Seeing the hidden architecture of environmental interfaces by bioCP-SEM
Published in Earth & Environment
The rhizosphere is often described as one of the most dynamic interfaces on Earth, but observing this interface without disrupting its spatial organization remains challenging. Roots, microorganisms, organic matter and mineral particles form a highly heterogeneous structure that is easily disturbed during conventional preparation. Roots can be observed separately, microorganisms can be cultured or detected by molecular methods, and soil particles can be analyzed as mineral components. However, observing these components together, while preserving their spatial relationships and ultrastructural details, remains technically difficult.
Our study began with a simple but long-standing question: can electron microscopy contribute more directly to environmental resource science by visualizing the hidden structures of complex biological and mineral interfaces?
For more than 20 years, I have worked mainly on electron microscopy of plant samples. During this time, I have been involved in many aspects of biological electron microscopy, including chemical fixation, resin embedding, ultrathin sectioning, transmission electron microscopy, scanning electron microscopy and correlative imaging. These approaches are powerful for observing plant cells and tissues at high resolution. At the same time, I often wondered whether techniques developed for biological electron microscopy could be extended beyond conventional laboratory specimens and used to observe more heterogeneous environmental samples.
This question became more important to me during the COVID-19 pandemic. I work at the RIKEN Center for Sustainable Resource Science, and for many years I had been asking myself how electron microscopy could contribute more directly to environmental resource science. While taking walks outside during the pandemic and reflecting on this question, I began to imagine the microorganisms hidden in natural environments: in soil, around plant roots, and on the surfaces of stones. I thought it would be fascinating if we could observe their fine structures not as isolated organisms, but together with the surrounding biological and mineral components. I also wondered whether electron microscopy could show how microorganisms, plant tissues, organic matter and mineral particles coexist within environmental interfaces.
That idea became the starting point of this study. We considered whether methods from biological and materials electron microscopy could be combined. From biological electron microscopy, we could use fixation, dehydration and resin embedding to preserve tissues, cells and microorganisms. From materials science, we could apply the idea of cutting and polishing hard, mineral-containing specimens to expose a flat cross section. If these approaches could be combined, perhaps even soil containing roots, microbes and minerals could be observed by scanning electron microscopy.
In principle, the idea seemed straightforward. In practice, it required extensive trial and error. Environmental samples are not uniform. They contain soft biological tissues, fragile microbial structures, organic matter, air spaces, water, mineral particles and hard inclusions. Each component responds differently to fixation, dehydration, resin infiltration, cutting and polishing. A method that preserves plant cells may not preserve soil structure. A method that produces a flat polished surface may damage fragile biological ultrastructure. We therefore had to design the workflow carefully, step by step.
Throughout the development of the method, our first priority was to preserve the ultrastructure of tissues, cells and microorganisms as much as possible. We did not want to obtain only a mechanically polished surface; we wanted a polished surface that still retained biological information. This required careful consideration of fixation conditions, resin infiltration, block preparation, sectioning and polishing. We repeatedly tested how to introduce fixatives into fragile root–soil samples, how to infiltrate resin into heterogeneous matrices, which instruments could cut embedded blocks without causing excessive damage, and how to polish the exposed surface finely enough for backscattered electron imaging.
Backscattered electron imaging by scanning electron microscopy was an important part of the workflow. Polished resin-embedded sections can provide broad views of complex samples, while also revealing fine structural contrast within biological specimens. The challenge was not simply to see something under the microscope, but to retain enough ultrastructural detail to interpret biological structures within a much larger environmental context.
