Instantaneous ex situ mineral carbonation in clays under ambient conditions
Published in Chemistry

Owing to the significant climate change caused by the steady increase of CO2 in the atmosphere along with industrial activity, the technology of CO2 capture and storage with respect to less-energy intensiveness, cost effectiveness, and environmental friendliness has been long-awaited. In power plants, electricity is generated by burning fossil fuels such as coal and natural gas, which could be a large point source through the combustion process. An approach to reduce CO2 emission is the efficient capture of CO2 and its storage in a stable manner before release to the atmosphere. An important factor of CO2 capture and storage is the long-term sequestration of CO2, where CO2 storage in geological reservoirs has been often considered. In this approach, CO2 is injected into a deep geological formation to be physically confined below an impermeable or very low permeability caprock, such as a shale, allowing for a sequence of possible trapping mechanisms. A fraction of injected CO2 is fixed as thermodynamically stable mineral carbonates, as e.g., CaCO3 or MgCO3 in the geological formation via the reaction with alkaline minerals there. The formation of stable carbonates in the deep underground geology, known as in situ mineral carbonation, could be suitable for long-term storage of CO2. On the contrary, ex situ mineral carbonation is the above-ground process involving the reaction of CO2 with alkaline earth metals extracted from basic rock.
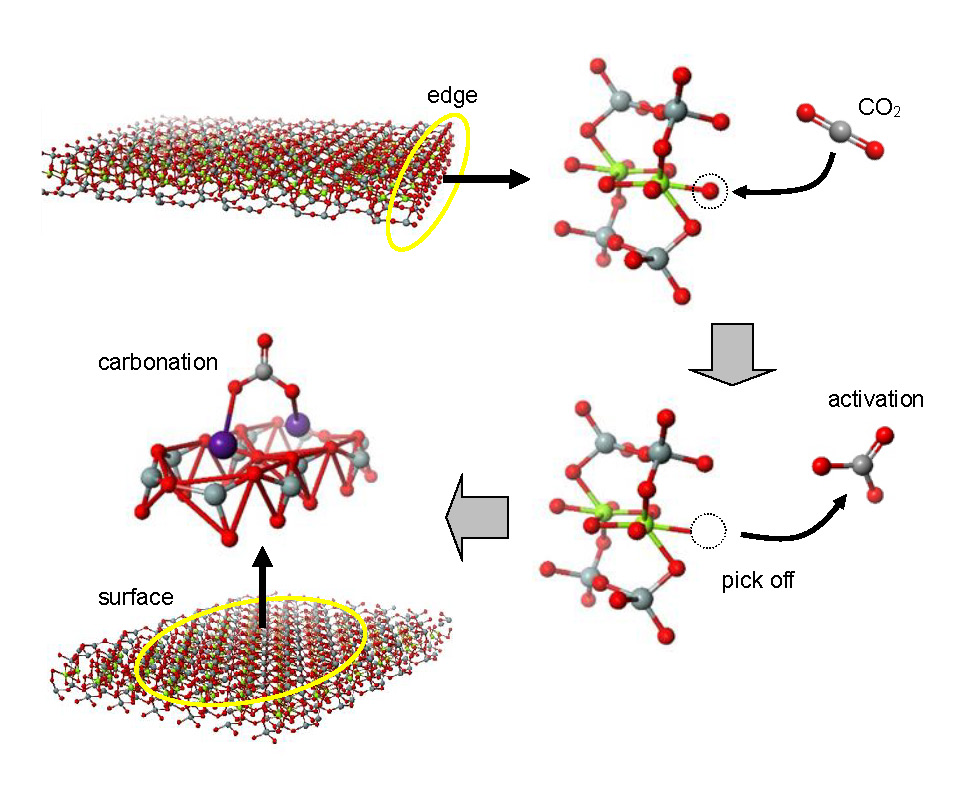
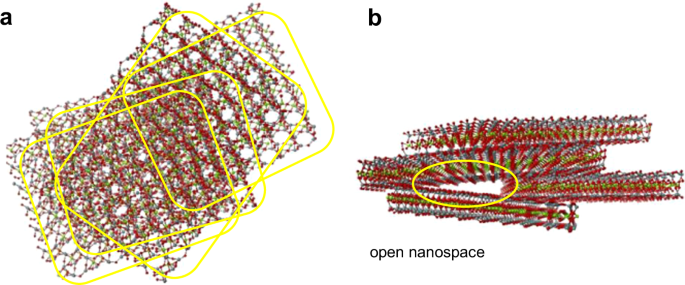
Saponite, a silicate clay mineral abundantly and ubiquitously available in nature, is structured through stacks of two dimensional (2D) nanosheets with thicknesses of a few nanometers, which are the minimum structural unit. The 2D nanosheets have a variety of sizes and cannot be perfectly stacked, resulting in partial overlapping. This results in the formation of nanoscale open spaces. Here, CO2 adsorption in the open spaces originated from overlapped nanosheets in saponite clay minerals is explored by solid-state nuclear magnetic resonance coupled with open space analysis using positronium. The following scenario of instantaneous ex situ mineral carbonation under ambient conditions was found. In saponite, the O atoms are weakly bound to the octahedron by ionic bonding, in contrast to the strong semi-covalent bonding of O and Si atoms in the tetrahedron. CO2 gas molecules thus easily pick up the O atoms from the octahedra at the nanosheet edges in the interior of the above open spaces to be activated as the carbonate ions of CO32− type (see right-hand side). The CO32− ions are then chemisorbed at the alkali metal cations on the surface of 2D nanosheets (see left-hand side). This instantaneous mineral carbonation and CO2 physisorption occurs in the absence of an energy-consumption process or chemical solution enhancement. The present finding is of potential significance for CO2 capture and storage and presents an approach of environmentally friendly recycling of low contaminated soil in Fukushima.
Kiminori Sato & Michael Hunger
Carbon dioxide adsorption in open nanospaces formed by overlap of saponite clay nanosheets, Kiminori Sato and Michael Hunger, Communications Chemistry 3, 91 (2020).
DOI: https://doi.org/10.1038/s42004-020-00346-5
Follow the Topic
-
Communications Chemistry

An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Experimental and computational methodology in structural biology
Publishing Model: Open Access
Deadline: Apr 30, 2026
Advances in Asymmetric Catalysis for Organic Chemistry
Publishing Model: Open Access
Deadline: Mar 31, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in