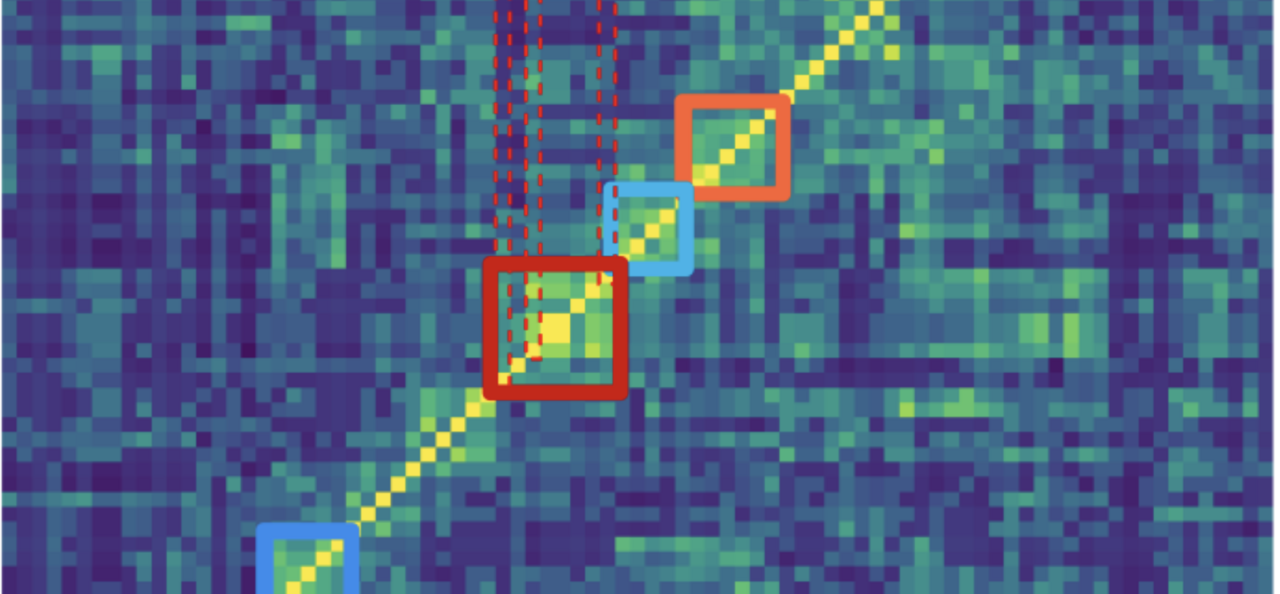
A central concept emerging from the literature on ensembles was that within ensembles, some neurons appear to play a central role. These so-called “core” neurons consistently participate when a given spiking activity pattern appears and seem to anchor the ensemble structure.
At first, this seemed perfectly consistent with classical theories of cortical organization. Since Hebb, repeated neural activity patterns have often been explained through attractor networks—circuits in which neurons belonging to the same pattern are strongly interconnected. If this were the case, neurons within an ensemble—especially core neurons—should show stronger mutual connectivity than average.
And around that time, an extraordinary dataset became available.
A turning point: MICrONS
The MICrONS project (https://www.nature.com/immersive/d42859-025-00001-w/index.html, https://www.microns-explorer.org) began releasing datasets combining two-photon recordings with electron-microscopy reconstructions of synaptic connectivity in mouse visual cortex. For the first time, researchers could examine both the activity of neurons and the precise wiring connecting them.
We immediately turned to this dataset.
If ensembles were stabilised by attractor-like circuits, the connectomic reconstruction should reveal dense connectivity among the neurons forming those patterns.
But the data told a different story.
The neurons repeatedly participating in ensemble patterns were not strongly connected to one another. Their wiring looked similar to that of the surrounding network.
When structure and dynamics diverge
This mismatch forced us to rethink the problem.
In spiking network models with distance-dependent connectivity, activity propagates across the network in cascades or waves. Because of the topology of the network, certain neurons lie along paths that activity frequently travels. These neurons may appear repeatedly in activity patterns—not because they form a special circuit, but because they occupy strategic positions in the network’s structural landscape.
From this perspective, ensembles may not correspond to rigid attractor circuits. Instead, they may reflect recurring pathways of activity through cortical networks.
The value of open datasets
Projects like MICrONS, and the other datasets from Svoboda Lab, Goard Lab, and Carandini Lab, make it possible to test ideas that previously remained theoretical. And they allow us to question established ideas. By combining functional recordings and connectomics, they allow researchers to examine not only how neurons fire together, but also how their wiring supports—or fails to support—those dynamics.
In our case, the results suggested that repeating cortical patterns may arise less from specialised attractor circuits than from the interaction between network topology and neural dynamics.
In a living animal, these recurrent trajectories may be the dynamical backbone that links sensation to action, allowing behaviour to emerge from the flow of activity through cortical networks.
Follow the Topic
-
Nature Neuroscience
A multidisciplinary journal that publishes papers of the highest quality and significance in all areas of neuroscience, with contributions in molecular, cellular, systems and cognitive neuroscience, psychophysics, computational modeling and diseases of the nervous system.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
The Gut-Brain axis and health
Publishing Model: Hybrid
Deadline: Dec 05, 2026
Lifespan changes in the human brain
Publishing Model: Hybrid
Deadline: Jul 31, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in