Solar-driven Efficient Heterogeneous Subminute Water Disinfection Nanosystem Assembled with Fingerprint MoS2
Published in Earth & Environment

As a renewable energy source, solar energy is an attractive option to disinfect water through the method of Solar Drinking Water Disinfection (SODIS), which is a widely accepted method, particularly in developing countries that lack distribution systems for drinking water. Solar energy can be used directly to inactive bacteria, as photons from the ultraviolet B (UVB) or ultraviolet A (UVA) portion of sunlight interact with nucleic acids or other macromolecules to cause inactivation, which is time-consuming. Solar energy also can be used for indirect disinfection, as photons from the ultraviolet or visible portion of sunlight convert into electrons by catalyst to produce reactive oxygen species (ROS) for disinfection. This heterogeneous water disinfection with sunlight can avoid secondary pollution and other shortcomings of homogeneous system. However, at present, its speed and efficiency are far from those of homogeneous processes, which can effectively attack bacteria though molecular/ion scale collision, such as chlorination. Here, we successfully address this issues of heterogeneous disinfection by developing discrete nanoflakes of (Al2O3@v-MoS2)/Cu/Fe3O4 in which vertically aligned 2-D nanofingerprint MoS2 is hybridized with Cu metal and magnetic Fe3O4 particles.
Three exciting features are integrated into such a novel structure of (Al2O3@v-MoS2)/Cu/Fe3O4: bifacial vertically aligned MoS2 grown on the top and bottom surfaces of light-transparent Al2O3 nanoflakes that can absorb not only UV light but also almost the full spectrum of visible light, and both sides can operate simultaneously; a Cu-MoS2 junction that enhances charge separation for the efficient generation of reactive oxygen species (ROS); and magnetic