Regenerate, repair and replace: advanced therapy clinical trials
Published in Bioengineering & Biotechnology, Biomedical Research, and Pharmacy & Pharmacology

Advanced therapy clinical trials focus on cutting-edge treatments known as advanced therapy medicinal products (ATMPs). ATMPs can be classified into three main types:
- Gene therapy involves transferring genetic material into cells to treat, prevent, or cure diseases.
- Somatic cell therapy involves administering cells or tissues that have been manipulated to change their characteristics.
- Tissue-engineered products contain cells or tissues that have been modified so that they can be used to repair, regenerate, or replace human tissue.
In this blog we take a look at a selection of the ATMP studies registered at ISRCTN: the UK’s Clinical Study Registry that are recruiting at the time of writing.
The Bardet-Biedl syndrome 1 (BBS1) mutation affects multiple organs, including the eyes and vision. The AXIS study (ISRCTN96250868) is assessing whether AXV-101 gene therapy injected into the eyes of participants with mutated BBS1 genes can prevent sight loss.
The FORTRESS study (ISRCTN12595783) is testing whether NG-350A, an experimental gene therapy derived from a virus, can kill cancer cells (rectal cancer specifically in this study) without affecting normal cells.
Neonatal onset OTC deficiency is a rare genetic condition that causes ammonia to build up in the blood because the enzyme OTC is missing. A new investigational gene-editing drug, ECUR-506, introduces a corrective gene which can produce a working OTC enzyme (ISRCTN10957794).
A long-term study at Great Ormond Street Hospital (ISRCTN91900773) is following up patients with inherited immune system conditions who were treated with gene therapy to correct the faulty gene in their blood stem cells. The study will monitor any long-term side effects and assess how well the treatment works over time.
Mucopolysaccharidosis Type II is an inherited disease where a gene that codes for an enzyme is missing, which causes waste products to build up in cells and results in delayed development, deteriorating mental status and behavioural problems. A study at the University of Manchester (ISRCTN12458940) aims to deliver the missing enzyme to the patients’ cells by collecting and genetically manipulating the patients' own stem cells with gene therapy to replace the missing gene before infusing them back into the body.
A coronary artery bypass graft (CABG) operation involves harvesting a patient’s veins to create grafts which are used to bypass blockages in the blood vessels of the heart. The PROTECT study (ISRCTN43650325) is investigating the safety of a new gene therapy that is administered to the vein grafts to prevent them from deteriorating and failing over time.
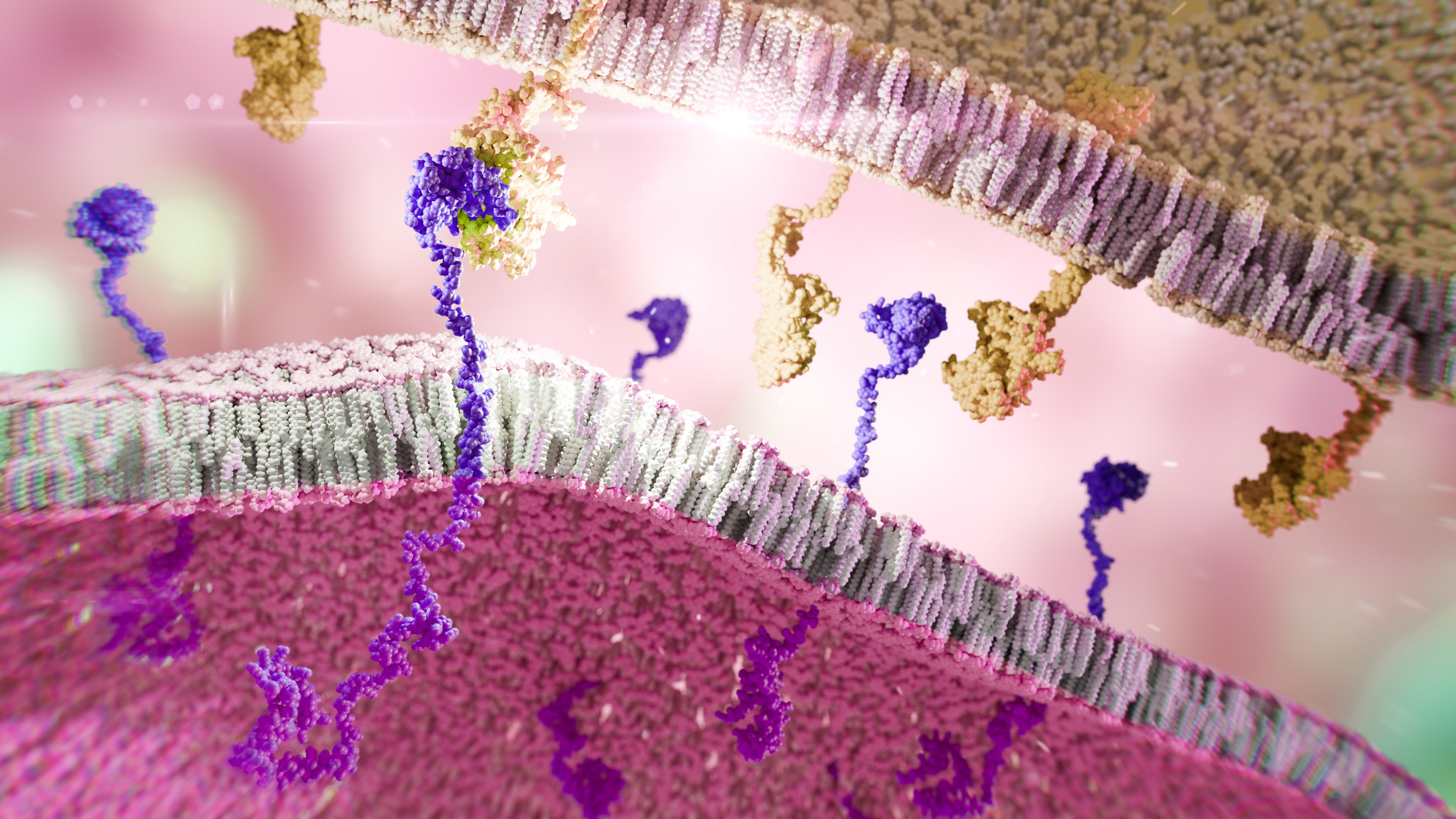