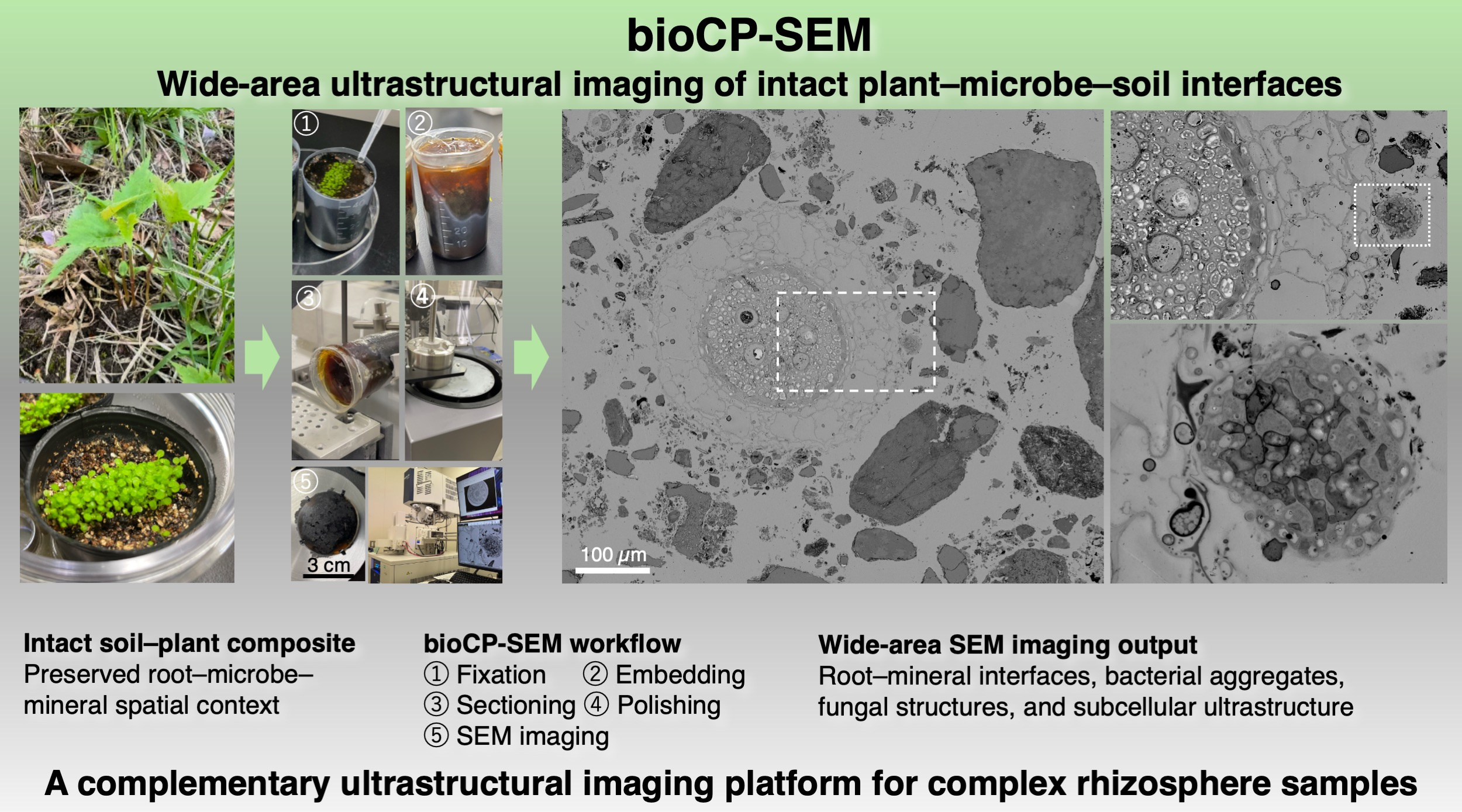
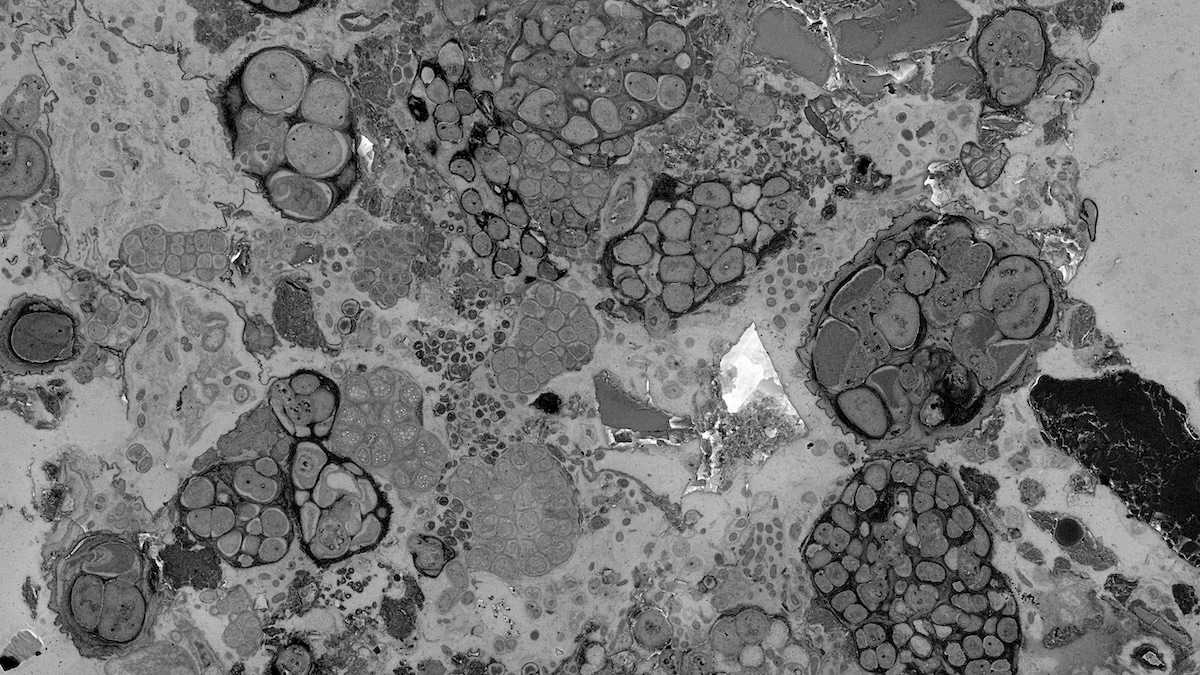
One of the most exciting moments came when we first observed structures resembling microorganisms in polished sections of root–soil samples. Later, when plant cellular structures also became visible in the same type of preparation, we felt that the approach was beginning to work. These observations were encouraging because they suggested that biological ultrastructure could be preserved within a sample that also contained mineral particles and other environmental components. For us, this was not simply a technical achievement. It was the moment when a long-standing question started to become a practical imaging method.
This work eventually developed into biological cross-sectional polishing scanning electron microscopy, or bioCP-SEM. The term “biological” is important because the method is not merely a polishing technique. It is a workflow that begins with the preservation of biological structures and then combines resin embedding, cutting, polishing and scanning electron microscopy to visualize complex biological–mineral interfaces. The aim is to observe the spatial organization of soft and hard components while retaining ultrastructural information.
bioCP-SEM does not by itself identify microbial species or reveal chemical function. It is not intended to replace molecular, isotopic or chemical analyses. Rather, it provides a structural map of complex samples. By showing where roots, microorganisms, organic matter and mineral particles are located in relation to one another, bioCP-SEM can help researchers understand the physical organization of environmental interfaces and select regions of interest for future correlative analyses.