Fe3O4 nanoparticles that have magnetic separation capability and conveniently regenerate after disinfection. (Fig. 1) This system exhibits outstanding water disinfection with thorough inactivation of over 5.7 log10 CFU ml-1 E. coli within 1 minute in real sunlight and with facile separation and reuse.
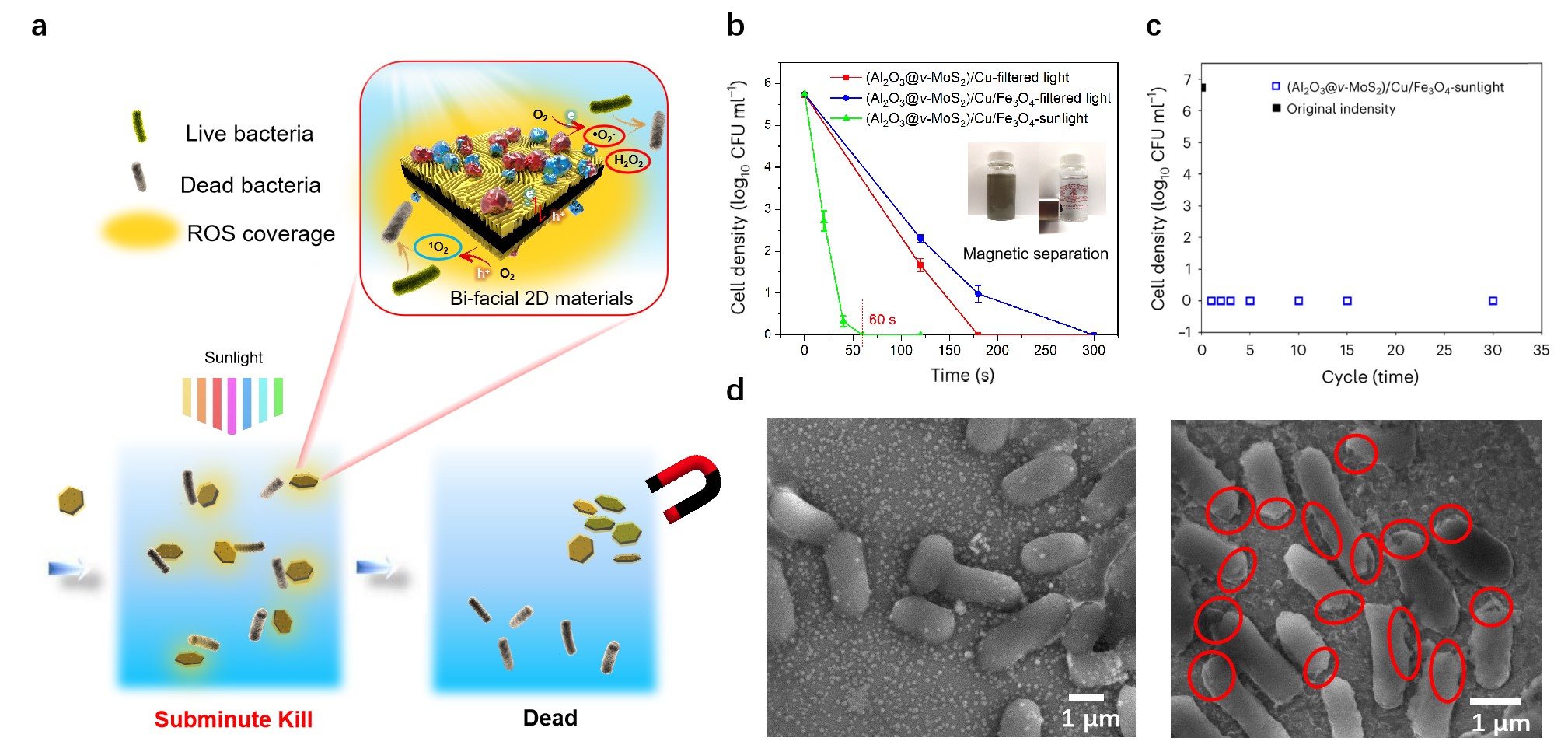
Fig. 1 Solar water disinfection test of recyclable (Al2O3@v-MoS2)/Cu/Fe3O4. a Schematic representation of photocatalytic mechanism and cyclic utilization for water disinfection. b Comparison of the disinfection performance of (Al2O3@v-MoS2)/Cu and (Al2O3@v-MoS2)/Cu/Fe3O4 under different light sources (UV-filtered light and real sunlight). In real sunlight, a thorough disinfection with (Al2O3@v-MoS2)/Cu/Fe3O4 is completed in 60 seconds (sec-kill). (The point at 0 CFU/mL indicate that all the E. coli cells are dead. Experimental conditions: T = 25℃, [pH]0 = 6.5, the initial concentrations of E. coli are 105.74 CFU/mL, the data are presented as mean ± s.d. (n = 3).). c Cyclic system proving the stability of (Al2O3@v-MoS2)/Cu/Fe3O4. d SEM images of solution samples (E. coli) separated at low speed before and after disinfection. After disinfection, the cell membrane was seriously damaged as shown in red circles.
The reasons why our photocatalytic system can disinfect in water so fast are based on the following three points. Firstly, the discrete bi-facial nanoflakes with vertically aligned 2-D nanofingerprint MoS2 can provide abundant active edges for ROS production (Fig. 1a). Secondly, our photocatalyst can absorb most part of sunlight (the cutoff of UV-Vis absorption at around 750 nm, Fig. 2a), which enhances the utilization rate of solar energy from ~7% to ~57%. Last but most important, our structure designed photocatalyst can reach an optimized band gap (Fig. 2e), which can perfectly produce all kinds of ROS (Fig. 2b-d).