T cells are part of the immune system and help fight infections. In CAR-T therapy, the patient’s own T cells are modified in the lab so that they can recognise and kill cancer cells. The reprogrammed cells are called CAR-T cells (short for Chimeric Antigen Receptor T cells).
Different varieties of CAR T cells are currently being tested in multiple Phase I studies, including blood cancers such as T-cell acute lymphoblastic leukaemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL) (FRACTALL, ISRCTN15341827) and B-cell cancers (ISRCTN17901467), in sarcoma (MIGHTY, ISRCTN75533638), and in brain tumours such as glioblastoma (CARGO, ISRCTN22366199) and glioma (GLIMPS, ISRCTN15356845). The long-term outcomes and adverse effects are being monitored in the LTalloCAR study (ISRCTN15432015), which follows up on leukaemia patients previously treated in Phase I trials.
CAR-T cells are also being modified to treat non-cancer conditions. Amyloidosis is a group of conditions where an abnormal protein, called amyloid, accumulates in the tissues. The ALARIC study (ISRCTN49320109) is testing whether CAR-T cells can be used to target and kill the cells producing the amyloid.
Other immune cell based therapies being studied include regulatory T cells for protecting transplanted hearts in children (ISRCTN15374803) and macrophages to promote liver regeneration after paracetamol overdose (ISRCTN12637839).
Stem cells are also categorised as ATMPs when these cells undergo substantial manipulation or are used for a different function. In Taiwan, umbilical cord-derived mesenchymal stem cells are being used to treat acute respiratory distress syndrome (ARDS), a life-threatening lung injury that allows fluid to leak into the lungs (ISRCTN47911040), and also ARDS and/or severe septic shock (ISRCTN52319075), along with circulating hematopoietic stem/progenitor cells for patients with heart disease (ISRCTN14054375).
Tissue-engineered products consist of cells or tissues engineered to regenerate, repair or replace a human tissue. In the AMELIE study (ISRCTN10591726) the patient’s muscle cells are combined with small porous carrier particles called microcarriers to make a product to treat obstetric anal sphincter injury.
In the HELP study (ISRCTN14716227) hepatocytes are being encapsulated with mesenchymal stromal cells in alginate microbeads for the treatment of acute liver failure in children.
About 12% of all ongoing global ATMP trials are in the UK, and there is significant investment to support ATMP development (source). ATMPs have the potential to revolutionise the way diseases are treated and could transform the lives of patients.
Follow the Topic
-
ISRCTN: The UK’s Clinical Study Registry
A primary clinical trial registry recognised by WHO and ICMJE that accepts studies involving human subjects or populations with outcome measures assessing effects on human health and well-being, including studies in healthcare, social care, education, workplace safety and economic development.

