Thermally-induced atropisomerism promotes metal-organic cage construction
Macrocycles have a crucial role in constructing self-assembled structures in both biological creatures and artificial systems.1,2 The behavior of molecules and their self-assemblies, including conformational inversion and reassembly, is in response to environmental stimuli. These phenomena are crucial for modifying the precise structure and associated characteristics of these systems. The stimuli include light,3 acid-base,4 guest molecules,5 and temperature.6 Temperature response is a widespread occurrence in biological systems. However, in artificial systems, the ability to respond to external stimuli is mostly found in extremely flexible macrocycles, such as calixarene. Thermally-induced conversion mainly involves the transformation between the two forms. In 2019, Gong’s group successfully created a set of stable atropisomers with a stiff intrinsic structure and demonstrated the ability to convert between these isomers at a temperature of 473 K.7 Controlling the conversion between three or more distinct rigid isomers of artificial systems through heating remains a challenge.
Meanwhile, macrocyclic molecules have an essential function as ligands in self-assembly systems. Recently, they have been widely used in the construction of metal-organic molecular cages. The coordination cages have potential as advanced functional materials because of their large interior volume, regulated cavity environment, and high porosity. Currently, it is noteworthy that macrocycles are often assembled in fixed forms. Functionalized metal-organic self-assembly, such as metal-organic cages, typically necessitates the prior modification of ligands, complex synthesis procedures, and/or the addition of guest molecules. However, the development of controlling macrocycle structure and its corresponding assembly structure is still in progress.
Gong et al. developed a nanoscale transformation that uses thermal response to create organometal cages from large macrocycle building blocks. Pyridine groups were incorporated in the design and synthesis of cyclo[4](1,3-(4,6-dimethyl)benzene)[4](1,3-(4,6-dimethyl) benzene)(4-pyridine), resulting in the formation of three different atropisomers.
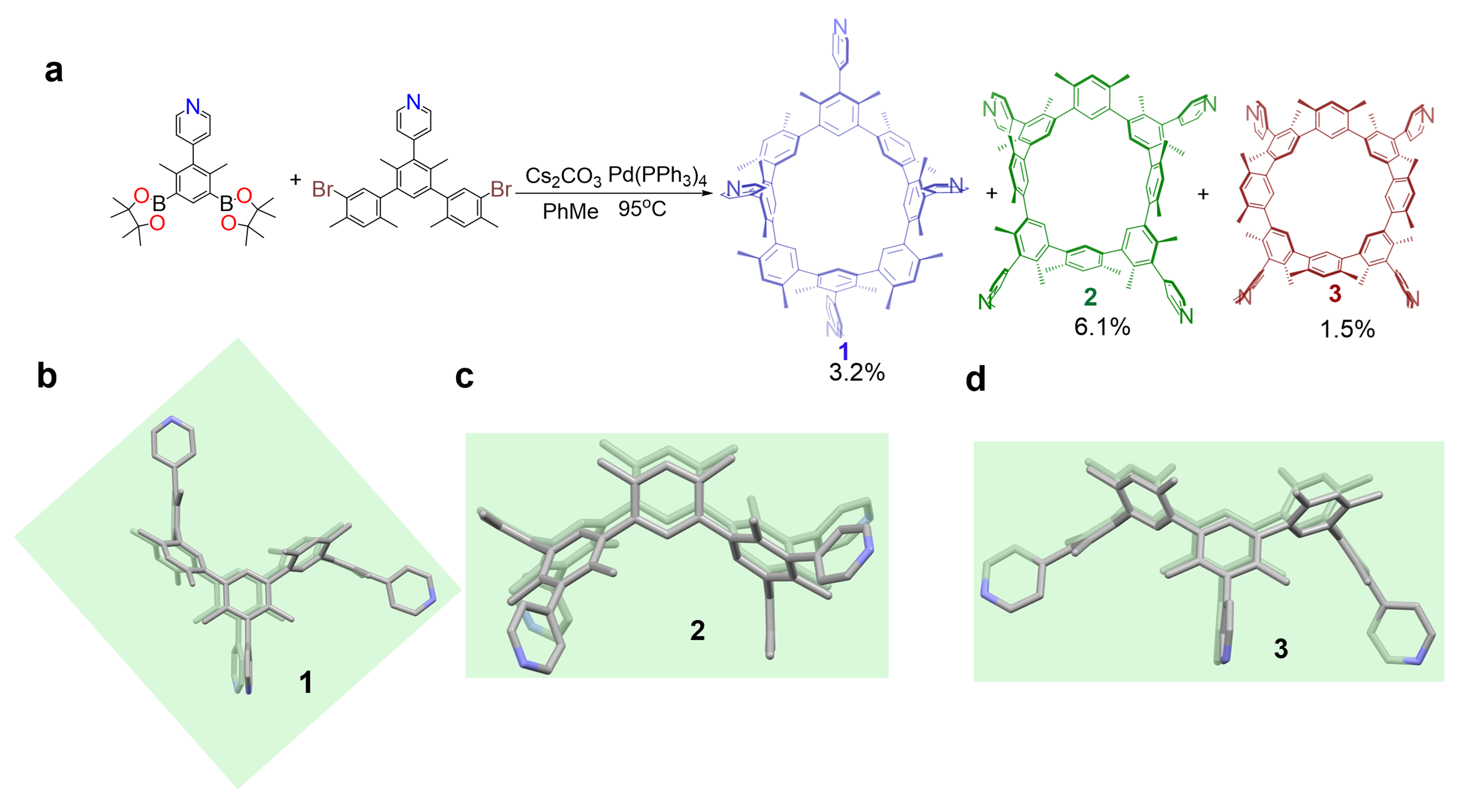
Fig. 1 Synthesis and single crystal X-ray diffraction structures corresponding to the macrocyclic atropisomers (1, 2, and 3).
Thermally Induced Controlled Conversion of Atropisomers
At 298k, there are three distinct rigid atropisomers that can exist in a stable manner. Two of these atropisomers exhibit Cs symmetry (1 and 2), while the third atropisomer exhibits C4v symmetry (3). When heated at different temperatures, the three atropisomers can achieve resistive isomerization between the inherent skeleton. At 393 K, the