Transformation of siderite to goethite by humic acid in the natural environment
Published in Chemistry
Iron is the fourth most abundant element, which can form complexes with elements such as C, N, O, and S, making it an essential element for nearly all living organisms. The environmental iron-bearing mineral phases (such as the iron oxides) are aggregates of nano-particles whose performance of adsorption and release of nutrients and metals are highly dependent on the particle size, crystallinity and transformation product. Hence, it is important to understand and reveal the transformation and crystallization mechanisms of such minerals in the natural environment.
As the scholar in the field of water treatment, we have been mesmerized for a while by the minerals transformation in the natural environment to understand the essential mechanism of the migration of heavy metals and nutrients. We noticed that previous studies reveal that mineral transformation occurs under experimental conditions that require high temperature and pressure, specific pH, and higher Fe concentrations. By studying the crystallization mechanism (Ostwald ripening and Oriented attachment), we found that the magnitude of the free-energy barrier of nucleation is a significant factor in determining the properties and the number of particles produced. At low concentrations, temperatures, and pressures, the free-energy barrier is relatively large, so the processes of nucleation and growth should be rare in the natural environment in those predicted by classical models. However, mineral transformation happens in nature all the time, if this transformation mechanism can be applied to water treatment engineering, it will greatly improve the water quality with minimal cost. It is a pity that the crystallization path in the natural environment is not well established and needs further investigation.
