Ultra Low Loading Palladium Catalysts Enable Selective and Scalable Synthesis of E-Alkyl Alkenes
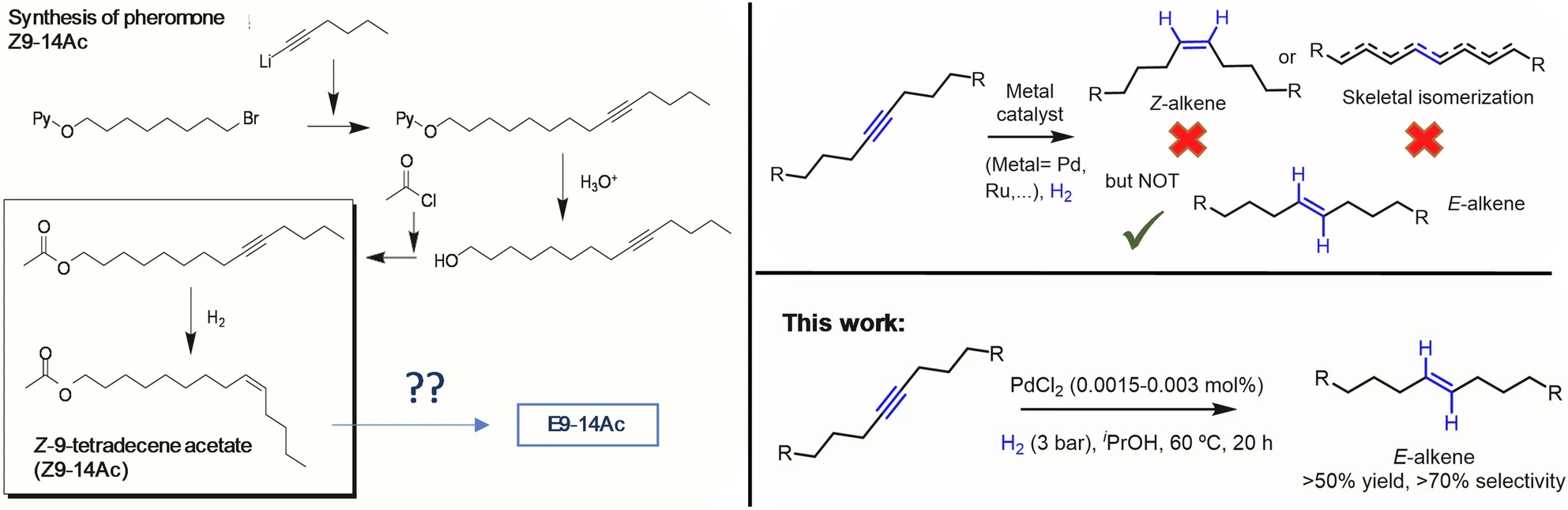
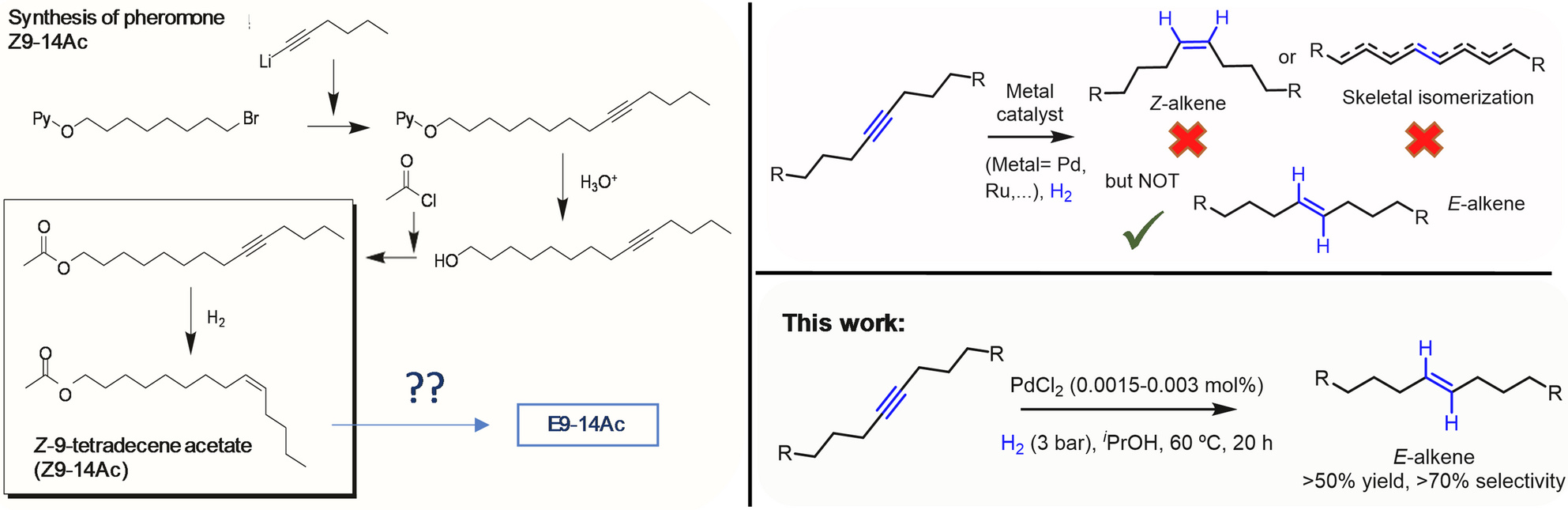
In our recent study, we report a practical and industrially viable catalytic methodology that enables the direct synthesis of E‑alkyl alkenes from internal alkynes using only parts‑per‑million (ppm) levels of palladium (Pd). By employing simple Pd salts such as PdCl₂—in the absence of ligands and at exceptionally low metal loadings—we demonstrate that ultrasmall Pd(0) clusters formed in situ can drive both the Z‑selective semi‑hydrogenation of internal alkynes and the subsequent Z/E isomerization, with minimal side reactions and excellent stereocontrol.
