Jumping genes spark bacterial evolution
Published in Microbiology and Genetics & Genomics

One of the most fascinating characteristics of bacteria is their exceptional ability to rapidly evolve. Although captivating, this aptitude turns out to be extremely threatening when it comes to antimicrobial resistance (AMR). Several reports from the last decades have shown how easily bacteria can evolve resistance to new antibiotics as they are either developed or discovered and introduced in the clinic. It is not surprising then that the emergency of AMR is nowadays considered as one of the main menaces in public health at world-wide scale. In this context, conjugative plasmids have gained importance as one of the main AMR disseminators due to their capacity to spread among diverse bacterial strains. Understanding how these plasmids impact the way in which their bacterial hosts evolve is crucial to tackle what is now considered the AMR “silent pandemic”. However, there is limited information on which factors determine the evolution of clinically relevant plasmid-bacteria associations. Hence, in the PBE lab, we try to close the gap between the basic science behind plasmid biology, and its applied implications in real clinical scenarios. When I arrived at the PBE lab a couple of years ago, the main objective of my master's thesis was precisely to decipher how a clinically relevant conjugative AMR-plasmid impacted the evolution of its bacterial hosts. Hands down, this would have been a much more difficult task without the support of Dr Alvaro San Millan, PI at the PBE lab, and Dr Alfonso Santos Lopez, PI at the Santos-Lopez lab.
To do so, we counted with an ideal dataset: isogenic pairs (same clone carrying/not carrying such plasmid) of resistant enterobacteria directly isolated from hospitalized patients. With these, we performed an experimental evolution (EE) assay in which we propagated these bacteria both with and without the plasmid in the same conditions for 15 days. This settled the perfect experimental design to study how an AMR-plasmid can alter the evolution of clinical enterobacteria. Sequencing the whole genome of the evolved populations, both with and without the plasmid, would allow us to analyze which genetic alterations are responsible for the success of this clinically relevant AMR-plasmid.
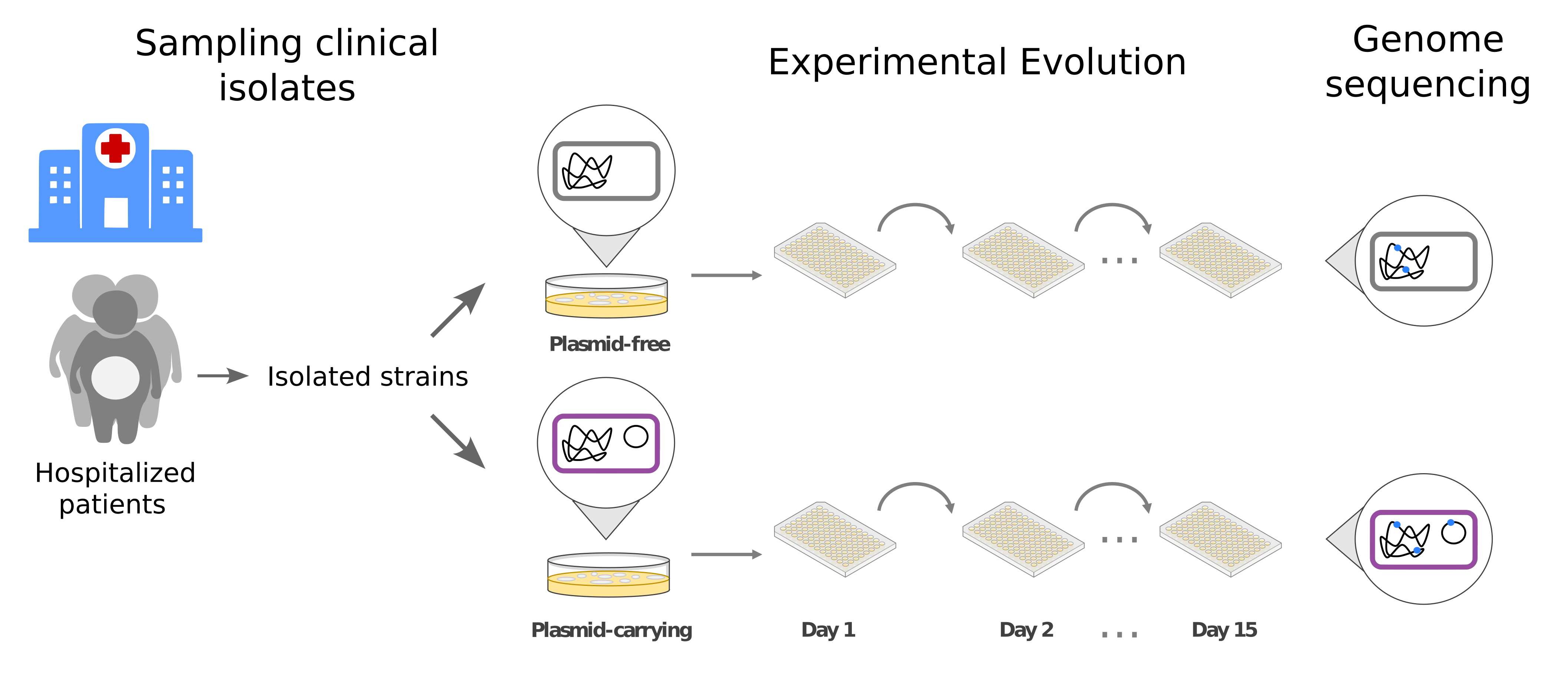
Interestingly, when we studied the genomes of the evolved bacteria, we found that the genetic targets altered were essentially the same both with and without the plasmid. However, the evolutionary pathways (i.e. the mechanisms through which bacteria evolved) differed. Plasmid-free bacteria mainly adapted through single substitutions (SNPs) in the genetic sequence of target genes, whereas plasmid-carrying bacteria adapted through insertion sequences (ISs) disrupting the same targets. ISs are the smallest genetic elements which can “jump” from one place of the genome to another one. If the IS happens to jump into a gene breaking it, it can be highly beneficial to the bacteria depending on the environmental conditions. Interestingly, the IS that moved during our experiment coincided with those carried by the AMR-plasmid under study.
