Metabolic rewiring of the probiotic bacterium Lacticaseibacillus rhamnosus GG contributes to cell-wall remodeling and antimicrobial production
Published in Microbiology
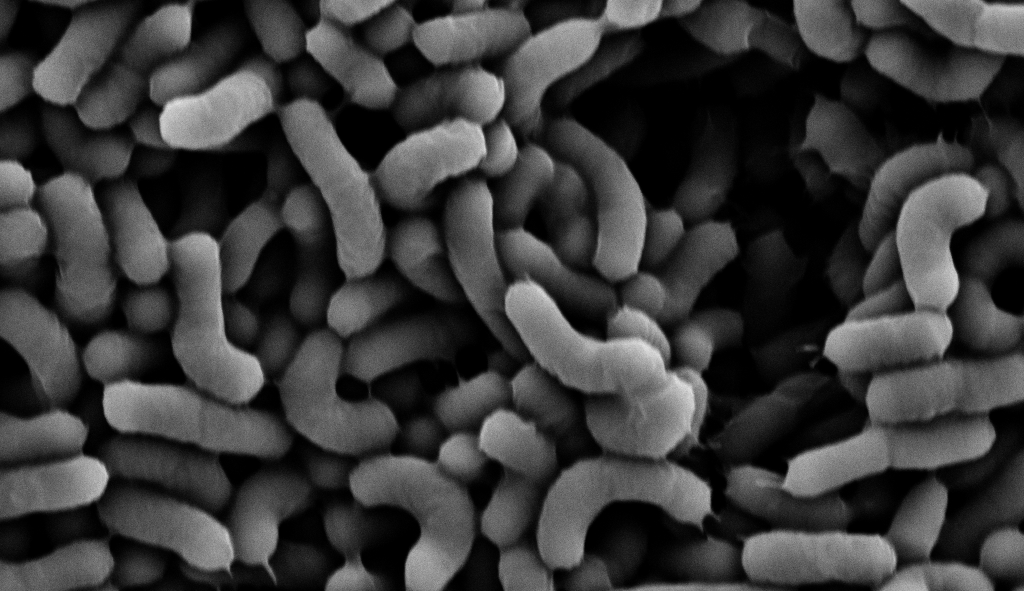
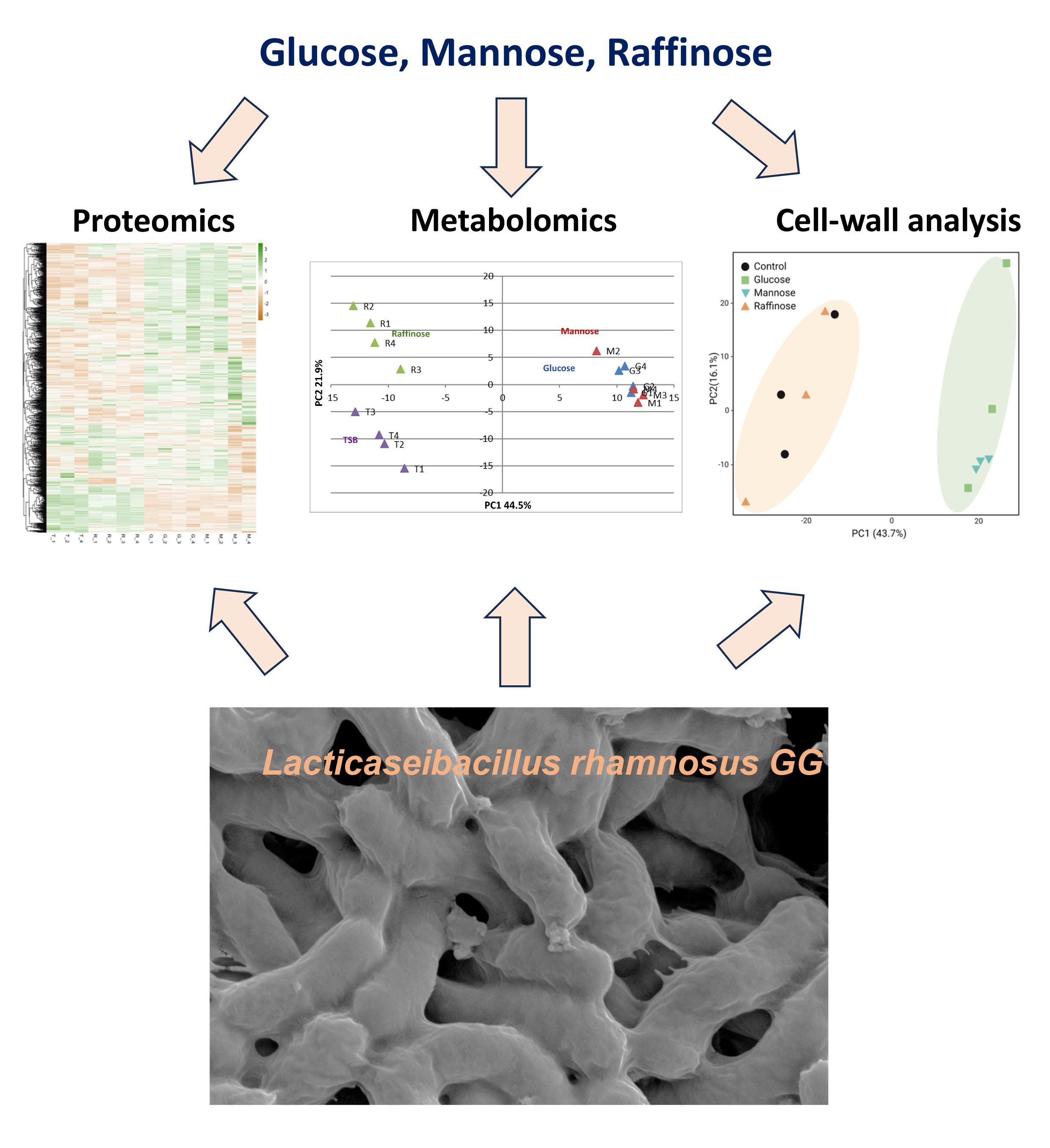
Lactobacillaceae belong to the lactic acid bacteria (LAB) group, as lactic acid is their main end-product of carbohydrate metabolism. These bacteria are Gram-positive rods. They are adapted to the low oxygen environments of humans and mammalians' gastrointestinal tract (GIT) and are facultative anaerobes. The probiotic performance in mammals' gut depends on nutrient composition and availability. Therefore, the performances of Lactobacillaceae and additional probiotic bacteria must be systematically explored and related to core metabolism. In our manuscript, we utilized metabolomic, proteomic, and volatilome (airborne metabolites) analysis (Figure 1) and in-depth microbial cell wall analysis to correlate simple carbohydrates' effect on a model probiotic bacterium Lacticaseibacillus rhamnosus GG (LGG) https://www.nature.com/articles/s41522-023-00431-2.
We found the availability of fermentable sugars, e.g., glucose and mannose, is imperative to physiological adaptations associated with increased fitness and competitiveness within a host: The accumulation of cytotoxic metabolites (Ethanol, organic acids, and butanol), volatiles, and antibacterial extracellular proteins.
Furthermore, while LGG eliminates a broad spectrum of pathogenic bacteria in a fermentation-dependent manner, LGG itself alters its cell wall and biofilm formation as a potential coping mechanism against the same conditions. In addition, our results indicated that cell wall remodeling precedes biofilm formation and that it is very likely that metabolic rewiring and the accumulation of toxic fermentation products trigger cell wall remodeling. Collectively, our data provide strong evidence that cell wall remodeling precedes an induced biofilm formation, and these behaviors are correlated with enhanced antimicrobial production. We also found that an improved biofilm formation occurs downstream of fermentation and may involve the upregulation of proteins associated with an increased adhesion.
Overall, we provide compelling evidence linking specific dietary changes with the performance of probiotic strains. Our results may yield improved interventions based on probiotic bacteria or their products.
For more details, read our work here: https://www.nature.com/articles/s41522-023-00431-2.
Follow the Topic
-
npj Biofilms and Microbiomes
The aim of this journal is to serve as a comprehensive platform to promote biofilms and microbiomes research across a wide spectrum of scientific disciplines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Microbial endocrinology
Publishing Model: Open Access
Deadline: Oct 21, 2026
Microbiome and energy metabolism
Publishing Model: Hybrid
Deadline: Dec 06, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in