Promising Breakthrough Treatment Offers New Hope for Spinal Cord Injury
Published in Biomedical Research
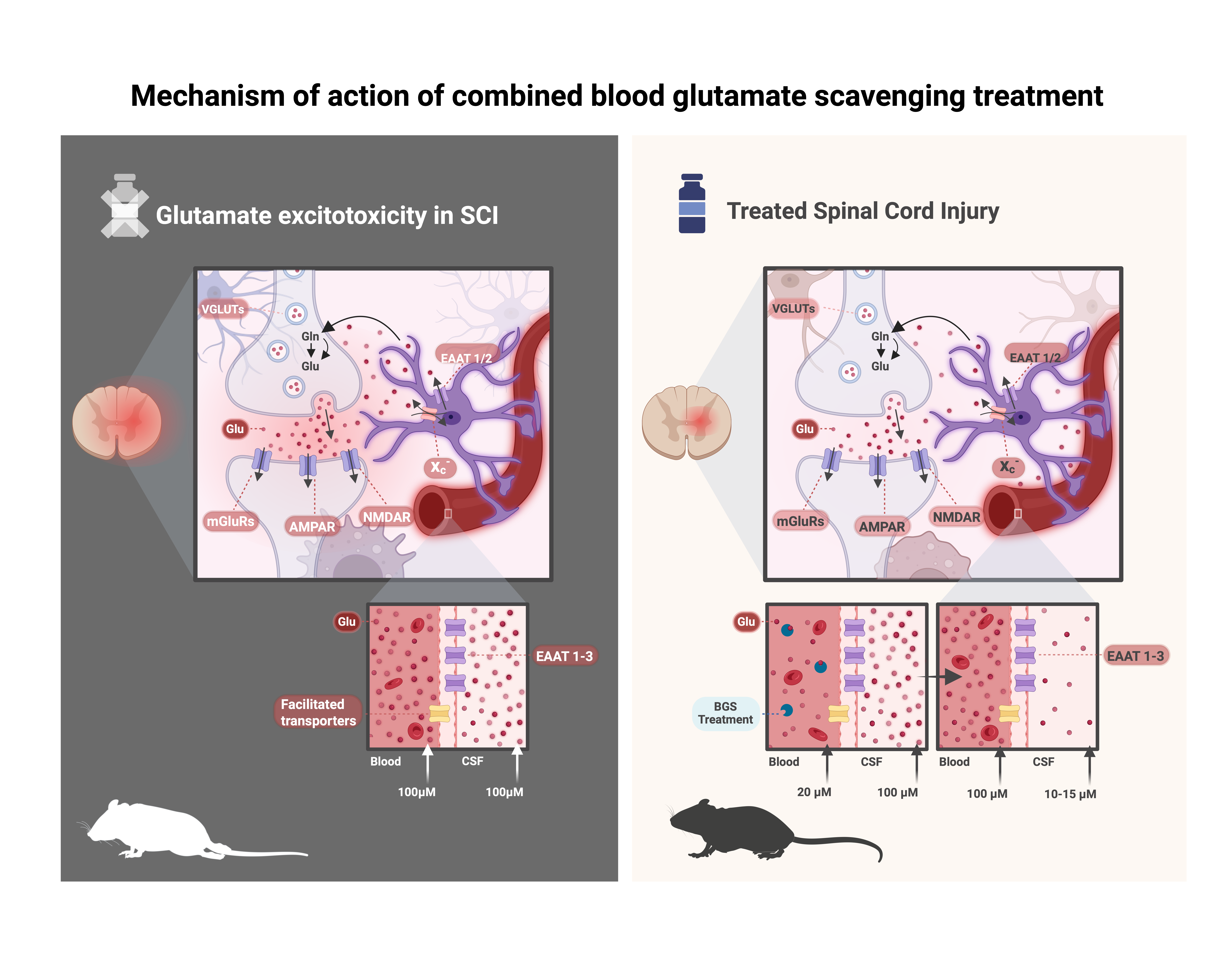
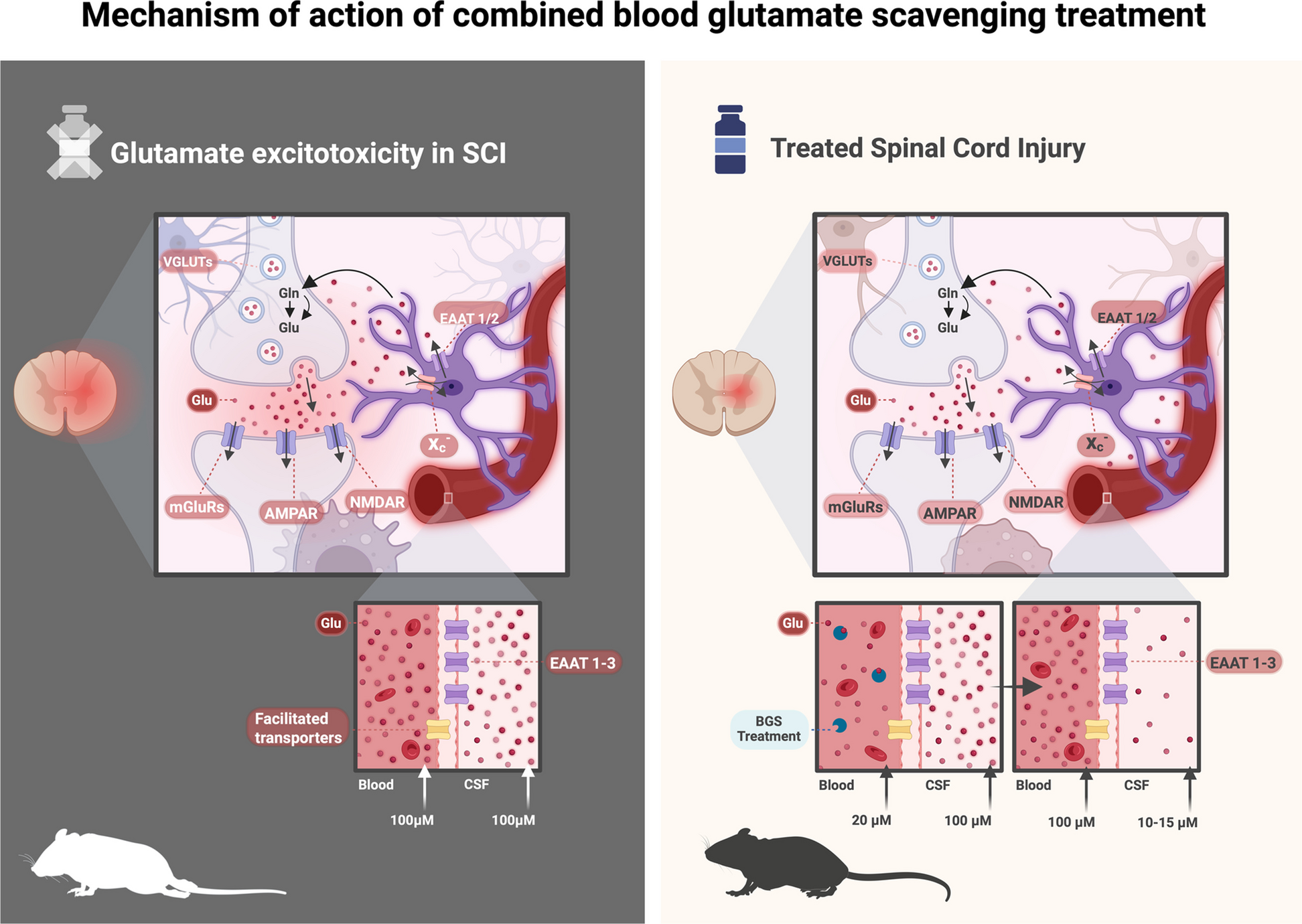
The research, led by Dr. Angela Ruban at Tel Aviv University, together with Dr. Yona Goldshmit, M.Sc. Josef Levine, M.Sc. Rosemary Lavender and Alexander Yakovchuk developed and validated a treatment platform that lowers toxic blood glutamate levels and helps remove excess glutamate from the cerebrospinal fluid. The results of the new treatment were striking: treated animals showed substantially less tissue damage, reduced inflammation and scarring, better preservation of nerve cells and axons, and up to 80% improvement in locomotor recovery compared with untreated animals.
Importantly, the treatment remained effective even when administered up to eight hours after injury, a time window that could make it practical for real-world emergency medicine. The findings also showed a strong safety profile, a critical step toward clinical translation.
The results were further strengthened by independent validation from an external contract research organization (CRO) in a severe rat model of spinal cord injury.
These findings are now moving beyond the laboratory. NeuroHagana, a biotechnology company founded by Dr. Ruban, has validated the results and is now advancing development of the treatment toward clinical use. The company is focused on transforming this academic breakthrough into a real emergency therapy for patients, with potential applications not only in spinal cord injury but also in other acute neurological conditions driven by glutamate toxicity, including traumatic brain injury and stroke.
Follow the Topic
-
Inflammation and Regeneration
Inflammation and regenerative medicine are interconnected and vital in translational research. Inflammation and Regeneration invites manuscripts that bridge these fields, offering an open-access platform covering a broad spectrum of basic and clinical regenerative medicine topics.
Related Collections
With Collections, you can get published faster and increase your visibility.
Regeneration of Immune Cells: Toward Regenerative Immunotherapy