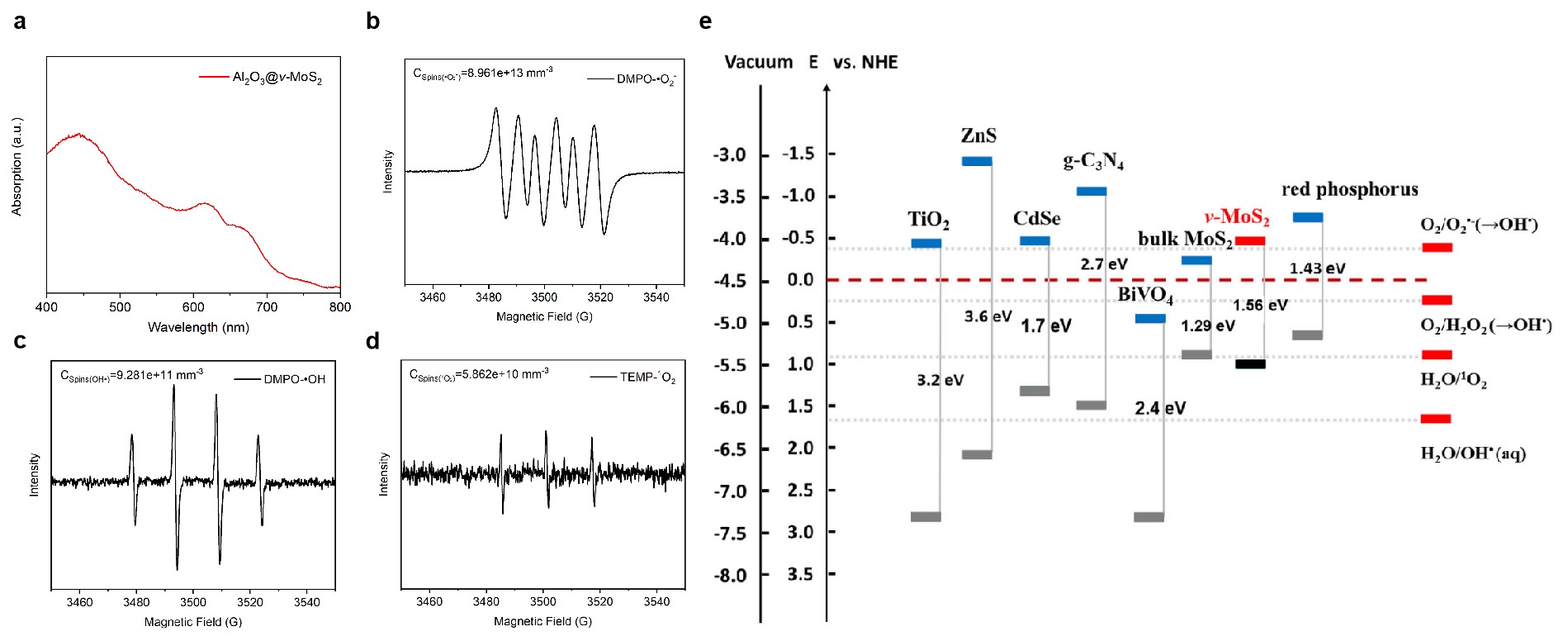
Fig. 2 Key characterization of our photocatalyst to explain its ultra fast water disinfection. a UV-Vis absorption spectra of Al2O3@v-MoS2. The cut-off wavelength of Al2O3@v-MoS2 is approximately 750 nm, whereas Al2O3 has no absorption (P = 1.5 × 10−3 mg l−1). b-d The signals of superoxide radical, hydroxyl radical and singlet oxygen in EPR spectrum, respectively. (1 sun; UV-filtered; 10 mg Al2O3@v-MoS2/Cu in 50 ml water; 25oC). e Band gap energy, valance band maximum, and conduction band minimum positions of several semiconductors for water disinfection, including TiO2, ZnS, CdSe, g-C3N4, BiVO4, bulk MoS2, and red phosphorus, in comparison to that of Al2O3@v-MoS2. The potentials in the absolute energy scale and in normal hydrogen electrode scale were also indicated.
In this study, we explored the ultrafast heterogeneous disinfection system by means of nanostructure predesign, experimental synthesis and performance verification. Our study shows that the selection of photocatalysts with the most suitable band gap width and as many active sites as possible is the key to achieve ultrafast heterogeneous water disinfection. The band gap of the catalyst should be narrow enough to absorb a large amount of the visible energy of sunlight, but wide enough to produce as many kinds of ROS as possible. At the same time, the vertical aligned 2-D fingerprint structure of MoS2 also provides sufficient amount of active edges. We believe that our catalyst design represents state-of-the-art photodisinfection and will inspire more innovations in this exciting interdisciplinary field.
More details can be found in our paper " Solar-driven Efficient Heterogeneous Subminute Water Disinfection Nanosystem Assembled with Fingerprint MoS2" published in Nature Water.
Follow the Topic
-
Nature Water

This journal publishes research on the evolving relationship between society and water resources on a monthly basis. It covers the natural sciences, engineering, and social sciences, with a particular interest in regards to interdisciplinary research.
Related Collections
With Collections, you can get published faster and increase your visibility.
Remote sensing and forests
Publishing Model: Hybrid
Deadline: Apr 30, 2026
Water pollution and advanced treatment processes
Publishing Model: Hybrid
Deadline: May 31, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in