Crystal and glass, two main states of solid, are both formed from high-temperature melts. The melt structures relate to the macro-properties of melts, the micro-processes of crystal growth, the formation of defects in crystals, and the structures and properties of glasses, and thus are an important fundamental issue involving a number of disciplines, such as physics, chemistry, material, and geoscience. However, the knowledge about melt structures is very limited so far due to the lack of suitable analytical tools. In 2008, our group discovered the universal principle of the structural transition from a crystal to its melt by using in-situ, high-temperature Raman spectroscopy (Cryst. Growth Des. 2008, 8, 412‒414), which paves a new way to explore melt structures. In 2013, a density functional theory (DFT) computational method was adopted for the first time to accurately simulate a melt Raman spectrum (CrystEngComm 2013, 15, 995‒1000), which ultimately helps to establish a full method to analyze melt structures. After that, a series of special molecular structures only present in high-temperature melts were discovered, the anomaly phenomenon of viscosity found in borate melt was interpreted, and the growth mechanisms of numerous functional crystals were revealed.
As a fundamental material, the structure of the GeO2 melt has attracted much attention for decades. It is widely accepted that germanium in the GeO2 melt is coordinated by at least four oxygen atoms. Recently, our group used high-temperature Raman spectroscopy and DFT computation to investigate the structural origins of two mysterious peaks located at 340 and 520 cm-1 in the Raman spectrum of a GeO2 melt. Threefold