How Stem Cell Research Is Revolutionizing the Treatment of Androgenetic Alopecia
Published in Bioengineering & Biotechnology, General & Internal Medicine, and Education
Every dermatology clinic specializing in trichology receives patients daily -both men and women- seeking solutions for androgenetic alopecia. Patients are increasingly younger: nearly 30% of men begin to notice it around the age of 30, and its prevalence rises by 10% per decade. Among women, around 40% experience some degree of hair loss by the age of 50. Many see baldness as a mere aesthetic issue, but behind it often lie deeper problems, such as low self-esteem, insecurity… and also an increased risk of skin cancer caused by the early loss of the hair’s natural protection against sunlight.
There are, in fact, solutions for androgenetic alopecia, such as pharmacological treatments (minoxidil and finasteride) or hair transplants. However, not all patients feel comfortable with the long-term use of drugs, nor are they all candidates for invasive surgical procedures. In any case, over the past decades we have witnessed how available therapies could slow down hair loss (provided the patient was a suitable candidate), but could not restore damaged follicles. That led us to ask ourselves: what if we could regenerate the follicles?
This story began with my research using an energizing molecule to improve the survival of follicular units in transplants -a key factor in achieving the best possible outcome. This work, which I successfully applied in my practice at Clínica Imema in Madrid, earned an R&D&I certification and the Innovative SME seal for the protocol “New Approach for the Treatment of Androgenetic Alopecia.” At the same time, in the corridors of Hospital Clínico San Carlos in Madrid, colleagues from the Cellular Therapy Research Unit introduced us to the potential of stem cells in other diseases.

But what about in androgenetic alopecia? Could stem cells, combined with a molecule that provides them with energy to enhance their survival “awake” the “dormant” follicles and achieve lasting hair growth? A hypothesis had been born -and with it, a goal: to reverse alopecia.
Launching a study of this kind was not easy. Androgenetic alopecia does not occur spontaneously in mice, so we had to develop a model to induce it in a controlled way. This involved determining the appropriate age of the animals, ensuring that the mice adapted properly to the laboratory environment, and defining the method for administering the inducing agents. In addition, the skin of mice is much thinner than that of humans, and their hair density is much higher, which made both the administration of the products and the evaluation of hair growth more challenging, as it tended to “saturate” the digital analysis methods. In any case, we were able to demonstrate that androgenetic alopecia can be induced in mice by administering dihydrotestosterone -the same hormone responsible for this condition in humans.
Additionally, stem cells have a limited lifespan outside the body, and from our experience with transplants we knew that hair follicles need time to establish a vascular network in the recipient tissue if they are to survive. To maximize their chances, we administered an energizing molecule, adenosine triphosphate (ATP) which prolongs cell survival, giving stem cells time to release growth factors and differentiate. In this way, if the stem cells could survive long enough in the scalp, they could regenerate damaged follicles and reverse baldness.
The study design included 12 experimental groups: a control group (sham), a reference group (wild type), a placebo group, and nine experimental groups. Among the latter, one group received only stem cells; two groups were treated exclusively with adenosine triphosphate (liposomal and non-liposomal); and six groups combined stem cells (at high, medium, and low doses) with adenosine triphosphate, either liposomal or non-liposomal.
Each group was evaluated using photographic observation models, digital hair growth analysis and histological verification to confirm that visible changes corresponded to follicular regeneration at the microscopic level. We also developed specific statistical models to interpret the results, as variability among animals and hair density required a rigorous quantitative approach.
The results: among male mice treated with a low dose of stem cells and adenosine triphosphate, up to 50% achieved complete hair regrowth and the other 50% showed intense regrowth, thus reaching a 100% success rate in hair regeneration. In females treated with a medium dose, up to 50% achieved total regrowth and 40% intense regrowth, meaning that 90% of the female mice experienced significant hair growth.
