Synthesis of Aroyl hydrazone, Medicinal hydrazone, metal complexes, DFT analysis Molecular docking study
Published in Chemistry
The aroyl tridentate hydrazones are a distinctive class of ligands (triatomic moiety (>C N—N—C<) possesses flexible donor azomethine (>HC N—) group that are effective metal ion chelators. They are perfect pioneers in biological, catalytic potential, increasing efficacy and decreasing toxicity. The structural motifs of aroyl hydrazone have been verified by their multifunctional potential in drug-design, favourable catalytic and ultra-trace detection of elements in biological samples [1-3].
In recent years, there is an enhanced interest in the chemistry of hydrazones and their complexes due to their wide range of antidepressant [4], antibacterial, antiviral, antifungal, analgesic, anti-inflammatory, anticonvulsant and antimalarial activities [5, 6]. The core structure of hydrazone ligand contains the nucleophilic imine, amino nitrogen, imine carbon (electrophilic and nucleophilic nature), and intrinsic character of (>C N—) bond are played the distinctive role in various potential activities [7]. The functional diversity of aroyl hydrazone ligands are responsible for their pharmacological as well as biological potentials viz., tuberculosis, antitumoral [8, 9] anticoagulant, antiplatelet [10], and enzymatic inhibitors [11].
The structural tautomerism of hydrazone ligand is also responsible for enhancing their flexibility in donation (ONO) and capable to stable metal complex synthesis. A ligand system having electronegative atoms like nitrogen, oxygen, sulphur and halide atoms also enhances the coordination possibilities [12, 13]. Further, the hydrazone Schiff bases also exhibit numerous biological applications such as insecticides, herbicides, rodenticides, nematicides and plant growth regulators [14]. Compared with simple hydrazone Schiff bases, aroyl or heteroaroyl Schiff base ligands have additional donor sites which introduces a wider range of properties for these molecules [15].
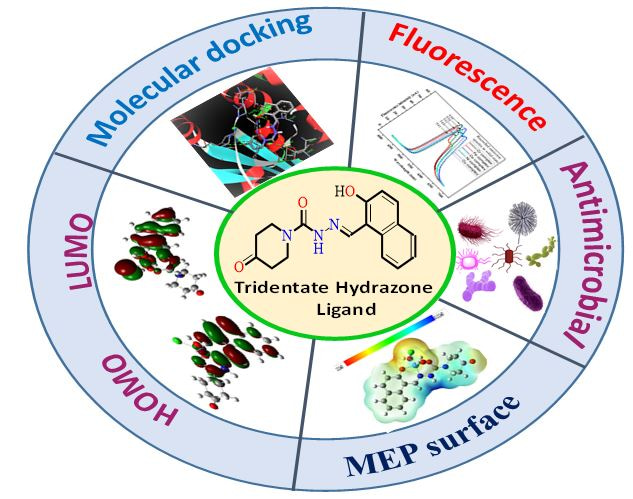
To the best of our knowledge, there is no such report was found in literature on synthesis of aroyl hydrazone bearing 4-oxopiperidone moiety and therefore our interest has been grown up to synthesis of importance associated hydrazone ligand and its metal complexes. Such tridentate ligand displayed their better coordinating properties with metal ions showing the pharmacological potential, fluorescence, thermo-kinetic stability. Furthermore, the aroyl hydrazones ligands demonstrated a paramount role in the field of medicinal chemistry as the iron chelators in vivo as well as in vitro for the iron overload treatment. The excellent biological sensitivities of aroyl hydrazones and their mode of chelation with transition metal ions present in living systems have been of significant interest.
Additionally, aroyl carbohydrazone are known to have strong chelating properties resulting in the several geometries e.g. bidentate to multidentate linkages via ONO and N2O4, NO donor system. The metal complexes of Mn(II)L, Co(II)L, Ni(II)L, Cu(II)L and Cr(III)L and Fe(III)L have been displayed role in the various diseases belonging to the antioxidant, DNA and cytotoxicity [16]. Therefore, it is envisaged that the 4-oxopiperidone moiety involving in the hydrazide, (E)-N'-((2-hydroxynaphthalen-1-yl)methylene)-4-oxopiperidine-1-carbohydrazide ligand (H2L,C) (Scheme 1) and its metal complexes would result in compounds of interesting biological activities.
Consequently, in the light of the foregoing deyails and as a continual of our work on the synthesis of Schiff base complexes, here we report synthesis, spectroscopic characterization, thermal behaviours and the antimicrobial susceptibility of Cr(III)L, Fe(II/III)L Mn(II)L, Co(II)L, Ni(II)L and Cu(II)L complexes with above ligand. DFT calculation has made to confirm the molecular structures and to determine the quantum chemical parameters. Moreover, in silico molecular docking has been used to determine possible binding interaction of the ligand with gram‐positive bacteria (E.coli) and fungal protein. In addition to above, an in silico-ADMET prediction has also been presented for the development of future potential research, pharmacological applications and drug developments.
Follow the Topic
-
Research on Chemical Intermediates
This journal publishes current research articles and concise dynamic reviews on the properties, structures and reactivities of intermediate species in all the various domains of chemistry.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in