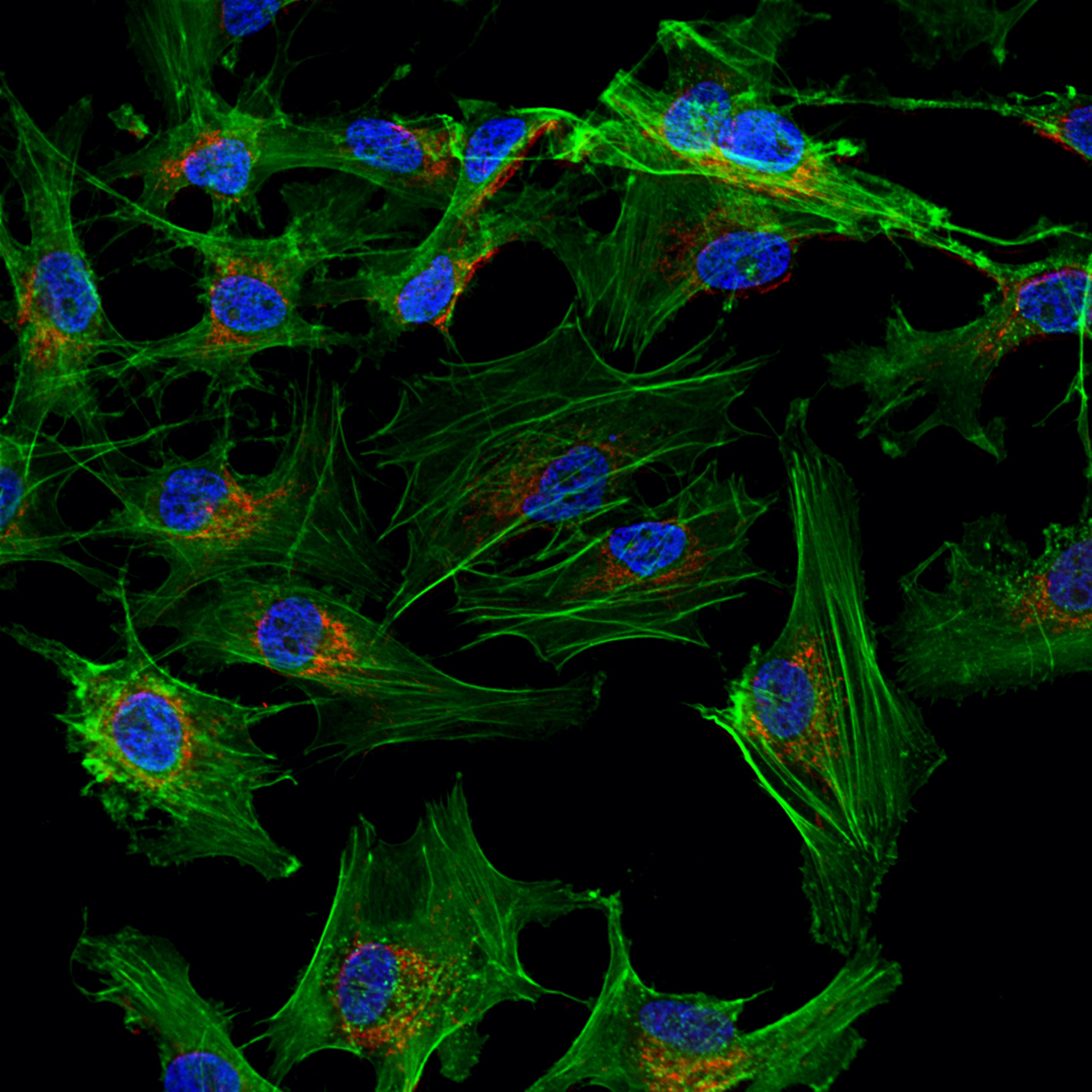 Stem cells are also categorised as ATMPs when these cells undergo substantial manipulation or are used for a different function. In Taiwan, umbilical cord-derived mesenchymal stem cells are being used to treat acute respiratory distress syndrome (ARDS), a life-threatening lung injury that allows fluid to leak into the lungs (
Stem cells are also categorised as ATMPs when these cells undergo substantial manipulation or are used for a different function. In Taiwan, umbilical cord-derived mesenchymal stem cells are being used to treat acute respiratory distress syndrome (ARDS), a life-threatening lung injury that allows fluid to leak into the lungs (



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in