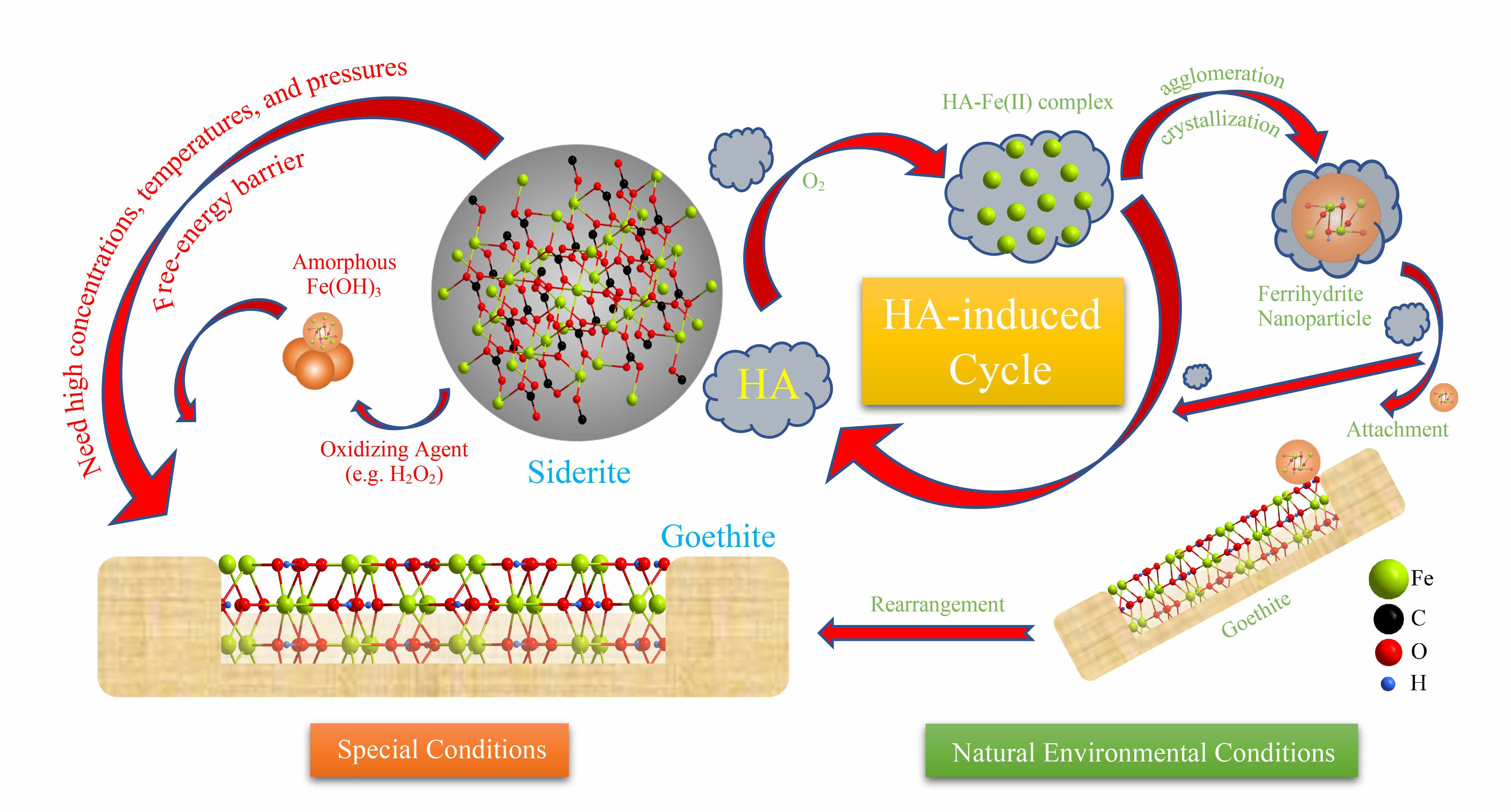
It caught our attention through the groundbreaking work of Nico A.J.M. Sommerdijk, who find many biomineral crystals form complex non-equilibrium shapes, often via transient amorphous precursors, and the organic matter (OM) can induce the formation of liquid precursor. Hence, the knowledge of the OM influence is critical for the understanding the mechanism of mineral transformation in the environment. Humic acid (HA) is a representative natural OM and a most frequently found organic compound in soil, possessing abundant hydroxyl and carboxylic groups that facilitate its adsorption onto minerals. To explore the different transformation paths caused by different precursor particles, the oxidation of siderite by H2O2 and dissolved oxygen (DO), and HA are considered and compared. The major difference between the three is that humic acid can chelate with the released ferrous ions to form a Fe(II)-HA complex, which accelerates the dissolution and oxidation of Fe(II) from the surface of siderite; then owing to the Fe(II)-HA complex keeping Fe atoms within the effective distance of each other, and the formation of ferrihydrite occurs by the agglomeration and crystallization of Fe atoms. In biomineralization, transient amorphous precursor particles (mesocrystals) enable the efficient transport of multi-ion complexes in low concentration to the crystallization site. Hence, with the effect of humic acid, the primary nanocrystals achieve crystallographic alignment despite spatial separation from each other. These findings are important in understanding the pathway of Fe-mineral transformation in the natural environment. The results not only support the supposition that the mesocrystal is a necessary precursor to the single-crystal attachment but also confirm a fundamental step for crystal growth.
