Human embryonic stem cells – from derivation to clinical regenerative applications
Published in Bioengineering & Biotechnology
The enormous potential of human pluripotent stem cells for the study of early human development, disease modeling, drug discovery, and regenerative therapy was appreciated from the early days of derivation of mouse embryonic stem cells (ESCs). Both ethical constraints on the use of human embryos for research, and evolutionary differences in stem cell biology between mice and humans impeded attempts to develop ESCs from human embryos. Therefore, our success to develop ESCs from surplus in vitro fertilized human embryos, which was published in the year 2000 in Nature Biotechnology (NBT) 1, was an important scientific and medical landmark, and a rewarding accomplishment for all co-authors who participated in the study.
As a young obstetrician and gynecologist with specialties in in vitro fertilization (IVF), my main vision upon joining this project with the research group of Professors Alan Trounson and Martin Pera in Australia, collaborating with Prof. Ariff Bongso from Singapore, was the opportunity to perform research in the area of IVF that would have broad scientific and medical implications. Given my medical background, I was most thrilled by the potential use of human ESCs (hESCs) for regenerative therapy.
Ethical considerations made research in the realm of human embryos difficult to navigate in Australia. Therefore, I travelled to Singapore to perform the initial isolation of the inner cellular mass (ICM) of pluripotent stem cells from surplus donated human embryos. I then flew back to Australia with the isolated ICMs in a small flask in my pocket. It was an exciting adventure that was covered within an editorial review in the journal Science 2.
The derivation of hESC lines and their publication in NBT had tremendous impact on shaping my research career. It paved my way to the establishment of the Sidney and Judy Swartz hESC Research Center at the Goldyne Savad Institute of Gene Therapy at my home institution, the Hadassah University Medical Center in Jerusalem. Given my great enthusiasm for regenerative therapy, we have been focusing on overcoming the barriers of developing hESCs for clinical transplantation therapy ever since. Nevertheless, it is worth noting that in the early days, cultivation of hESCs was work intensive, on a low scale, and with significant spontaneous differentiation, hence their potential use for cell therapy appeared beyond reach.
Our first step towards fulfilling the goal of cell therapy was the derivation of new hESC lines that would be suitable for clinical applications. At that time, the existing hESC lines were only suitable for research purposes. We were first to develop hESC lines without the use of animal-derived reagents and under GMP conditions 3, with the vision that they would serve the research community worldwide as starting materials for developing clinical transplantation applications. We have been providing these cell lines to multiple academic research groups and companies that are developing cell therapy products globally. It is rewarding that the lines are already in clinical trials with two projects-- for age-related macular degeneration (AMD, at Cell Cure Neurosciences Ltd), and for amyotrophic lateral sclerosis (ALS, at Kadimastem Ltd).
