Integrative analysis of kindlins, the mechanochemical super-adapters in cancer
Published in Cancer, Genetics & Genomics, and General & Internal Medicine
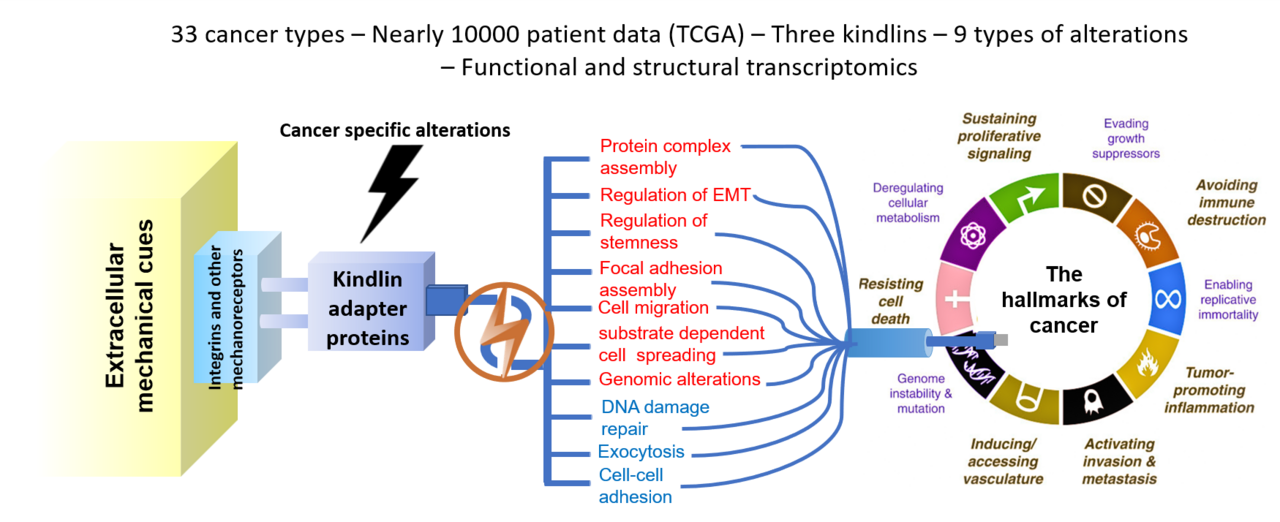
In the ever-evolving landscape of cancer research, understanding the intricate molecular mechanisms driving tumor progression is paramount for developing effective therapeutic strategies. Mechanical cues are pivotal in shaping tumor onset, progression, metastasis, and recurrence1. However, the processes by which these cues are converted into intracellular chemical crosstalk remain elusive. Specifically, mechanical adapter proteins such as kindlins, which translate mechanical cues into chemical signals, still require further study2. Central to multiple cellular functions, primarily through their involvement in integrin regulation3, kindlins consist of three FERM domain-containing proteins – FERMT1 (kindlin1), FERMT2 (kindlin2), and FERMT3 (kindlin3)4. Previous studies on FERMT genes have demonstrated the development of tissue-specific pathologies upon generating knockouts and mutants5. Dysfunctional kindlin proteins, via their integrin-binding activity, are also implicated in cardiovascular conditions, neurodegeneration, and cancer6. We embarked on a journey to explore the role of kindlin family proteins in cancer, shedding light on their significance as mechanochemical adapters in the complex signaling of cancer biology. Due to their involvement in numerous signaling pathways and biological activities, we performed a comprehensive analysis using whole transcriptome and proteome data from large patient cohorts (e.g., TCGA). Our study aimed to unravel the role of kindlins in connecting external mechanical cues with internal cellular events, thereby influencing tumor heterogeneity, chemoresistance, and relapse.
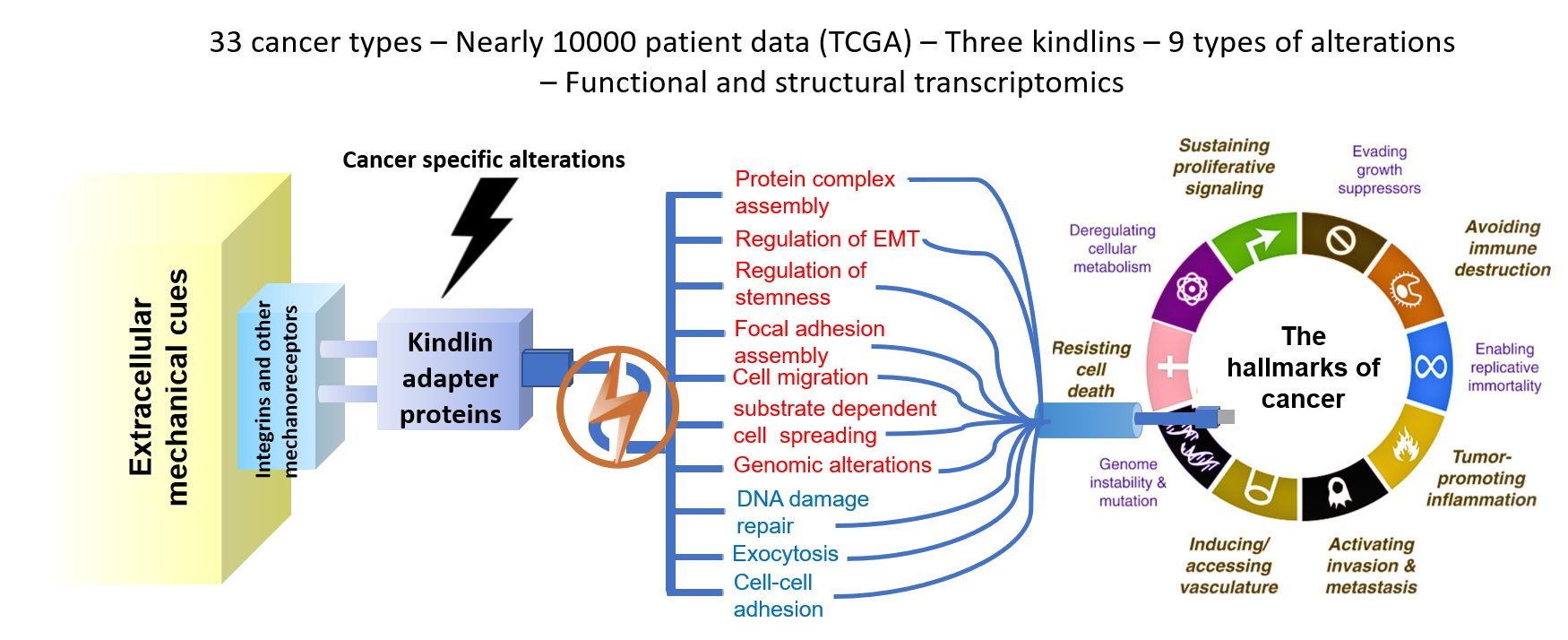
Given their widespread involvement, we first examined the somatic mutation and mRNA expression profile for all the FERMT genes across multiple cancer types. This preliminary analysis set us up for the bulk of our study. A mutated profile with altered mRNA and protein expression levels, as well as a poor overall and relapse-free survival prognosis across multiple cancers, begged the question – what is the potential mechanism of pathogenicity conferred via mutations in kindlin proteins? Akin to a mechanosensory cell, kindlins can transduce mechanical cues from a range of structural proteins and receptors to their downstream interactors, almost acting like signaling bridges between the cell interior and the extracellular milieu. We hypothesized that perturbed signal reception and subsequent transduction could be at the heart of this pathogenicity. To develop this idea, we first generated structural templates for all three Kindlin family proteins and introduced mutations using in silico platforms corresponding to those recorded in patients. We tested how stability parameters for the protein’s global conformation and its region-specific flexibility were impacted for all three candidate proteins. We also computed domain-specific changes in signal reception and transduction for all mutants. Next, by performing docking studies, we tested how these structural alterations might impact their ability to interact with their direct interactors. Together, our structural studies demonstrate a broadly destabilized interaction state with most partner proteins, pointing at an inability to transduce signals downstream efficiently.
