The Nanoplastic Trojan Horse: Evaluating Interfacial Adsorption and Intracellular Toxicity Vectors
Published in Chemistry, Earth & Environment, and Ecology & Evolution
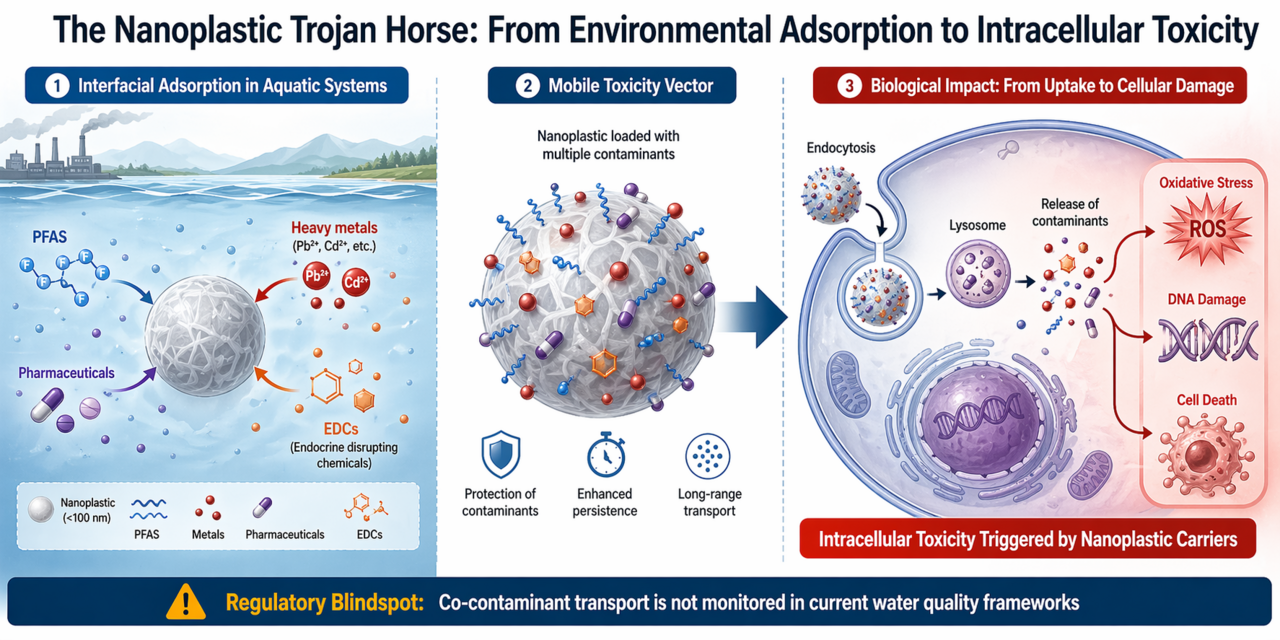
Beyond Physical Pollution in Aquatic Ecosystems
When discussing plastic pollution, the environmental engineering community often focuses on the macroscopic and physical hazards of microplastics—such as gastrointestinal blockages in aquatic life or the bio-fouling of municipal membrane filtration systems. However, as these polymers undergo progressive photo-oxidation and mechanical fragmentation down to the nanoscale (<100 nm), the fundamental nature of their threat changes.
At the nanoscale, plastics cease to be merely inert physical pollutants. Because of their immense surface-area-to-volume ratio, they transition into highly active interfacial vectors. They are no longer just floating debris; they are dynamic chemical sponges with profound implications for the health impacts of nanoplastics in drinking water.
The Trojan Horse Mechanism: Co-Contaminant Adsorption
In complex aquatic matrices, nanoplastics do not exist in isolation. Through a combination of hydrophobic interactions, pi-pi stacking, and electrostatic forces, their surfaces rapidly scavenge and concentrate legacy pollutants from the bulk water.
This includes heavy metals, endocrine-disrupting pharmaceuticals, and even other highly recalcitrant organics like PFAS. A nanoplastic particle traveling through a wastewater effluent stream acts as a mobile concentrator, gathering a highly toxic payload onto its surface. This phenomenon of co-contaminant adsorption on microplastics is commonly referred to in ecotoxicology as the "Trojan Horse" effect, and it fundamentally alters how contaminants move through the environment.