conversion from between 1 and 2 is the only chemical equilibrium that occurs, whereas 3 does not take part in the process. At 473 K, the conversion of 1 and/or 2 to 3 is quantitative. The conversion energy barrier between the substrates was determined based on the experimental data. The experimental results demonstrate that the interconversion among the three atropisomers is controllable.
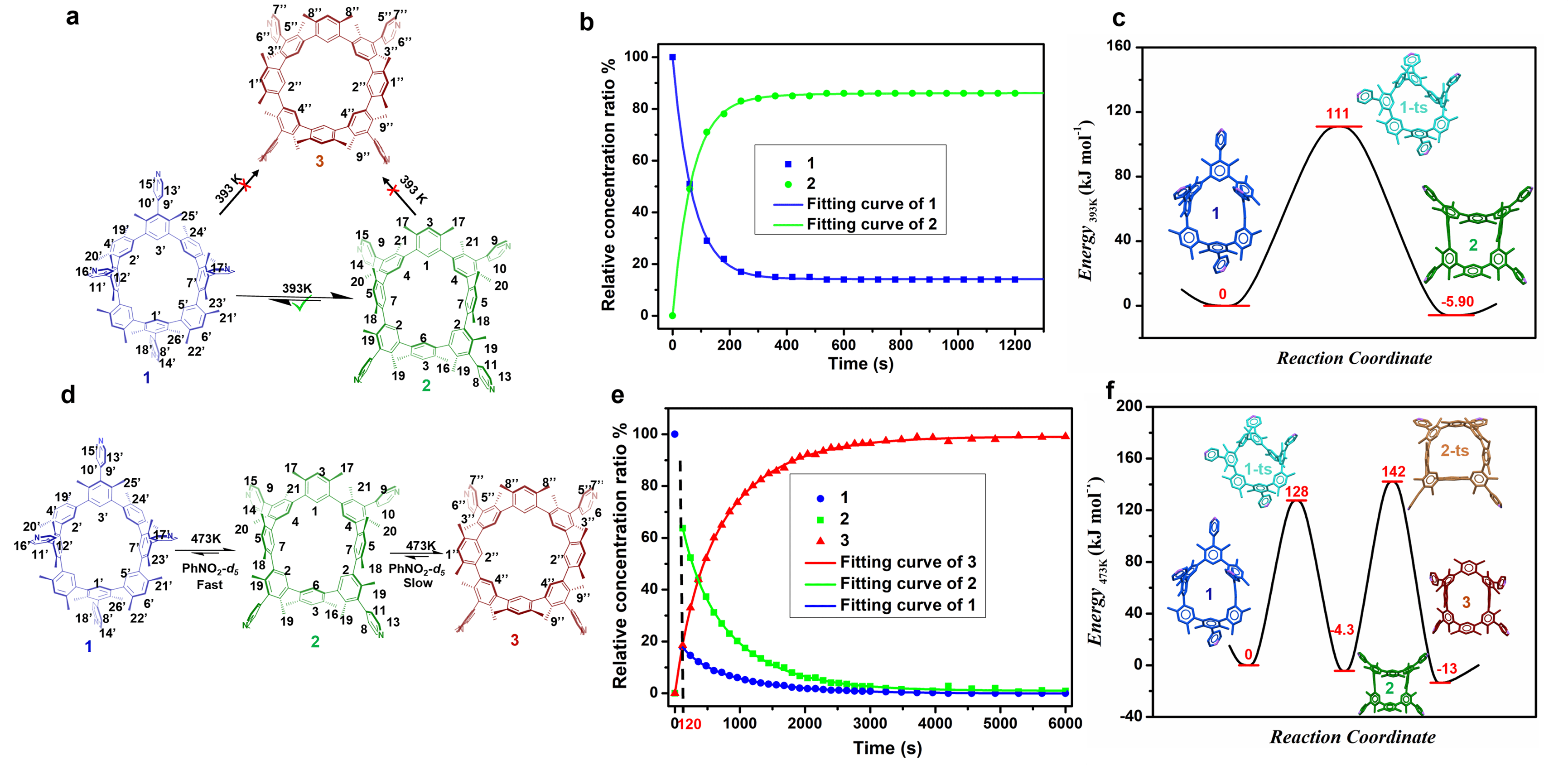
Fig. 2 (a) Schematic representation of the conversion between 1, 2, and 3 in PhNO2-d5 at 393 K. (b) Time-dependent relative concentration ratio changes of 1 (blue dot) and 2 (green dot) in PhNO2-d5 at 393 K. (c) the potential energy diagram of the conversion between 1 and 2 in PhNO2-d5 at 393 K. (d) Schematic representation of the transformation between 1, 2, and 3 in PhNO2-d5 at 473 K. (e) Time-dependent conversion between 1 (blue dot), 2 (green dot), and 3 (red dot) and related non-linear fitting curves (solid lines) at 473 K. (f) The potential energy diagram for the conversion between 1, 2, and 3 in PhNO2-d5 at 473 K.
Thermal Promoted Construction of Metal−organic Cage
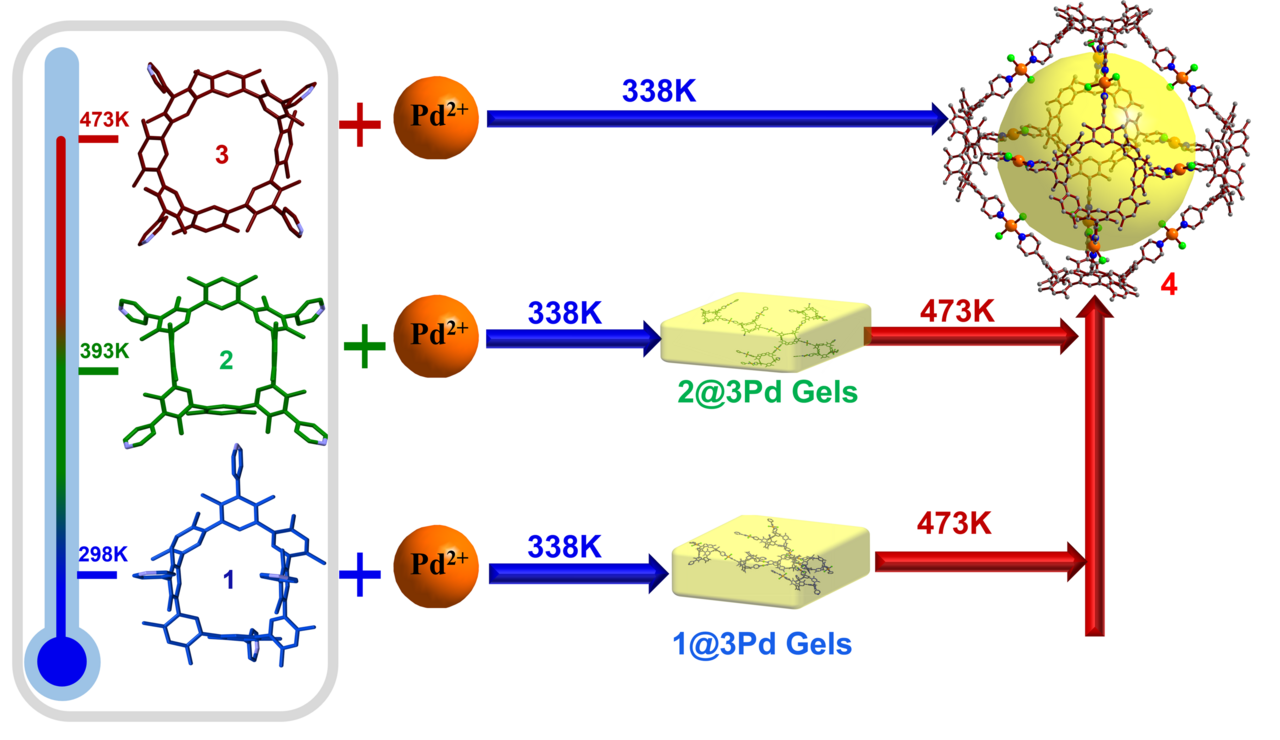
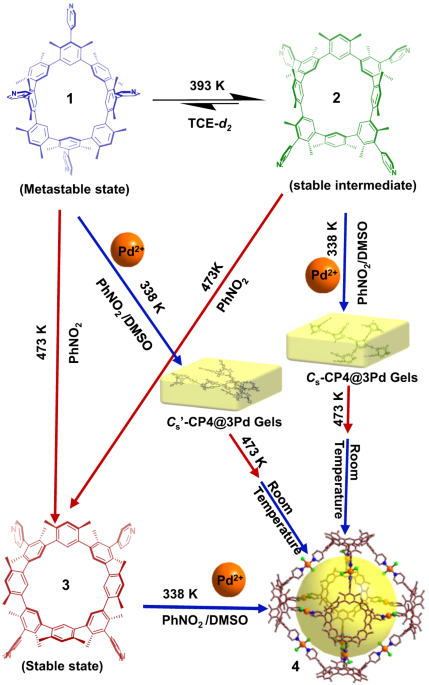
Furthermore, we have successfully accomplished the specific conversion of thermally induced atropisomers, while also further controlled the assembly structure of macrocycle involvement. Octahedron-like metal-organic cage 4 was