Neutron diffraction provides molecular insight into carbon capture solutions
Published in Chemistry, Physics, and Sustainability
Our planet is undergoing a climate crisis, with CO2 emissions from industrial processes and energy production being one of the leading causes of climate change. Scientists worldwide are pioneering innovative solutions to mitigate climate change and secure a sustainable future. Carbon capture technologies are part of this revolution.
This process involves removing CO2 from industrial CO2-rich flue gases by using chemical agents that selectively bind to carbon dioxide, allowing the remaining purified air to be safely released into the atmosphere. The captured CO2 can later be released from the solvent via heat for geological storage or utilised. As renewable energy alone cannot meet current demand and vital industries, like cement and steel, remain difficult to decarbonise, carbon capture technologies are essential for a sustainable future. An ideal carbon capture solvent maximises CO2 absorption rates and capacities, while minimising the energy required for regeneration. This requires an understanding of the structure of reaction complexes in unloaded and carbon dioxide loaded carbon capture solutions. We were motivated to address this challenge by applying the technique of neutron diffraction and structural refinement modelling. These experiments are performed at the ISIS Neutron and Muon Source, a world leading research centre in the UK that is funded by the Science and Technology Facilities Council (STFC). ISIS supports over 3000 scientists from both academic and industrial settings every year, allowing scientists to probe matter at the atomic scale in ways no other tools can, driving breakthroughs in clean energy, healthcare, engineering, advanced technologies and more. At ISIS, high-energy protons from a particle accelerator are collided with a metal target to eject neutrons through the process of spallation. The neutrons are directed at samples to reveal precise, non-destructive, atomic-level insights. This can provide access to structural and dynamic information for a diverse range of samples which is simply not possible in traditional laboratory settings. When combined with structural refinement modelling, the neutron diffraction approach reveals a clear picture of the atomic positions present in the carbon capture solutions.
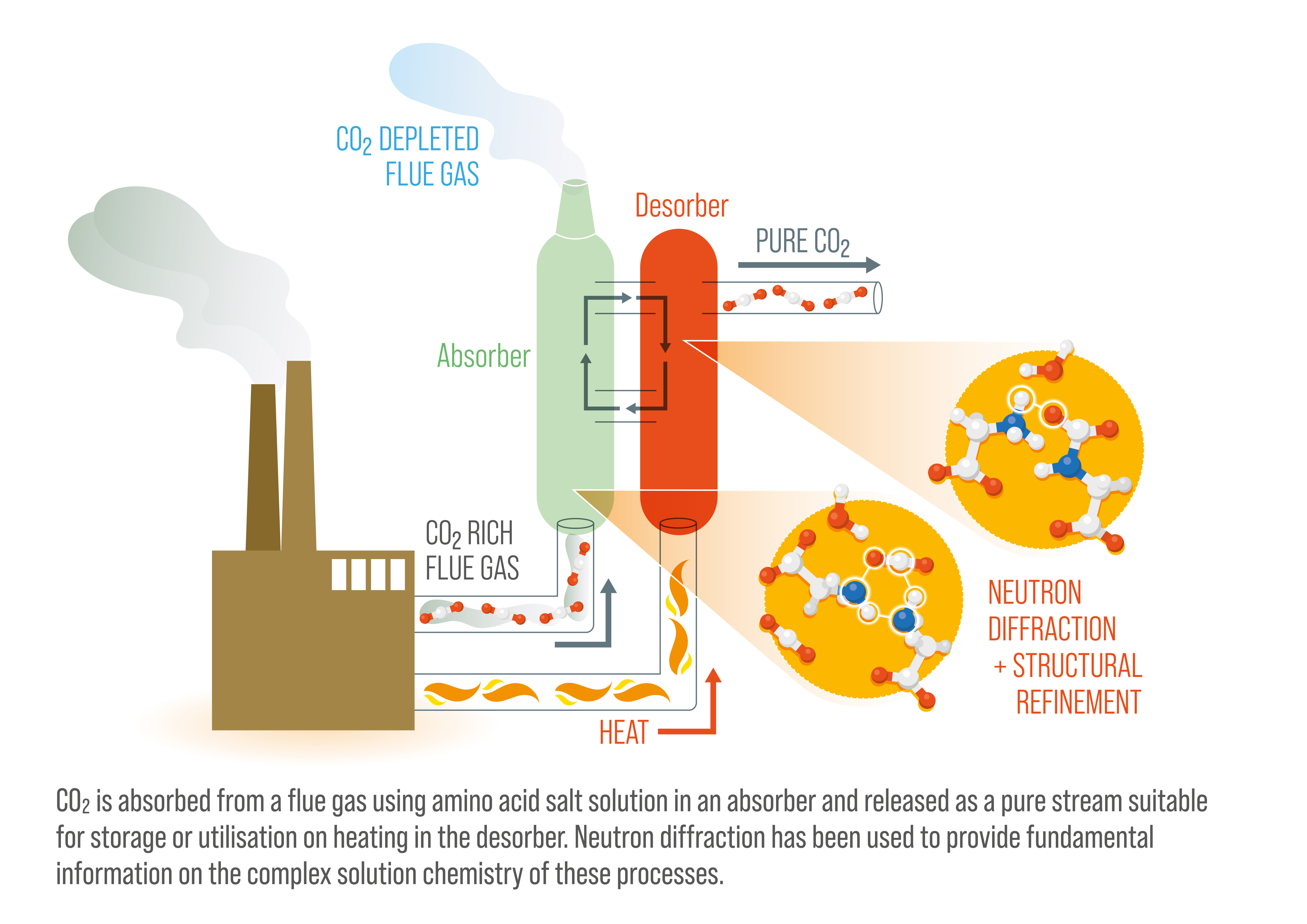
We studied two carbon capture solutions pre and post CO2 capture that are currently of wide interest due to their efficient capture, low energy regeneration requirements, and biocompatibility. Amino acids are fundamental building blocks that comprise all proteins in all living organisms, and when combined with alkaline salts, form amino acid carboxylate salts. This combination alters the chemical structure of the amino acids, rendering them capable of binding CO2 molecules. Two different amino acid salt systems were made by dissolving the simplest of the 20 naturally occurring amino acids, glycine, with either sodium or potassium hydroxide in water. Preparation of these salts and determination of their CO2 uptake rates and reaction products were done in collaboration with Dr Daniel Sault and Prof Christopher Rayner from University of Leeds School of Chemistry and senior chemist James Wheatley from University of Leeds spinout company C-Capture Ltd. who are all leading scientists in the area of carbon capture chemistry.