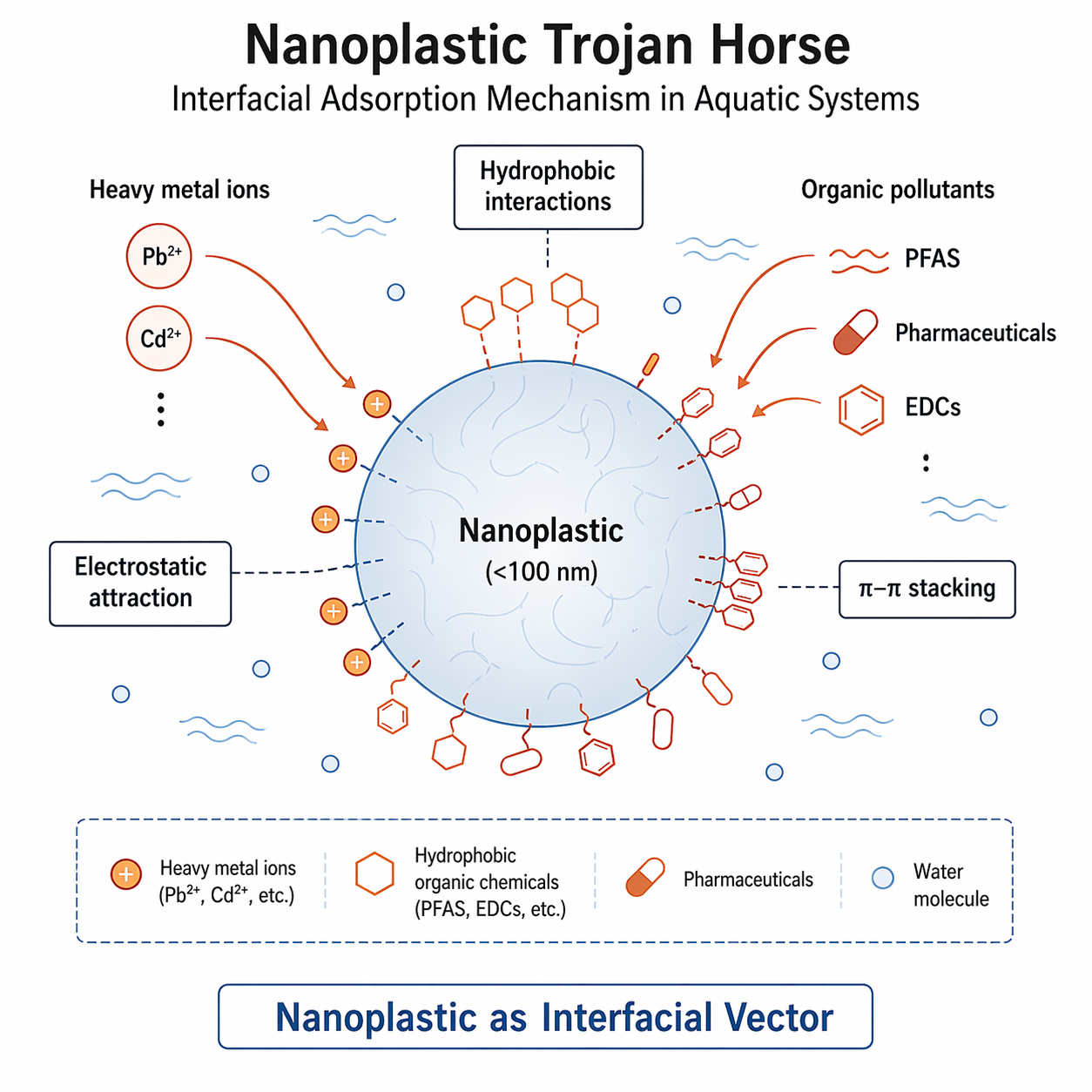
Crossing the Biological Barrier: Intracellular Toxicity
The true danger of the nanoplastic Trojan Horse is its ability to bypass advanced cellular defenses. Due to their sub-cellular dimensions and lipophilic nature, the cellular uptake of nanoplastics is alarmingly efficient. They can readily cross highly selective biological barriers, including the gut epithelium, the placental barrier, and even the blood-brain barrier.
Once internalized via cellular mechanisms like endocytosis, the nanoplastic-pollutant complex is transported into the intracellular space. Crucially, when these complexes reach the acidic environment of the lysosomes, the sudden pH shift alters the interfacial binding affinities. This triggers the "Trojan Horse" to open—rapidly desorbing and releasing its concentrated payload of heavy metals and EDCs directly into the cytosol. This localized release causes severe oxidative stress, DNA damage, and cellular apoptosis.
The Regulatory Blindspot (An Industry Opinion)
As researchers, we must call out a glaring gap in our global infrastructure: our regulatory frameworks are entirely unprepared for this. Current water quality guidelines from the EPA or WHO treat heavy metals, PFAS, and plastics as completely separate regulatory categories.
There are currently no standardized protocols for monitoring how these contaminants interact together at the nanoscale. If our biosensors and analytical labs are only looking for dissolved lead or free-floating PFAS, they will completely miss the massive concentrations of these toxins being smuggled inside nanoplastic vectors. We cannot regulate what we do not accurately measure.
The Danger of Incomplete Remediation
This cellular reality exposes a massive vulnerability in how we design water treatment systems. If we rely on conventional treatments that merely fragment microplastics without achieving full mineralization, we are inadvertently engineering a more dangerous problem. Breaking a single microplastic into a thousand nanoplastics exponentially increases the surface area available for toxic adsorption and biological infiltration.
Conclusion
The presence of nanoplastics in our water cycle forces a paradigm shift in environmental toxicology. We can no longer evaluate the toxicity of plastics in isolation; we must account for their role as intracellular transport vectors for co-contaminants.
Furthermore, this underscores the critical thesis I advocate for: we cannot settle for the incomplete oxidation or physical filtration of emerging contaminants. True water security requires deep-tech, advanced oxidation processes for nanoplastic mineralization. We must engineer targeted catalytic systems capable of absolutely destroying polymeric backbones before they become vectors.
As we push the boundaries of environmental engineering, how should global regulatory frameworks adapt to measure and restrict the "Trojan Horse" effect of nanomaterials in our drinking water? Should standard compliance testing require intracellular toxicity screening?
Follow the Topic
Introducing the Palgrave Macmillan Campaign for the Humanities
At Palgrave Macmillan we publish cutting-edge humanities research that has real-world impact. This research community brings together the voices of our authors and editorial team to highlight and publicize the value of the humanities and humanities research in our world today.
Continue reading announcement

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in