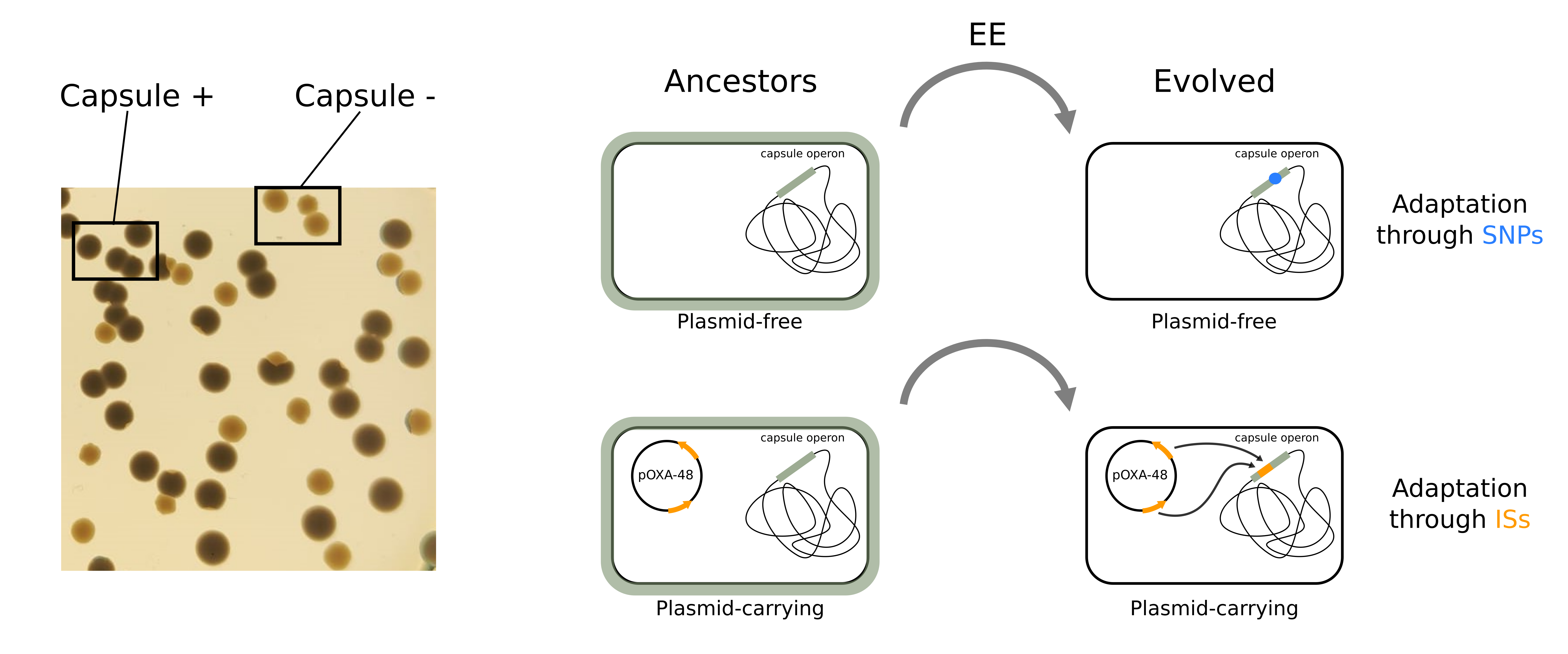
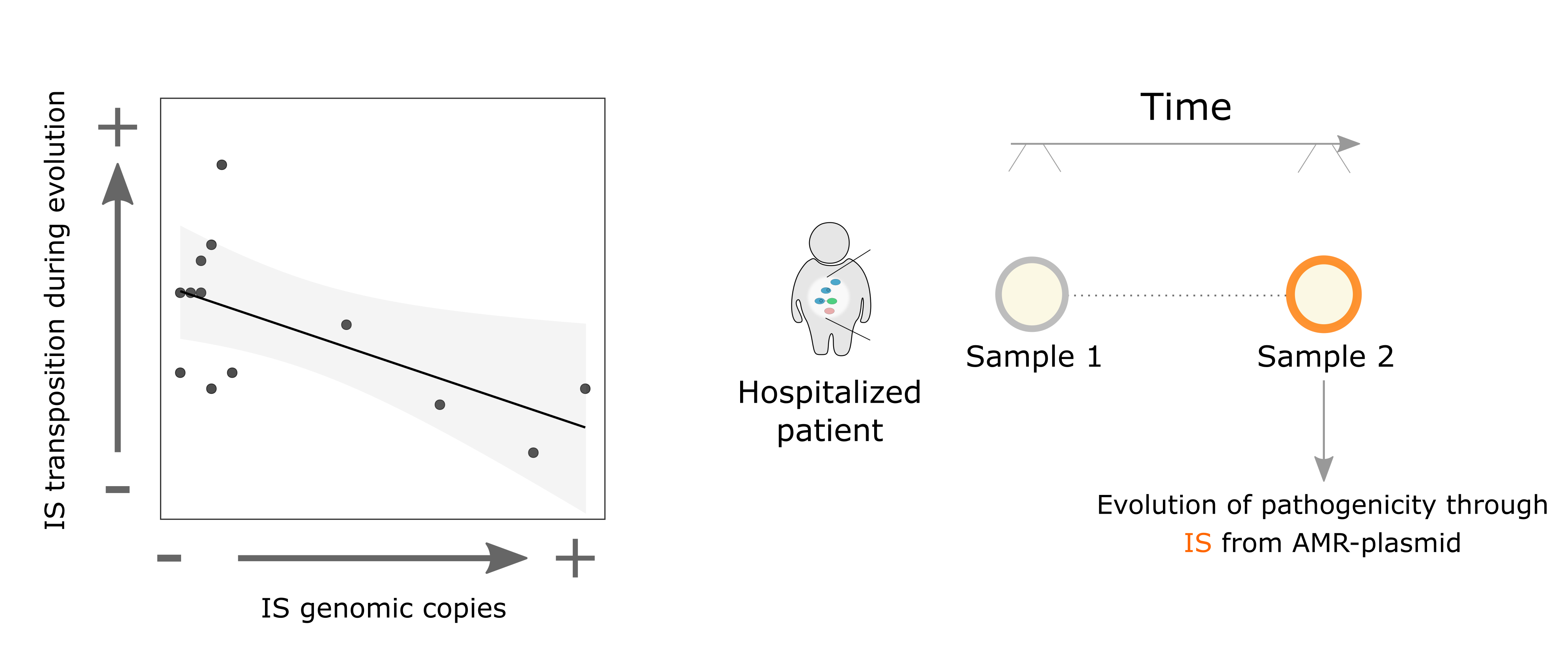
What amazed us was that this happened in most of the clones we analyzed, but not in others, so… what was making the difference? Taking a deeper look at some articles from the 90's studying the regulation of these IS elements, and correlating them with our data, we were able to decipher the mechanism underlying this puzzle: the lower the IS copies in a strain, the higher the transposition activity from the plasmid during evolution. Furthermore, we tried to translate our experimental results to real life scenarios. Luckily, thanks to previous work in the lab, we had access to the sequences of the bacterial lineages sampled over time from the same colonized patients, which allowed us to study their within-patient evolution. Analyzing them, we observed that these jumping genes could also potentiate the evolution of bacterial pathogenicity and virulence in the gut of hospitalized patients.
From a broader perspective, our results suggest that AMR-plasmids do not only confer resistance to their bacterial hosts, but also provide them with alternative strategies to evolve, specifically through the movement of transposable elements. Barbara McClintock, who discovered the transposable elements in corn during the 40's and 50’s, had to postpone the publication of her results due to the skepticism of the scientific community at that time. She opened the door to the study of a completely new genetic mechanism, and, fortunately, 30 years later she received the nobel prize for her discovery. Even nowadays with the use of the latest sequencing technologies, the study of jumping genes, which can move from plasmids to the chromosome and vice versa, and show multiple copies in the host, is an extremely challenging task. We hope to have done our bit with this work by shedding light into the role of transposable elements in bacterial evolution.
Follow the Topic
-
Nature Ecology & Evolution

This journal is interested in the full spectrum of ecological and evolutionary biology, encompassing approaches at the molecular, organismal, population, community and ecosystem levels, as well as relevant parts of the social sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Biodiversity and ecosystem functioning of global peatlands
Publishing Model: Hybrid
Deadline: Jul 27, 2026
Understanding species redistributions under global climate change
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in