Innate immunity and lipids: resistance to immunotherapy in the peritoneal cavity
Published in Cancer, Biomedical Research, and Immunology

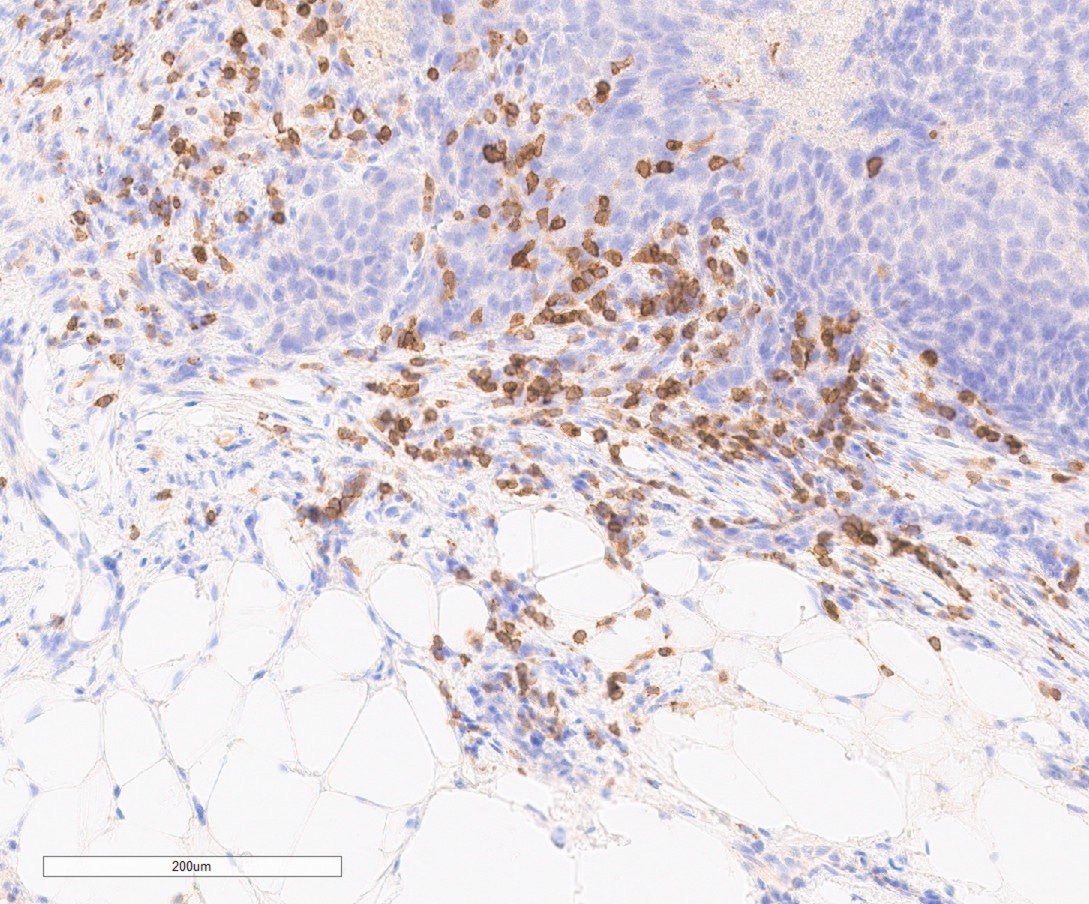
Immunohistochemical image (CD3 staining for T cells) of an ovarian cancer section with adipose cells in the lower part and tumor cells in the upper part with ample T cells sitting at the interface between the two compartments.
Breaking up resistance to immunotherapy: the two sides of adipose tissue in the tumor microenvironment of the abdominal cavity
„What drives resistance to immunotherapy?“ was the initiating question of an exploration of the immunologic specifics of the tumor microenvironment. Seeing checkpoint-inhibitors generating so much clinical benefit in malignant melanoma patients and on the other hand practically no benefits in advanced metastatic (maicrosatellite stable) colorectal cancer patients clearly raised this question. While there was a plethora of data indicating the impact of infiltrating T cells in the tumor environment in colorectal cancer (Galon et al. Science 2006), treatment with checkpoints did not work for the vast majority of microsatellite stable colorectal cancer cases. This obvious divergence between different tumor entities offered a glimpse into the complexities of how the tumor microenvironment can differ between tumors. With the view on the resistance of immunotherapy especially in liver and peritoneal metastases, the central point emerged: what is the difference in the specific organ-microenvironments that makes it so difficult for immunotherapy? A shared environment between the immunologically „cold“ (or hard-to-treat) tumors is the peritoneum or the abdominal cavity. The abdominal cavity is also challenging field for oncology, having a huge unmet medical need as it is hard to pursue resections and patients typically have severe symptoms from tumor progress there. Seeing that there are (some) patients with ovarian cancer that do respond to immunotherapy put the focus on this tumor: what do we see in the microenvironment? Again, previous work showed that T cells can infiltrate there as well (Zhang et al. NEJM 2004), but why is immunotherapy so limited in it´s effect then? Histopathology offers a look into this microenvironment that is based on morphology and phenotypes: patterns inside the tissue. With this expertise, a detailed look inside the immunological landscape of ovarian cancer confirmed, what we had seen in liver metastases of colorectal cancer (Halama et al. Cancer Res 2012, Halama et al. Cancer Cell 2016): there are „islands“ (i.e. conglomerates) of T cells, especially in the vicinity of adipose tissue. But how does „fat“ contribute to these „islands“? With a deeper look at phenotypes, the peculiar type of macrophages emerged clearly, showing off their lipid inclusions and at the same time assembling T cell crowds around. An intriguing observation that led to the next explorations.