coordinated germanium was found in the GeO2 melt (Fig. 1). The results may settle down the long-standing debate on the GeO2 melt structure. The electronic structures of the GeO2 melt (Fig. 2) indicate that not only stable Ge–O bonds but also fluxional Ge–O bonds exist in the GeO2 melt, which can be used to interpret the fluidity, as well as the viscosity, of the GeO2 melt at the molecular level. The discovery of threefold coordinated germanium provides a new insight into the structures of germanate melts, which is helpful for better understanding the formation, defect structures, and properties of germanate crystals/glasses. In addition, as an analog of SiO2, the knowledge of the GeO2 melt structure also has an important implication in geological research.
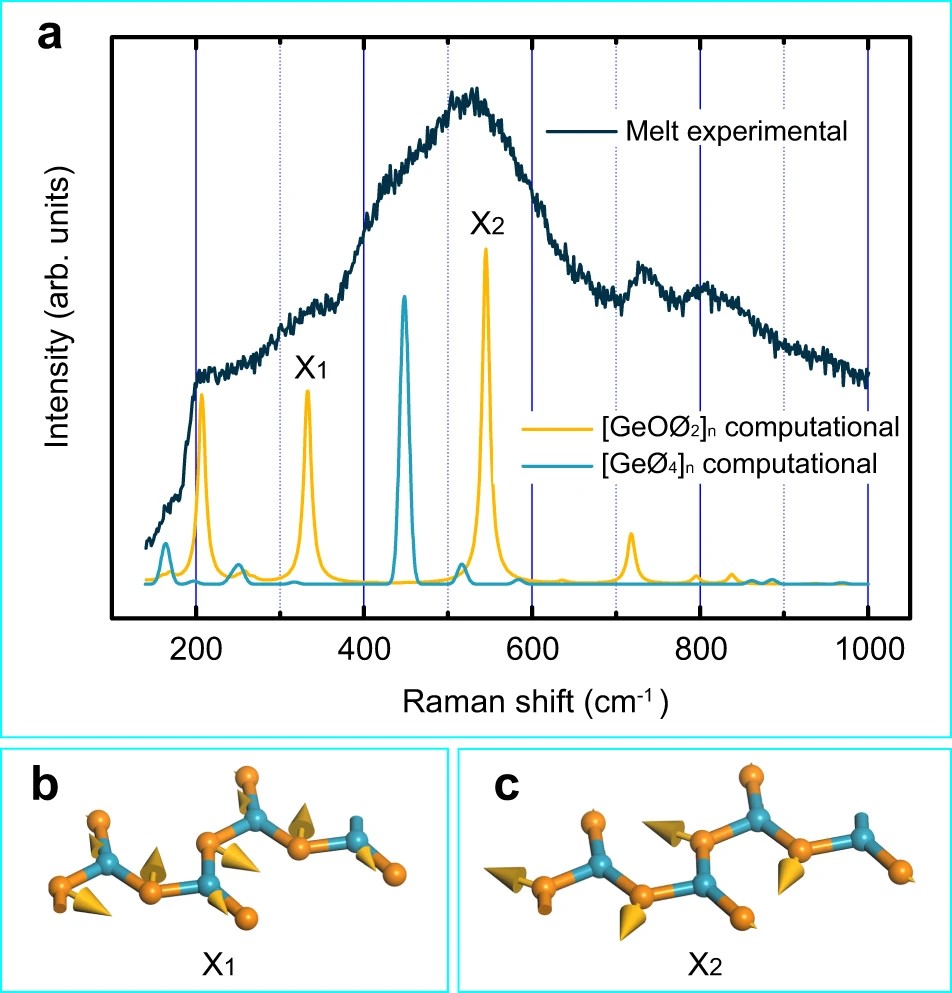
Fig. 1 | Raman spectra of the GeO2 melt and vibrational modes of the two mysterious Raman peaks. (a) Experimental and computational Raman spectra of the GeO2 melt. The melt is composed of [GeOØ2]n chains and [GeØ4]n networks. (b) The vibrational mode of the 340 cm‒1 peak. (c) The vibrational mode of the 520 cm‒1 peak. Blue and orange spheres represent germanium and oxygen atoms, respectively.
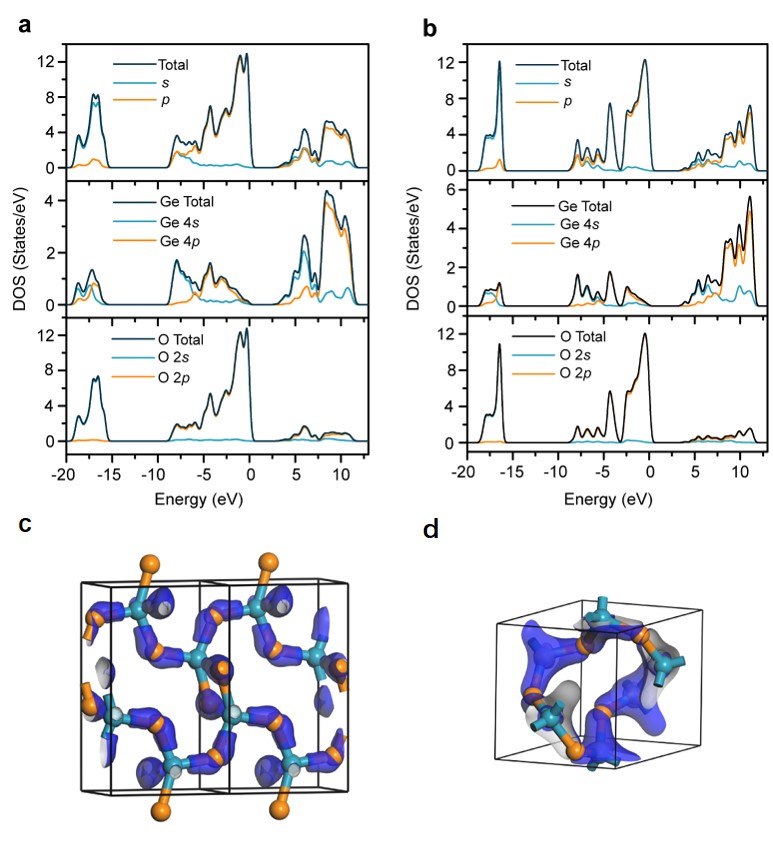
Fig. 2 | Electronic structures of the [GeOØ2]n chain and the [GeØ4]n network (two building units of the GeO2 melt). (a) Total and projected DOSs for the [GeOØ2]n chain. (b) Total and projected DOSs for the [GeØ4]n network. (c) The bonding orbitals of the [GeOØ2]n chain in the energy range from –8.0 to –7.1 eV. (d) The bonding orbitals of the [GeØ4]n network in the energy range from –8.6 to –7.5eV.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in