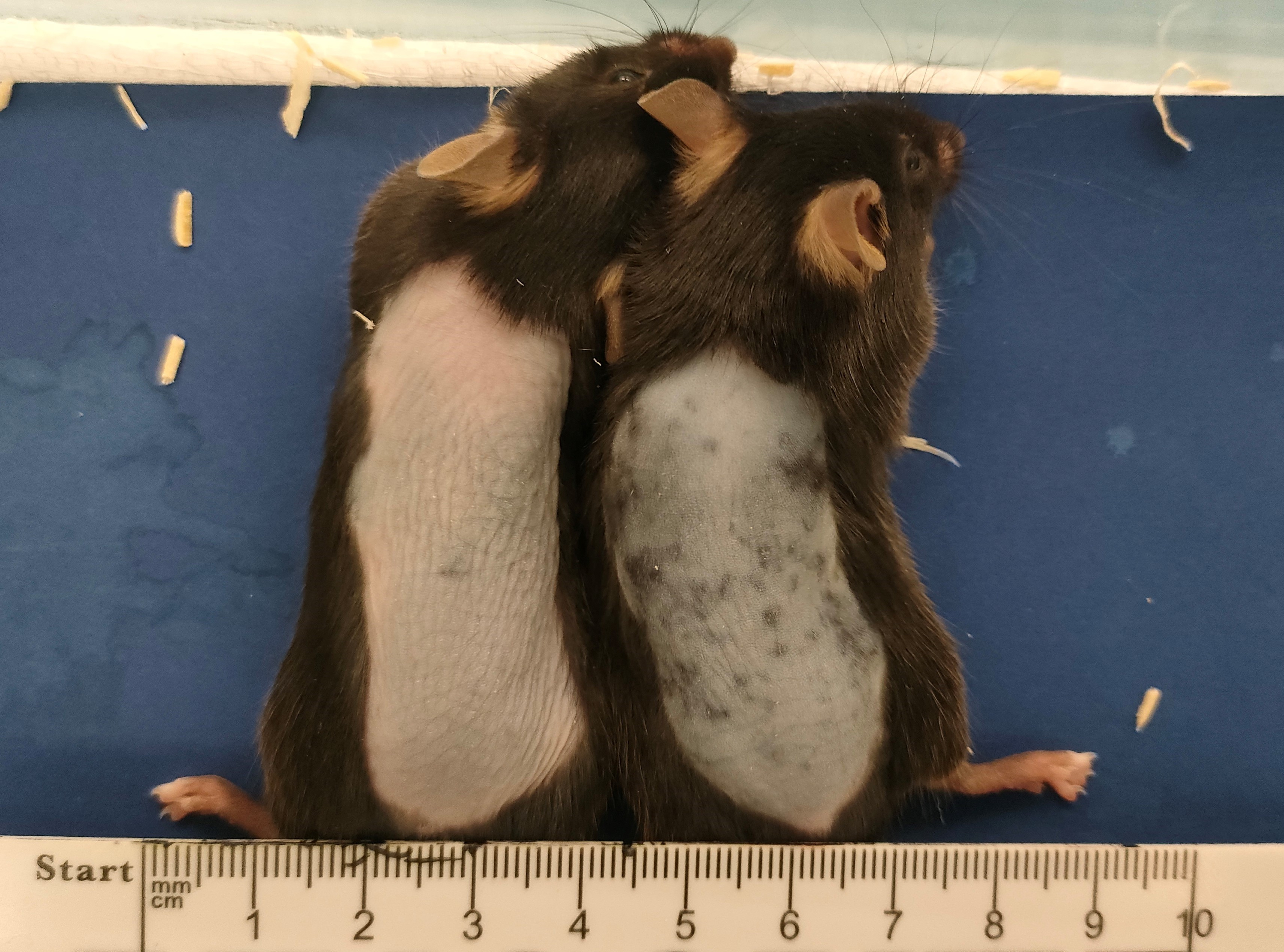

Mice in day 10 (first picture) and day 21 (second picture).
These results were undoubtedly unexpected. Compared to current treatments, which depend on continuous use and offer much more modest outcomes, here we are seeing intense and -apparently- long-lasting growth. This opens up a horizon of hope that did not exist before. It has been nearly 40 years since the first effective drug for alopecia was marketed, and during all this time, pharmacological options have shown limited efficacy and dependence on ongoing use. Now, we are opening an entirely new path: an advanced therapy capable of regenerating follicles and delivering long-term results.