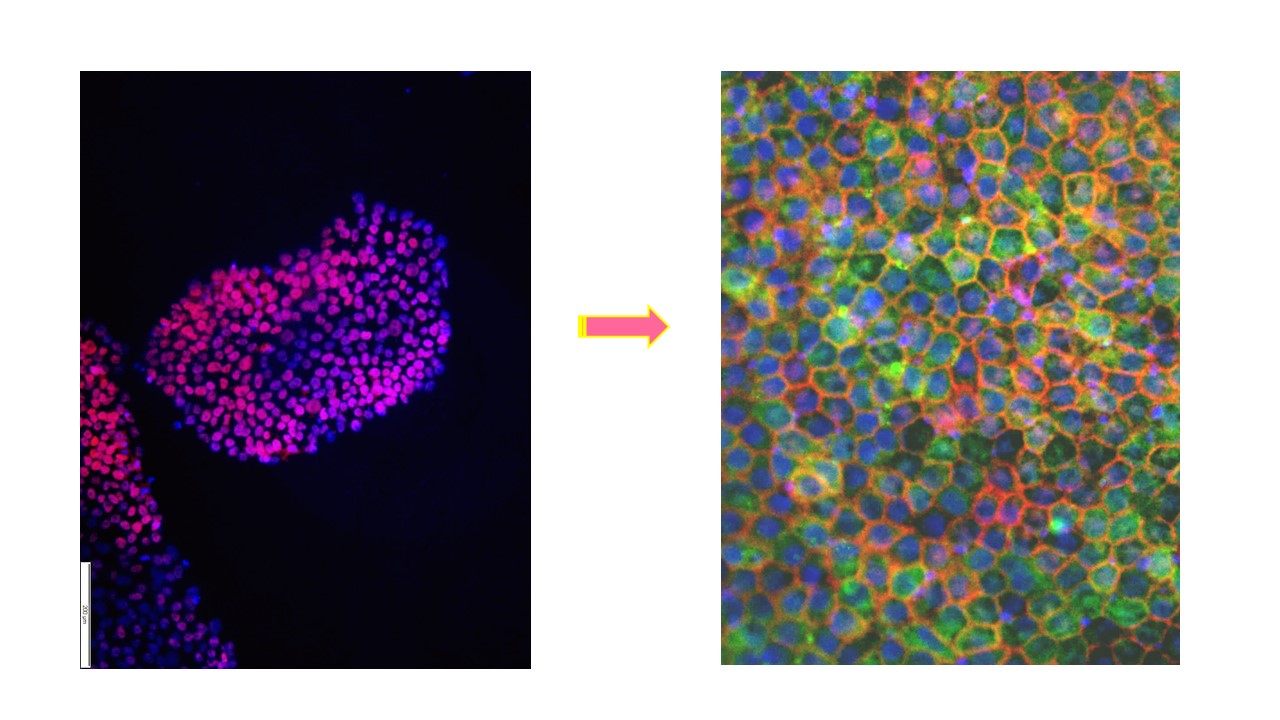
In the next step, we were searching for a clinical indication that may be a suitable candidate for developing a hESC-based cell therapy. AMD evolved in our hands as the most promising indication. Age-dependent progressive loss of function and degeneration of retinal pigment epithelium (RPE) cells, in AMD patients, leads to degeneration of photoreceptors, which causes progressive vision loss and eventual legal blindness. Given the lack of an FDA-approved effective treatment for AMD, indications in the literature suggesting that RPE regeneration may halt progression of AMD, and our success in efficiently directing the differentiation of hESCs into functional RPE cells 4, led us to focus in collaboration with Professor Eyal Banin from the Department of Ophthalmology at Hadassah, on developing hESC-derived RPE cells for transplantation in AMD.
In conjunction with Cell Cure Neurosciences, Ltd (1) that I co-founded (currently a fully owned subsidiary of Lineage Cell Therapeutics, Ltd.), we successfully manufactured batches of RPE cells, under xeno-free and GMP conditions. Subject to approvals by the Israeli Ministry of Health and the FDA, Cell Cure launched a multi-center phase I/IIa clinical trial of RPE cell transplantation in advanced dry-AMD in Israel and the USA. Initial results from the clinical trial are encouraging (2).
The beginning of my journey in the area of pluripotent stem cells began with the derivation of the hESC lines and their publication in NBT. We were fortunate to contribute to the scientific advancement of this field and to provide both our initial research-grade and the clinical-grade lines to multiple research groups worldwide. It has taken many years to cross barriers from the initial derivation of hESC lines to clinical trials, and we have been privileged to be part of this endeavor. I am optimistic that in the coming years, the field will cross the barrier of phase II and III clinical trials, and the vision of off-the shelf hESC-derived products for regenerative therapy will become a reality.
REFERENCES:
- Reubinoff, BE, Pera MF, Fong CY, Trounson A, Bongso, A. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat Biotechnol, 2000 Apr; 18(4):399-404. doi: 10.1038/74447
- Vogel, Gretchen. In the Mideast, Pushing Back the Stem Cell Frontier. Science, 08 Mar 2002; vol. 295, Issue 5561, pp. 1818-1820. DOI: 10.1126/science.295.5561.1818
- Tannenbaum SE, Turetsky TT, Singer O, Aizenman E, Kirshberg S, Ilouz N, Gil Y, et al. Derivation of Xeno-Free and GMP-Grade Human Embryonic Stem Cells – Platforms for Future Clinical Applications. PLoS ONe, June 20, 2012, https://doi.org/10.1371/journal.pone.0035325
- Idelson, M, Alper R, Obolensky, A, Ben-Shushan, E, Hemo, I, Yachimovich-Cohen, N, Khaner H, et al. Directed differentiation of human embryonic stem cells into functional retinal pigment epithelium cells. Cell Stem Cell. 2009 Oct 2; 5(4):396-408. doi: 10.1016/j.stem.2009.07.002
Follow the Topic
-
Nature Biotechnology
A monthly journal covering the science and business of biotechnology, with new concepts in technology/methodology of relevance to the biological, biomedical, agricultural and environmental sciences.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in