synthesized by stepwise or in situ synthesis. The coordination of 3 with Pd2+ can directly form metal-organic cage 4 at 338 K. A yellow gel is generated in situ when Pd2+ is mixed with either 1 or 2. Upon heating, the macrocycles undergo a stiff skeleton inversion, leading to a transformation of the initial gel to the self-assembling cage structure (i.e., 4). This phenomenon can be considered a unique instance of the transformation into self-assembly of thermally responsive macrocycles on the nanoscale, based on current knowledge. This technique offers a novel approach for the fabrication of thermally responsive materials. To the best of our knowledge, this is a unusual example of the transition to self-assembly containing thermally responsive macrocycles at the nanoscale. It provides a new strategy for the preparation of thermally responsive materials.
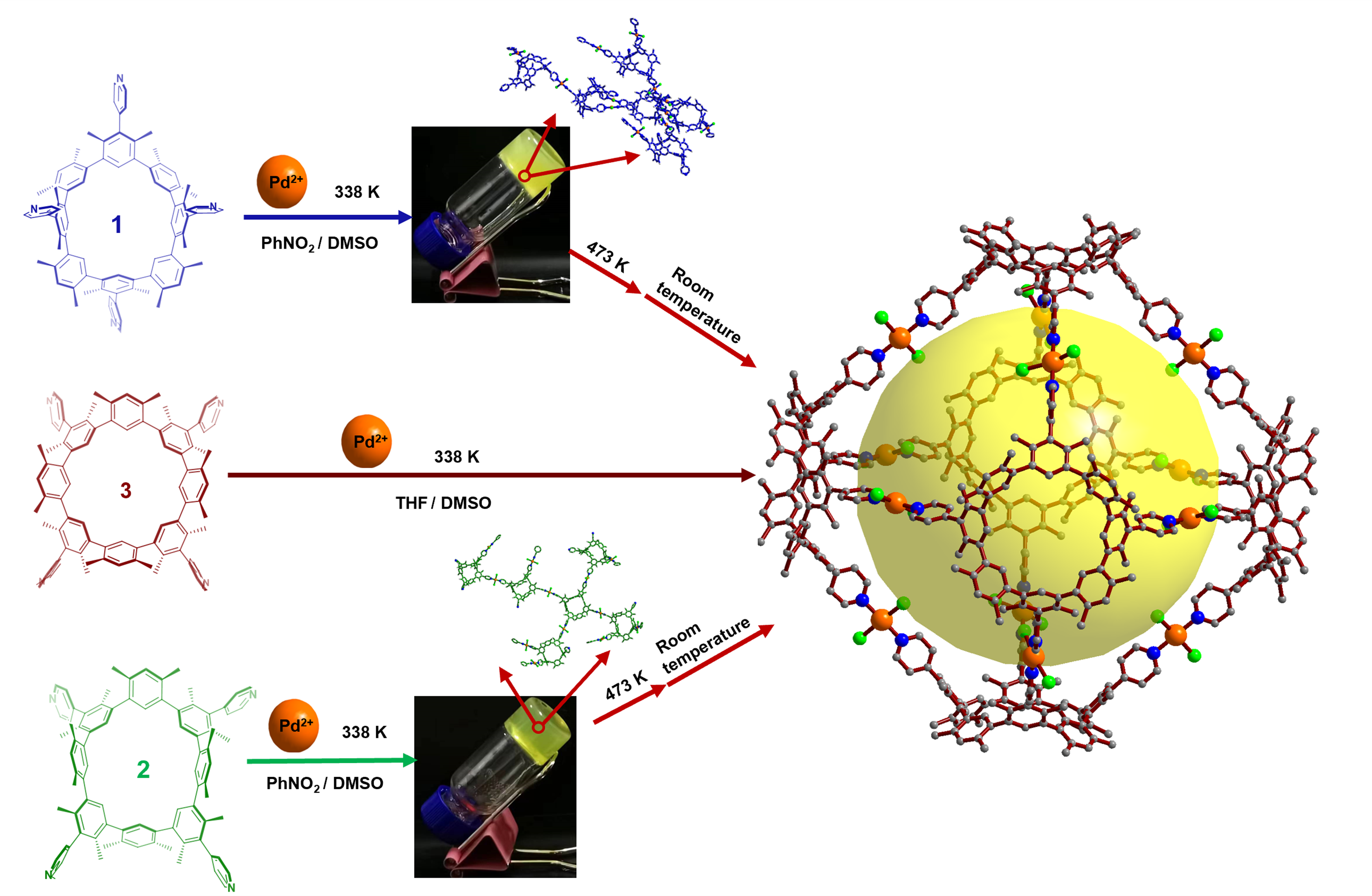
Fig. 3 The cage 4 construction from the mixture containing 1, 2, or 3 and Pd2+
Summary and Outlook
In this study, we report the macrocycle involving three rigid atropisomers with stability at room temperature. Controlled conversion between rigid atropisomers can be achieved with thermal induction at different temperatures. Moreover, the self-assembly structure is additionally governed by manipulating the conformational inversion of the atropisomers. This work provides a complementary strategy for controlling the structure and properties of macrocycles and related self-assembled systems.