Follow the Topic
-
Stem Cell Research & Therapy
The major forum for translational research into stem cell therapies. An international peer-reviewed journal, it publishes high-quality open access research articles with a special emphasis on basic, translational and clinical research into stem cell therapeutics and regenerative therapies.
Related Collections
With Collections, you can get published faster and increase your visibility.
Immune re-programming and long-term effects mediated by mesenchymal stromal cell therapy
A significant body of evidence suggests that culture expanded mesenchymal stromal cells (MSCs) are short-lived, paracrine signalers that briefly calm inflammation following in vivo administration before disappearing. However, emerging preclinical and early clinical evidence suggests that MSCs may also induce longer-term immunological changes through mechanisms such as trained immunity and epigenetic imprinting of hematopoietic or precursor stem cells in the bone marrow.
This Collection welcomes original research or review articles describing novel findings and insights on immune re-programming mediated by MSCs and evidence of long-term effects in vivo.
All submissions in this Collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Dec 02, 2026
Pluripotent stem cell-enabled therapies for neurological disorders
The discovery of pluripotent stem cells (PSCs) has empowered the production of large quantities of desired human cell types. With methodological advancements, PSCs can be differentiated into multiple cell types of the brain and spinal cord, including dopaminergic, GABAergic, serotonergic, cholinergic, and glutamatergic neurons, as well as astrocytes, microglia, and oligodendrocytes, paving the way for several novel findings for neurological disorders. Apart from 2D models, over the past few years, PSC-derived brain organoids have also provided an unprecedented opportunity to decode the aetiology of many neurological disorders at the cellular and molecular levels. Further, PSC-derived cells have been extensively tested for their efficacy in treating various neurological disorders by replacing damaged or dysfunctional cells, thereby creating opportunities for PSC-based cell therapies. Based on preclinical data, several pluripotent stem cell-based therapies are currently under clinical trials. These PSC-derived cells and models have also facilitated the testing of repurposed drugs, novel drugs, antioxidants, anti-inflammatory compounds, epigenetic modulators, DNA/RNA-based therapeutics, small-molecule inhibitors, natural products, and nutraceuticals to promote neural regeneration. Additionally, PSC-derived cells and products derived from PSCs, such as extracellular vesicles, are being investigated for their potential as drug delivery vehicles or bioengineered for the nano delivery of specific biomolecules to treat brain disorders. Overall, PSCs, derived cells and brain organoids have provided multiple therapeutic opportunities for treating neurological disorders.
To expand our understanding of the translation aspects of PSC-enabled therapies for neurological disorders, we invite researchers to contribute to this Collection by submitting original research articles in the following areas. A limited number of high-quality review articles will also be considered.
Possible topics include, but are not limited to:
- Robust cell transplantation studies of PSC-derived progenitor cells in established preclinical models of neurological disorders
- Studies identifying the durability and long-term safety of PSC-based therapies.
- Evaluation of methods/molecules that support abolishing PSCs in the transplant pool.
- Improved immunosuppression strategies for preventing transplant rejection.
- Directed differentiation protocols that yield high-purity cells for transplantation.
- Therapeutics identified from a high-throughput screening of drugs for neurological disorders based on PSC-derived models.
- PSC-derived models used for patient-specific drug screening.
- Biomarker discovery for neurological diseases identified through PSC-derived models.
- Pharmacological interventions for neurological disorders through drug-loaded extracellular vesicles derived from PSC-derived cells.
- Impact of bioengineered EVs derived from iPSC-derived cells for neurological disorders.
- Identification of novel pathophysiology of neurological diseases identified through PSC-derived models that open significant therapeutic opportunities.
- PSC-based studies that identified repurposed drugs, novel drugs, antioxidants, anti-inflammatory compounds, epigenetic modulators, DNA/RNA-based therapeutics, small-molecule inhibitors, natural products, nutraceuticals, etc, to promote neural regeneration.
- Any other relevant original contributions.
All submissions in this Collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Jan 15, 2027





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in