We performed two experimental studies at the ISIS facility using the NIMROD and SANDALS instruments to examine carbon capture solutions in the unloaded and CO2 loaded state respectively. Both experiments lasted for 4 days, with samples running 24 hours a day, and were supported by a dedicated team of specialists, including Disordered Materials group instrument scientists Tom Headen and Terri-Louise Hughes.
Using the combined neutron diffraction and computational modelling approach gave us atomic level insight into these complex systems, allowing us to probe the occurrence of interactions between molecules required for the various chemical routes which enable CO2 loading and offloading. Briefly, a single hydrogen atom must be lost at the absorbing site on an amino acid molecule, such that it can be replaced by a CO2 molecule. The hydrogen atom must then navigate its way through the solution to find a second amino acid molecule, which will accept the hydrogen. The total process of CO2 capture in these solutions therefore requires two amino acid molecules to absorb a molecule of CO2. The reaction occurs in the reverse direction for the liberation of CO2 to enable the solvent to then be reused in further capture cycles.
Through this technique we chose to focus on a handful of interaction structures, investigating the immediate chemical environment around the CO2 absorbing group of the amino acid in the unloaded solutions, and around the newly absorbed CO2 molecule on the amino acid in the CO2 loaded solutions. Study of these areas are crucial for accurate insight into the CO2 loading and offloading rates in these solvents. We also played careful attention to the structure of the water present in these systems, as it is this aqueous network that CO2 absorbing amino acids, CO2 molecules, and single hydrogen atoms must navigate for these chemical reactions to occur.
Our study revealed that the key interaction governing CO2 loading in amino acid solutions is the hydration shell surrounding the amino acid’s active site. This is exciting because it suggests that taking steps to destabilise this interaction through careful design of the chemical sites adjacent to the carbon absorbing group when designing new carbon capture solvents could enhance their CO2 absorbing ability. We also showed that the association of potassium around the CO2 absorbing site on the amino acid molecule is considerably less stable than the equivalent sodium interaction around the CO2 absorbing site. This indicates a lower barrier for incoming CO2 molecules, providing a fundamental explanation for why CO2 absorbs more rapidly in potassium-based amino acid salts that in sodium-based alternative, confirming both our observation and previous research. Finally, we observed that the key interactions to be considered for CO2 offloading are the associations both water and the potassium/sodium around the loaded CO2 molecule on the amino acid. This provides a route towards designing new carbon capture solvents which can exploit these interactions.
Our study highlights neutron diffraction as a universal, high-resolution toolkit for studying CO2 loaded solutions, demonstrating its potential to advance the investigation of any carbon capture solvent system. In the future, neutron diffraction may support the design of the next generation of carbon capture solvents, supporting our journey towards a more sustainable future.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
What are SDG Topics?
An introduction to Sustainable Development Goals (SDGs) Topics and their role in highlighting sustainable development research.
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in