References
1 Xie, T. Z., Guo, C., Yu, S. Y. & Pan, Y. J. Fine-tuning conformational motion of a self-assembled metal-organic macrocycle by multiple C-H...anion hydrogen bonds. Angew. Chem., Int. Ed. 51, 1177-1181 (2012).
2 Yang, Y.-D., Chen, X.-L., Sessler, J. L. & Gong, H.-Y. Emergent Self-Assembly of a Multicomponent Capsule via Iodine Capture. J. Am. Chem. Soc. 143, 2315-2324 (2021).
3 Yao, J. et al. Overtemperature-protection intelligent molecular chiroptical photoswitches. Nat. Commun. 12, 2600 (2021).
4 Lin, H.-Y., Wang, Y.-T., Shi, X., Yang, H.-B. & Xu, L. Switchable metallacycles and metallacages. Chem. Soc. Rev. 52, 1129-1154 (2023).
5 Huang, G.-B., Wang, S.-H., Ke, H., Yang, L.-P. & Jiang, W. Selective Recognition of Highly Hydrophilic Molecules in Water by Endo-Functionalized Molecular Tubes. J. Am. Chem. Soc. 138, 14550-14553 (2016).
6 Hitosugi, S., Nakanishi, W., Yamasaki, T. & Isobe, H. Bottom-up synthesis of finite models of helical (n,m)-single-wall carbon nanotubes. Nat. Commun. 2, 492 (2011).
7 Yang, Y.-D. & Gong, H.-Y. Thermally activated isomeric all-hydrocarbon molecular receptors for fullerene separation. Chem. Commun. 55, 3701-3704 (2019).
Follow the Topic
-
Nature Communications

An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Mar 24, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in