Utilizing the fully human explant model system: macrophages as orchestrators
So to study this from a functional perspective, we utilized our fully human model system, which was already validated in producing results that could be seen in comparison to actual human patients with the same treatment (Halama et al. Cancer Cell 2016). With this model system at hand, the systematic assessment of the environment in a diverse set of ovarian cancer samples followed and it became clear: macrophages orchestrate in both directions. For T cells by providing a better „environment“ for T cells to thrive, but at the same time binding T cells to specific regions, not directly in the tumor. So this prompted the idea of a „dichotomy“, with adpiose tissue at the same time helping T cell responses but also in many cases limiting the anti-tumoral activity. This also allowed to come to understand that „resistance“ is not a single one-way-street but could appear in the form of an interlocking network.
This brought up the question, whether this influence on macrophages to „orchestrate“ the immune response can be modulated. And this revealed that indeed, many factors come into play. As the innate arm of the immune system is a complex hub for interactions, the effect of curbing inflammatory signals was also coming into sight. Due to the rapid access program, the material from the operation room could be directly used to „fish out“ an interesting microbial species from multiple patients: Bacteroides vulgatus. This allowed to better integrate the view on the modulation of the innate arm of the immune system: not only lipids, but also inflammation (triggered via bacteria sensing) is contributing to the bigger picture. In a disease, where the abdominal cavity is beset with spreading cancer, the barrier function (e.g. towards the gut) is often compromised and (low-grade) inflammation can impact the abdominal cavity compartment. So it became clear that multiple (clinically relevant) factors weigh in on the activation (or „polarization“) of lipid-laden macrophages. But can this be also identified in another model system? Utilizing the possibility of the humanized mouse platform, the findings could be replicated and further explored, going beyond the observations in human epithelial ovarian cancer, revealing a conceptual framework of lipid-laden macrophage modulation.
From models to humans: using scientific insights to stratify patients
To look beyond the animal model and the more generalizable findings there, the translational angle moved into the focus again. This is twofold: one is the proof-of-concept for radiology to identify ovarian cancer patients that potentially could respond to checkpoint-inhibition and the other of course is to think about the way we can modulate the environment. As the presence of many of these „islands“ of T cells induced by lipid-laden macrophages identified a more favorable microenvironment, the access to the trial samples from the immunotherapy trial run by Junzo Hamanishi allowed to test this hypothesis: can we „predict“ which patient might respond to the checkpoint-inhibitor? Having received the „blinded“ samples, it turned out that the prediction was very accurate, based on the histopathology alone. But what if there is no extensive histopathological specimen? For the radiology approach, a random encounter with Vickie Baracos sparked this idea. Her interest in cachexia and expertise in abdominal imaging to measure fat deposits and distribution brought up the idea of having a non-invasive tool to „predict“ immunotherapy response. Utilizing this approach in the clinic, the proof-of-concept provided valuable insight as indeed the stratified patient, receiving the checkpoint-inhibitor, responded. But it is also clear at the same time, that this (so far limited) tool needs improvement and better technologies and more cases will allow to expand and develop this approach.
