Mapping dental stem-cell research: looking beyond publication counts
Published in Bioengineering & Biotechnology, Cell & Molecular Biology, and Biomedical Research
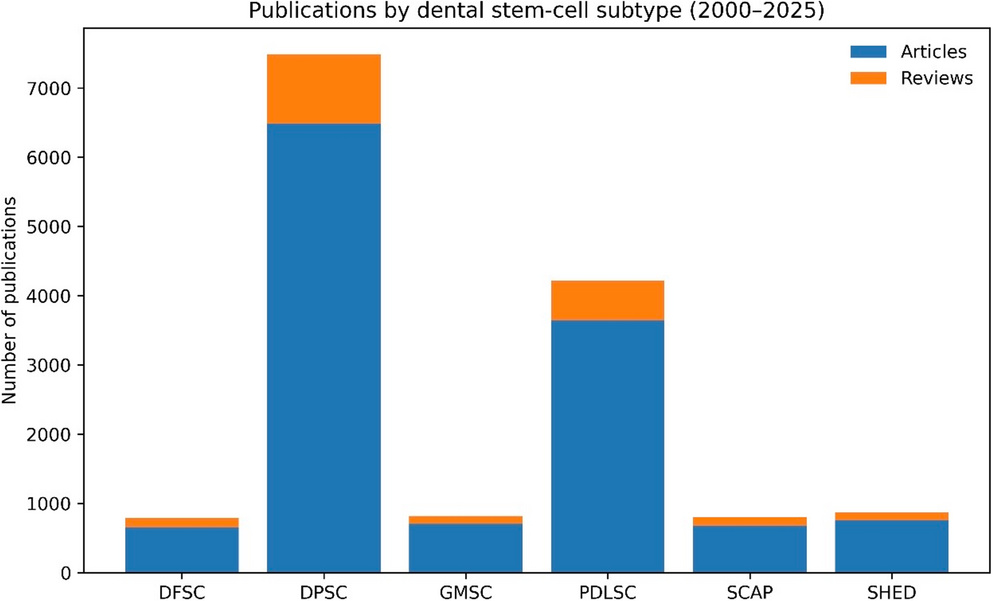
When I started working with dental stem cells and regenerative dentistry, I often noticed that some cell types appeared much more frequently in the literature than others. Dental pulp stem cells and periodontal ligament stem cells were widely represented, while other subtypes, such as SHED, SCAP, DFSC, and GMSC, seemed to follow different research trajectories.
This made me wonder: are the most frequently published dental stem-cell subtypes also the ones more often associated with translation-oriented language in the literature?
That question became the starting point for my recently published article in Oral and Maxillofacial Surgery. Instead of looking only at publication volume, I wanted to compare six dental stem-cell subtypes within the same analytical framework and examine how the literature has developed over 25 years.
As a single-author study, the process also required careful checking of terminology, overlap, and interpretation at each step.
The study used Web of Science-indexed records from 2000 to 2025 and focused on six major dental stem-cell subtypes: dental pulp stem cells, periodontal ligament stem cells, stem cells from human exfoliated deciduous teeth, stem cells from the apical papilla, dental follicle stem cells, and gingival mesenchymal stromal/stem cells.
One challenge was that these literatures overlap. A single paper may mention more than one dental stem-cell subtype, which can inflate simple publication counts. To reduce this issue, the study used a deduplicated corpus and an overlap-aware fractional counting approach for corpus-level comparisons.
I chose Web of Science Core Collection as the single database because the study depended on a consistent citation backbone for document co-citation and science-mapping analyses. Combining databases such as Web of Science and Scopus can broaden coverage, but it can also introduce differences in indexing, duplicate records, citation formats, and network structures. For this reason, I treated the Web of Science-only design as a methodological choice for internal consistency, while also recognizing it as a limitation rather than complete coverage of the field.
I also wanted to be careful with interpretation. The study did not assess therapeutic efficacy, clinical readiness, or regulatory maturity. Instead, it examined selected translation-oriented signals in the literature, including terms related to biomaterials/scaffolds, in vivo models, extracellular vesicles/exosomes, and clinical-trial-related language. These signals should be understood as indicators of research framing, not as evidence of clinical effectiveness.
Within these limits, the analysis suggested that publication volume and translation-oriented framing were not always aligned. Earlier-established subtypes continued to dominate total publication output, while some later-emerging subtypes showed proportionally higher representation of selected translation-oriented signals.
For me, the value of this work is not in ranking one subtype above another. Rather, it provides a structured map of how the field is being discussed and where evidence gaps may remain. I hope it can help researchers interested in regenerative dentistry, dental stem cells, biomaterials, extracellular vesicles, and translational tissue engineering think more carefully about subtype-specific research emphasis.
Published article:
Zahedah R. Bibliometric and science-mapping analysis of translation-oriented signals across six dental stem-cell subtypes: a Web of Science study (2000–2025). Oral and Maxillofacial Surgery. 2026;30:79.
Follow the Topic
-
Oral and Maxillofacial Surgery
This is a peer-reviewed online journal that caters to the needs of professionals in oral and maxillofacial surgery.
Ask the Editor - Oral and Maxillofacial Surgery
Got a question for the editor about Malocclusions or misalignment of the teeth? Ask it here!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Current State and Future Opportunities for 3D Printing/Additive Manufacturing in Oral and Maxillofacial Surgery: from Basic Science to Clinical Applications
Publishing Model: Hybrid
Deadline: Ongoing





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in