Although this study focused on plant–microbe–soil interfaces, we believe that the concept has broader potential. Many biological samples from natural environments contain both soft tissues and hard components. These include organisms associated with mineral surfaces, biofilms on stones, lichens, mosses, plant roots growing in soil, and field-collected biological materials that contain hard tissues or mineral particles. Conventional biological electron microscopy is often difficult to apply to such samples because they are too large, too heterogeneous, or too hard for standard sectioning. By combining biological preservation with cross-sectional polishing, bioCP-SEM may provide a way to observe a wide range of field-collected specimens that have previously been difficult to examine by electron microscopy.
Looking ahead, we hope that bioCP-SEM will serve as a bridge between ultrastructural imaging and broader environmental analysis. The structural information obtained by this method could be combined with elemental mapping by EDX, isotopic imaging by NanoSIMS, X-ray computed tomography, fluorescence microscopy and AI-based image analysis. Such combinations may allow researchers to connect structure, composition, spatial context and biological activity in complex environmental samples.
For me, this study represents an attempt to bring electron microscopy closer to the natural complexity of environmental systems. The original question was simple: can electron microscopy help us see hidden structures in the environments around us? bioCP-SEM is one answer to that question. It allows us to look at roots, microbes, organic matter and minerals not as separate entities, but as parts of an intact interface. We hope that this approach will open new opportunities for studying the microscopic architecture of the rhizosphere and many other biological habitats in the natural world.
Follow the Topic
-
Communications Earth & Environment
An open access journal from Nature Portfolio that publishes high-quality research, reviews and commentary in the Earth, environmental and planetary sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Climate extremes and water-food systems
Publishing Model: Open Access
Deadline: May 31, 2026
Drought
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in