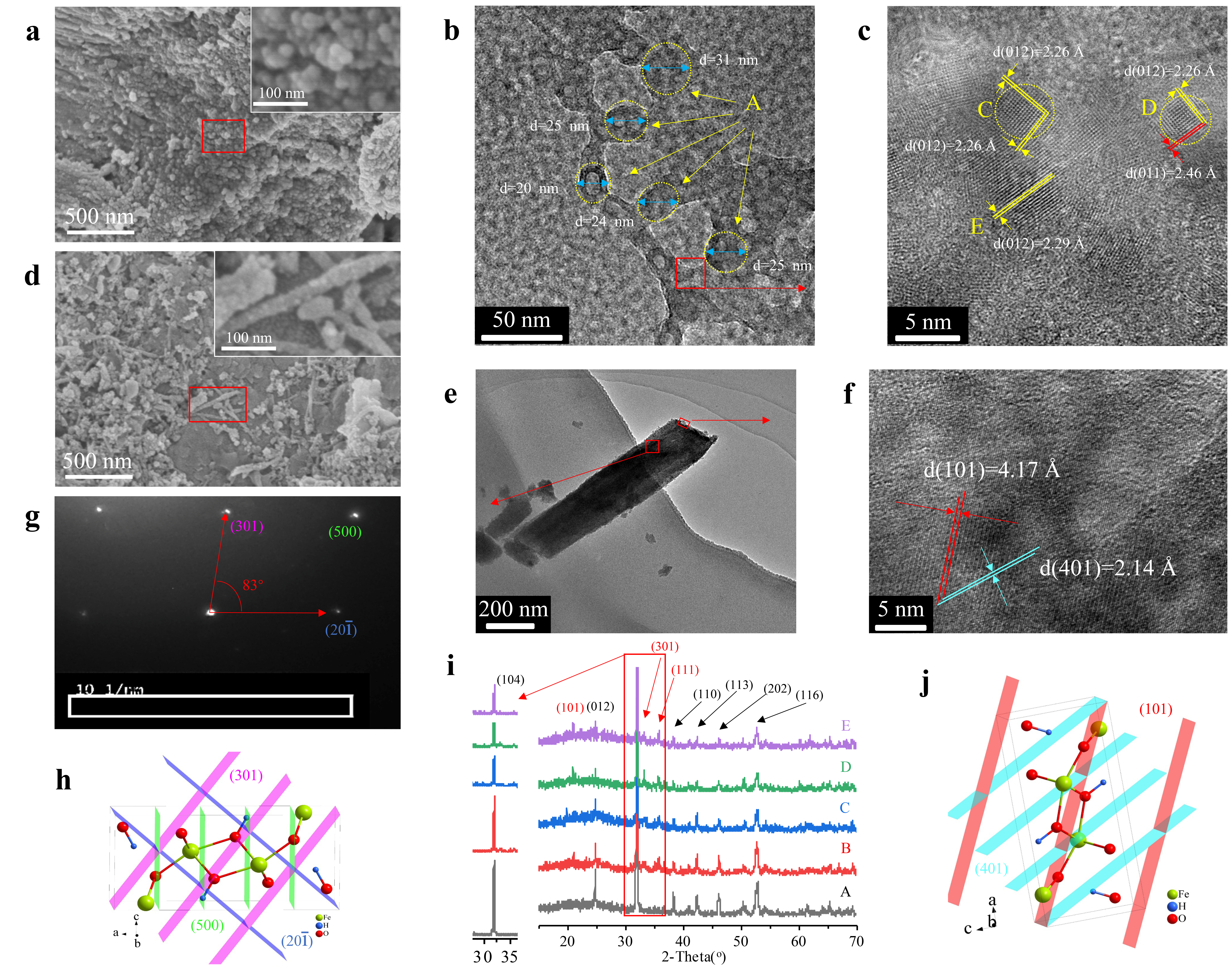
Fig. 1. Characterization of the precipitates. Representative SEM (a and d) and TEM (b and e), HRTEM (c and f), and SAED (g) images of the precipitates of siderite after reacting with HA (2 mg/L); the XRD (i) pattern of the synthesized siderite after reacting with 0 mg/L (i-A), 0.5 mg/L (i-B), 1 mg/L (i-C), 2 mg/L (i-D) and 10 mg/L HA (i-E); schematic diagrams of goethite (h and j) unit cell structures, which were drawn by the Diamond 3.2 software (Crystal Impact GbR, Germany). Samples were freeze-dried for 12 h after 7 days of aging. The concentration of dissolved oxygen (DO) were 4 mg/L.