Human induced pluripotent stem cells (iPSCs) have created new opportunities to deliver immune-cell therapies to patients who currently lack effective treatment options. iPSC platforms can provide a renewable, standardized supply of therapeutic immune cells with robust function, supporting indications that range from cancer and chronic viral infection to autoimmunity and transplantation-related complications. In this Thematic Series, four experts review the biology and translation of iPSC-derived iNKT cells for head and neck cancer, antigen-presenting cell products for cancer translational research, CD8+ T cells for cancer and HIV, and CD4+ regulatory T cells for autoimmunity and transplant immunology. Together, these articles highlight engineering and manufacturing challenges, safety and efficacy considerations, and emerging directions toward scalable, next-generation immunotherapies.
Please note that this Collection is invite-only and will not accept unsolicited submissions.
All submissions in this collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Oct 09, 2026
Decoding the Multilayered Nature of Inflammaging: Molecular Mechanisms, Organ Crosstalk, and Environmental Influences
Inflammaging, defined as low-grade chronic inflammation that accompanies aging, gradually disrupts systemic homeostasis and underlies the onset of multiple age-related disorders. Recent advances have expanded this concept beyond cellular and molecular mechanisms, revealing complex interactions that encompass immune modulation, metabolic reprogramming, organ crosstalk, the gut environment, and even social and environmental determinants. This Collection aims to explore inflammaging from an integrative and multilayered perspective. We welcome studies that investigate the molecular basis of cellular and immune aging, systemic inflammatory networks, their impact on regeneration and tissue repair, and the interplay with environmental and lifestyle factors. Original research, reviews, and conceptual or perspective articles that advance the understanding of inflammaging across biological scales are all encouraged.
All submissions in this collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Oct 09, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in