Metabolic restoration: a new indicator of treatment success beyond pathogen clearance
Published in Microbiology

Classical infectious disease drug development metrics focus on pathogen clearance. However, there are now numerous examples of diseases where pathogen killing is not automatically followed by symptom improvement. Examples include some manifestations of long COVID, post-Giardia irritable bowel syndrome, chronic Lyme disease, and many more. Chagas disease is caused by infection with Trypanosoma cruzi parasites. Parasite-induced and immune-derived tissue damage lead to progressive cardiac and gastrointestinal symptoms. Delaying antiparasitic treatment until late-stage symptomatic Chagas disease, for example because of limited diagnosis or treatment access, leads to parasite clearance but without preventing cardiac events or mortality 1. This has historically been attributed to persistent tissue damage, including irreversible cardiac fibrosis. However, given the strong role the McCall lab had found for metabolism in acute T. cruzi infection outcomes, we were curious about whether metabolism could also play a part in the clinical failure of benznidazole in late-stage Chagas disease.
To answer this question, in collaboration with partners at the National School of Tropical Medicine (NSTM) and Texas Children’s Hospital Center for Vaccine Development (TCH-CVD) at Baylor College of Medicine, in this paper we used a mouse model, in which mice were infected, treated with benznidazole, and then samples were collected 56 days later for metabolomic analysis 2. By collecting systematic sections across the heart, we were able to build a picture of metabolic changes post-treatment across heart regions. Specifically, we found that treatment with the antiparasitic benznidazole, followed by 56 days of recovery, only led to minor improvements in the cardiac small molecule profile at the bottom of the heart (Figure 1). Strikingly, Chagas disease patients can develop characteristic damage at the bottom of the heart, called apical aneurysms, so this lack of metabolic restoration at the bottom of the heart following benznidazole treatment matches with a major site of symptoms in patients. These results also matched with a parallel study that we performed using urine samples, where we found that urine metabolic changes also persist even after all parasites have been killed by benznidazole 3. Jointly, these findings provide a metabolic explanation for the clinical failure of benznidazole in advanced Chagas disease, expanding the known collection of responsible mechanisms and providing novel opportunities for intervention.
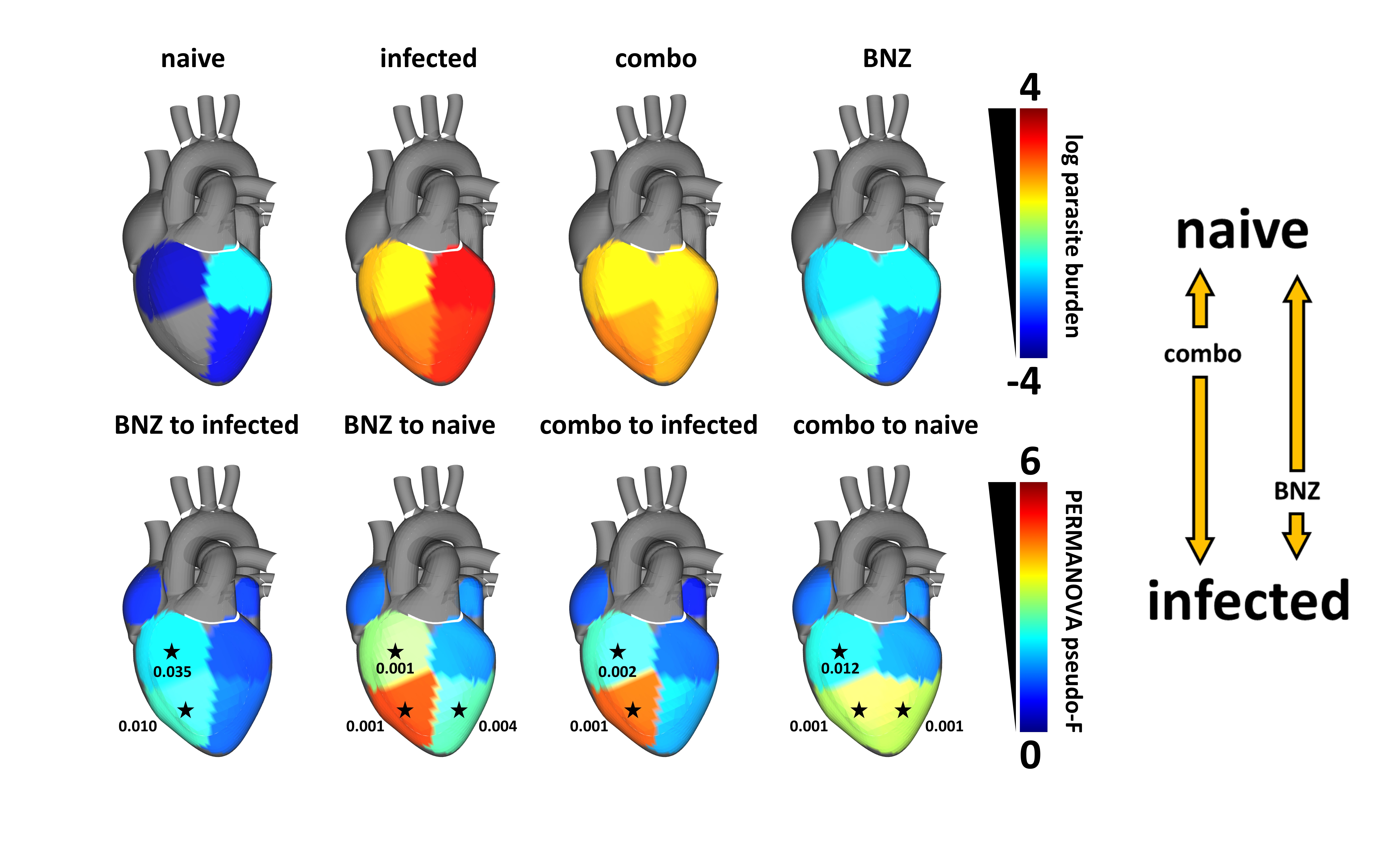
Figure 1. Superior metabolic restoration with vaccine-linked chemotherapy (“combo” treatment) than with benznidazole (BNZ), bottom and right-hand arrows, even though benznidazole is more effective at clearing parasite burden (top). Adapted from Liu et al, Nature Communications [ this paper], https://www.nature.com/articles/s41467-023-42247-w. 3D model purchased from 3DCADBrowser.com.