The other half of our study adopted a more panoramic view, where we asked how this perturbed interaction profile and subsequent signal transduction function could ultimately impact downstream cellular functions. To develop this line of thought, we analyzed the cell-wide interactome of Kindlin family proteins, pinpointing direct and indirect interactors. This exploratory approach revealed an incredibly vast nexus of interlinked pathways via the involvement of kindlin proteins. Utilizing whole-genome correlation network analysis (WGCNA), we identified kindlin alteration-specific gene clusters that were predominantly perturbed compared to the unaltered kindlin cohort. Employing DEseq (differential expression analysis) and mechanointeractome analysis, we further identified mechanosensitive and mechanotransducing protein genes that were predominantly co-altered with kindlin alterations. Using these findings as inputs, we constructed a kindlin-specific network to elucidate the potential effects of FERMT alterations on cancer hallmark signatures. Our analysis revealed an interesting finding – FERMT alterations significantly upregulated genes implicated in epithelial-to-mesenchymal transition (EMT). This struck us in two ways – first, given that kindlin proteins are focal-adhesion proteins interacting with integrins, one would expect to see an enrichment of genes implicated in cellular migration. Second, if that was the case, we should see most cancer-associated mutations from patients in the late-primary to early-metastatic stages. Our patient data analysis showed the same trend and validated our hypothesis. Overall, this part of our study allowed us to attribute the precise role of FERMT alterations in cancer and, more specifically, cancer progression.
Even so, exciting findings from our modules require further in vitro and in vivo characterization to fully validate our ideas. That does not, however, take away from the impact of our work, potentially laying the foundation for multiple cancer-associated projects involving the kindlin family of proteins in the future.
References:
- Chaudhuri, P. K., Low, B. C. & Lim, C. T. Mechanobiology of Tumor Growth. Chem. Rev. 118, 6499–6515 (2018).
- Rognoni, E., Ruppert, R. & Fässler, R. The kindlin family: functions, signaling properties and implications for human disease. J. Cell Sci. 129, 17–27 (2016).
- Li, H. et al. Structural basis of kindlin-mediated integrin recognition and activation. Proc. Natl. Acad. Sci. 114, 9349–9354 (2017).
- Meves, A., Stremmel, C., Gottschalk, K. & Fässler, R. The Kindlin protein family: new members to the club of focal adhesion proteins. Trends Cell Biol. 19, 504–513 (2009).
- Zhan, J. & Zhang, H. Kindlins: Roles in development and cancer progression. Int. J. Biochem. Cell Biol. 98, 93–103 (2018).
- Plow, E. F. & Qin, J. The Kindlin Family of Adapter Proteins. Circ. Res. 124, 202–204 (2019).
Follow the Topic
-
Communications Biology

An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Your space to connect: The Cancer in understudied populations Hub
A new Communities’ space to connect, collaborate, and explore research on Cancers, Race and Ethnicity Studies and Mortality and Longevity!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
From RNA Detection to Molecular Mechanisms
Publishing Model: Open Access
Deadline: May 05, 2026
Signalling Pathways of Innate Immunity
Publishing Model: Hybrid
Deadline: May 31, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in