Our findings address a long‑standing synthetic challenge: unlike Z‑alkenes, which can be routinely accessed via classical Lindlar catalysis, the catalytic preparation of E‑alkyl alkenes from alkynes has remained largely inaccessible, forcing industry to rely on cost‑intensive and environmentally burdensome stoichiometric reagents. The methodology presented in our work offers an unprecedented alternative—both economical and scalable—that is immediately applicable to the pheromone industry, where stereochemical purity directly impacts biological activity and commercial viability.
Key Insights
- Ultrasmall Pd(0) clusters are the true catalytic species
Through HAADF‑STEM, XANES, and EXAFS analyses, we confirm that ppm‑level PdCl₂ is reduced under hydrogenation conditions to generate sub‑nanometric Pd(0) clusters and individual atoms, which catalyze both key reaction steps while suppressing unwanted skeletal isomerization and over‑hydrogenation.
- Solvent dictates the E‑selectivity
We find that isopropanol (iPrOH)—unlike ethanol—triggers rapid Z/E isomerization, enabling the formation of E‑alkenes in synthetically meaningful yields. Under optimized conditions, E‑alkene selectivity reaches 53% from the model pheromone precursor, with negligible alkane formation.
- Ligand‑free catalysis at ppm metal loading
PdCl₂ performs effectively at 0.0015–0.006 mol%, drastically lower than traditional heterogeneous systems. These minimal loadings significantly lower costs and enhance sustainability without compromising reactivity.
- Exceptional selectivity for alkyl vs. aromatic alkynes
The catalyst system displays an unusual capability: while internal alkyl alkynes are selectively converted to E‑alkenes, aromatic alkynes show no Z/E isomerization, even though they readily undergo semi‑hydrogenation. This selectivity is rare in Pd‑catalyzed hydrogenation chemistry.
- Scalable to industrial conditions
We validate the method at 1‑kg scale, achieving 94% yield of the Z‑alkene intermediate without unwanted by‑products using standard reactor setups and low H₂ pressures (1.5–4 bar). These results demonstrate the robustness and transferability of the catalytic system.
- Electrochemical compatibility
Beyond thermal catalysis, the Pd system also functions under electrochemical conditions, enabling room‑temperature synthesis of E‑alkenes with 67% selectivity—eliminating the need for external hydrogen gas.
Significance of This Advancement
Our methodology offers a cost‑effective, scalable, and sustainable solution for producing E‑alkenes—an advance with immediate implications for agrochemical pheromone synthesis, where E‑isomers often command significantly higher value but are difficult to produce selectively.
Economic analysis indicates:
- Up to 60% reduction in production cost per kg of E‑alkene or Z‑alkene compared with Lindlar/Na‑NH₃ methods.
- 77.8% reduction in hydrogenation‑related cost when replacing Lindlar catalyst with PdCl₂ at ppm loading.
- Recyclability of the Pd catalyst, further improving sustainability metrics.
Taken together, these results establish a new paradigm for the catalytic synthesis of E‑alkyl alkenes and open opportunities for broader industrial applications beyond pheromones.
Authors & Affiliations
Miquel Molina–García †
- Francesco Orlando †
- Ramón Manzorro
- Juan C. Hernández–Garrido
- Vlad Martin‑Diaconescu
- Judit Oliver‑Meseguer
- Marta Mon*
- Jordi Aragón
- Antonio Leyva–Pérez*
Affiliations:
- Instituto de Tecnología Química (Universitat Politècnica de València–CSIC), Valencia, Spain
- Universidad de Cádiz, Spain
- ALBA Synchrotron Light Facility, Spain
- SEDQ Healthy Crops SL., Barcelona, Spain
Corresponding Authors
-
Marta Mon*
Email: marmoco@itq.upv.es -
Antonio Leyva–Pérez*
Email: anleyva@itq.upv.es
How to Cite This Article
Molina–García, M., Orlando, F., Manzorro, R., Hernández‑Garrido, J. C., Martin‑Diaconescu, V., Oliver‑Meseguer, J., Mon, M., Aragón, J., & Leyva–Pérez, A. (2026).
Synthesis of alkyl trans‑alkenes from alkynes by catalytic semi‑hydrogenation reaction with parts‑per‑million of palladium: application to pheromone synthesis.
Catal, 2, 9. https://doi.org/10.1007/s44422‑025‑00015‑5
Follow the Topic
-
Catal
Catal is an open access journal covering full spectrum of catalysis critical advances. From biocatalysts to heterogeneous catalysts, it integrates fundamental and applied sciences. Catal offers a primary platform for researchers and practitioners in the field.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in