RNA-seq and flow cytometry analyses demonstrated that immune alterations also persisted post-treatment. Metabolism and immunity are tightly linked. Furthermore, Chagas disease progression has been tied to pathologic immune responses that aggravate cardiac damage. Our group at NSTM and TCH-CVD has been actively working to develop therapeutic vaccines for Chagas disease, to re-shape these immune responses to a more beneficial, balanced pattern 4,5. Combining this therapeutic vaccine with benznidazole, an approach called “vaccine-linked chemotherapy”, is designed to clear the T. cruzi parasites responsible for initiating and maintaining pathological immune responses, while at the same time re-balancing immunity. This treatment uses lower levels of benznidazole, so more parasites remain than with the standard, benznidazole-only treatment regimen. However, strikingly, metabolism was improved following vaccine-linked chemotherapy, becoming more similar to the patterns seen in uninfected animals. These findings demonstrate a dominant role for immunity in driving metabolic maladaptations in chronic Chagas disease. Indeed, most of the metabolite features impacted by chronic infection and correlated to disease severity metadata were not restored by treatment. This observation further reinforces a link between metabolism and Chagas disease pathogenesis. Several of these metabolic features refractory to treatment are correlated with immune system metrics. Treatment access and treatment delays are major challenges in Chagas disease. Encouragingly, we did not find a temporal pattern at the level of chemical classes with regards to ease of post-treatment metabolic restoration.
Importantly, these improvements in metabolism were observed at timepoints preceding cardiac functional improvement by the vaccine-linked chemotherapy 6, suggesting that a metabolism-centric approach may provide earlier readouts of functional treatment efficacy. Current assessment of Chagas disease treatment efficacy requires long-term patient follow-up, a burden for these patient populations that may not have easy access to healthcare, and increasing drug development costs. There is therefore a strong need in the Chagas disease drug development field for new and faster metrics of treatment success. While cardiac samples are unsuitable for routine assessment of treatment efficacy, our observation that this lack of metabolic restoration is also seen in urine samples 3 suggests that metabolomic analysis of urine samples could meet this need.
Driven by a desire to find new candidates for metabolism-restoring therapies, we also quantified the proportion of infection-impacted chemical classes that were not restored by treatments. Lipids and related molecules were more easily restored than nucleotide-related molecules, suggesting that treatments targeting purine metabolism should be further studied with regards to effects on infection-induced metabolic changes and functional improvement, looking beyond effects on parasite burden.
Overall, these results suggest a need to move beyond traditional drug development metrics, into a comprehensive, functional assessment of treatment efficacy that involves quantification of post-treatment metabolic restoration. Research going forward will need to assess whether this paradigm holds true beyond Chagas disease.
Read the paper here: https://www.nature.com/articles/s41467-023-42247-w
References:
- Morillo, C. A. et al. Randomized trial of benznidazole for chronic chagas’ cardiomyopathy. N. Engl. J. Med. 373, 1295–1306 (2015).
- Liu, Z. et al. Localized cardiac small molecule trajectories and persistent chemical sequelae in experimental Chagas disease. Nat Commun. 14, 6769 (2023). [this paper]
- Dean, D.A. et al. Persistent Biofluid Small-Molecule Alterations Induced by Infection Are Not Restored by Parasite Elimination. ACS Infect Dis, in press (2023).
- Dumonteil, E. et al. Accelerating the development of a therapeutic vaccine for human Chagas disease: rationale and prospects. Expert Rev. Vaccines.11,1043-55 (2012).
- Jones, K. M. et al. Preclinical advances and the immunophysiology of a new therapeutic Chagas disease vaccine. Expert Rev. Vaccines. 21, 1185-1203 (2022).
- Jones, K. M. et al. Vaccine-linked chemotherapy improves cardiac structure and function in a mouse model of chronic Chagas disease. Front. Cell. Infect. Microbiol. 13, 1106315 (2023).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in