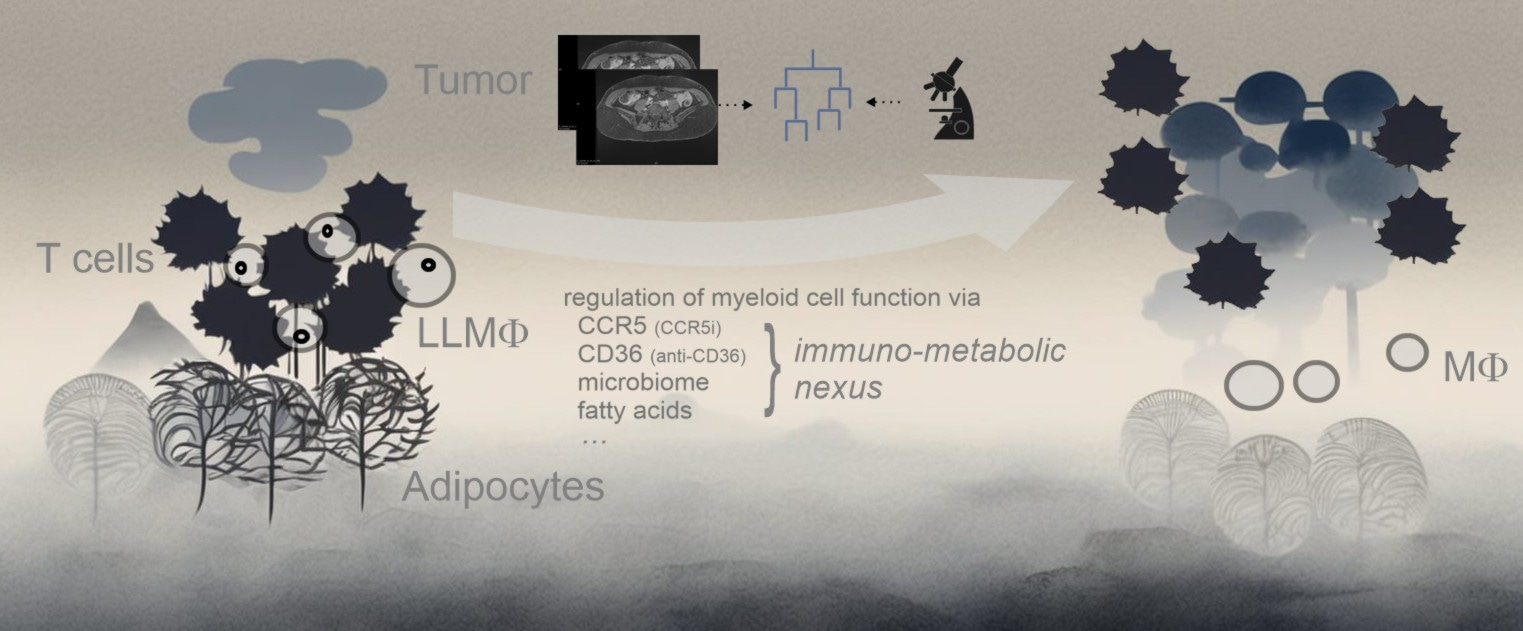
Conclusion: the challenges and opportunities of innate immunity modulation
So this work not only exemplifies how the complex regulation of macrophages affects immunotherapies, but it also clarifies how lipid metabolism influences oxidativ stress and with it the reaction to inflammatory stress. It not only identifies lipid-laden macrophages as central regulators of immune responses and resistance, but also makes clear: adipose tissue plays an important but dichotomous role. Supporting T cell responses and at the same time „keeping the T cells out“. These findings underscore the improved understanding of immunobiology of the innate immune system and also points to new diagnostic possibilities and new aspects of immunomodulation. The time for synergistic immunomodulation has come.
Follow the Topic
-
Signal Transduction and Targeted Therapy
This is an international, peer-reviewed, open-access journal publishing articles related to signal transduction in physiological and pathological processes, alongside signal transduction-targeted therapeutics in the form of biological agents and small molecular drugs used to treat human diseases.
Ask the Editor - Immunology, Pathogenesis, Inflammation and Innate Immunity
Got a question for the editor about the complement system in health and disease? Ask it here!
Continue reading announcement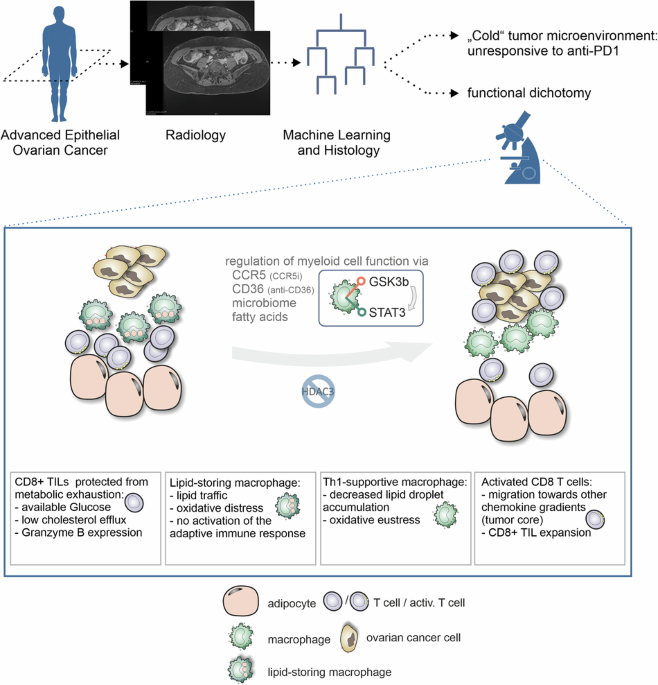




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in