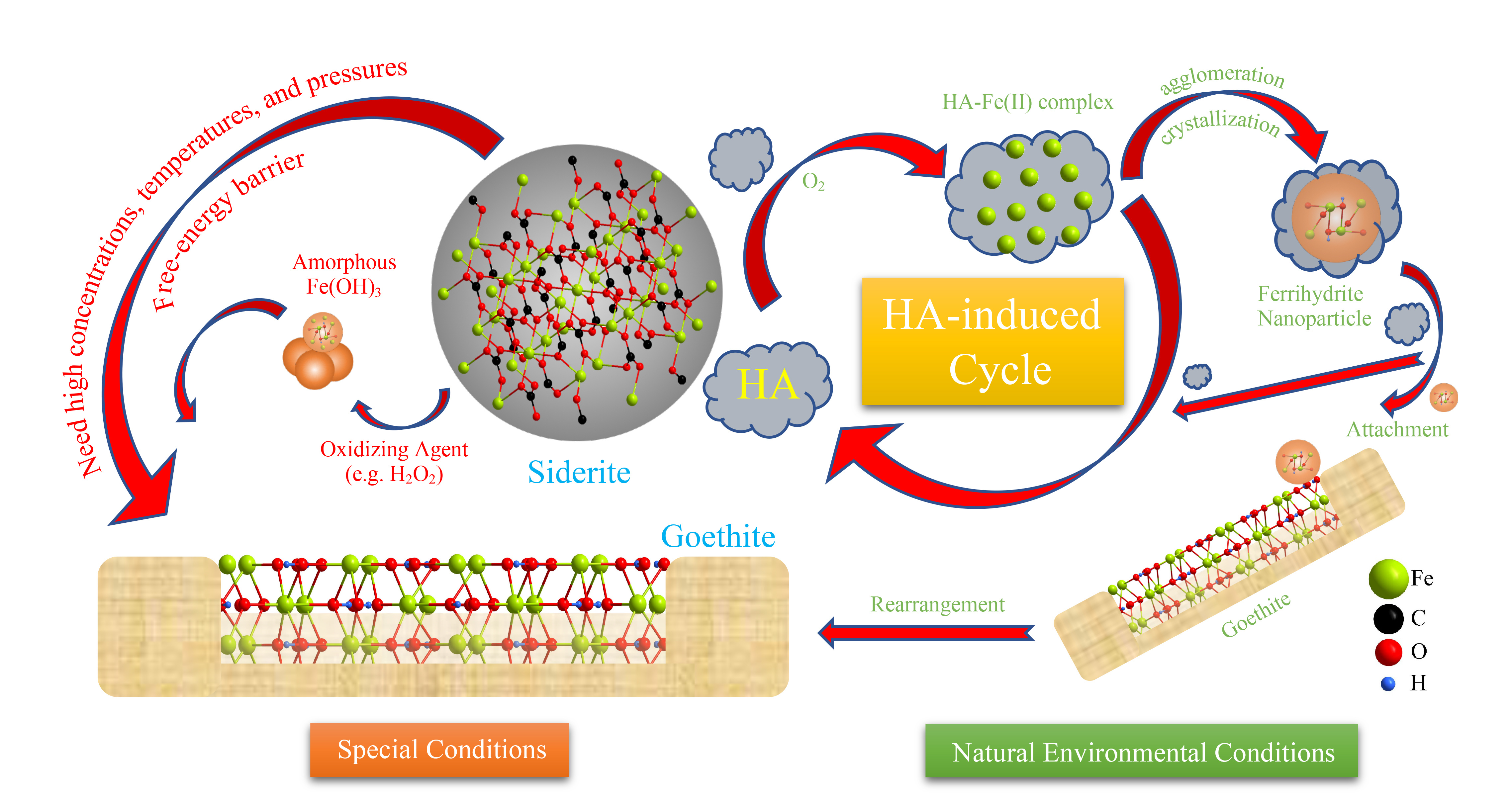
Fig. 2. Path diagram of mineral transformation. Possible pathways of mineral transformation from siderite to goethite by humic acid.
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Sustainable waste management through polymer upcycling
Publishing Model: Open Access
Deadline: Aug 31, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in