Unusual Trisulfide Chemistry
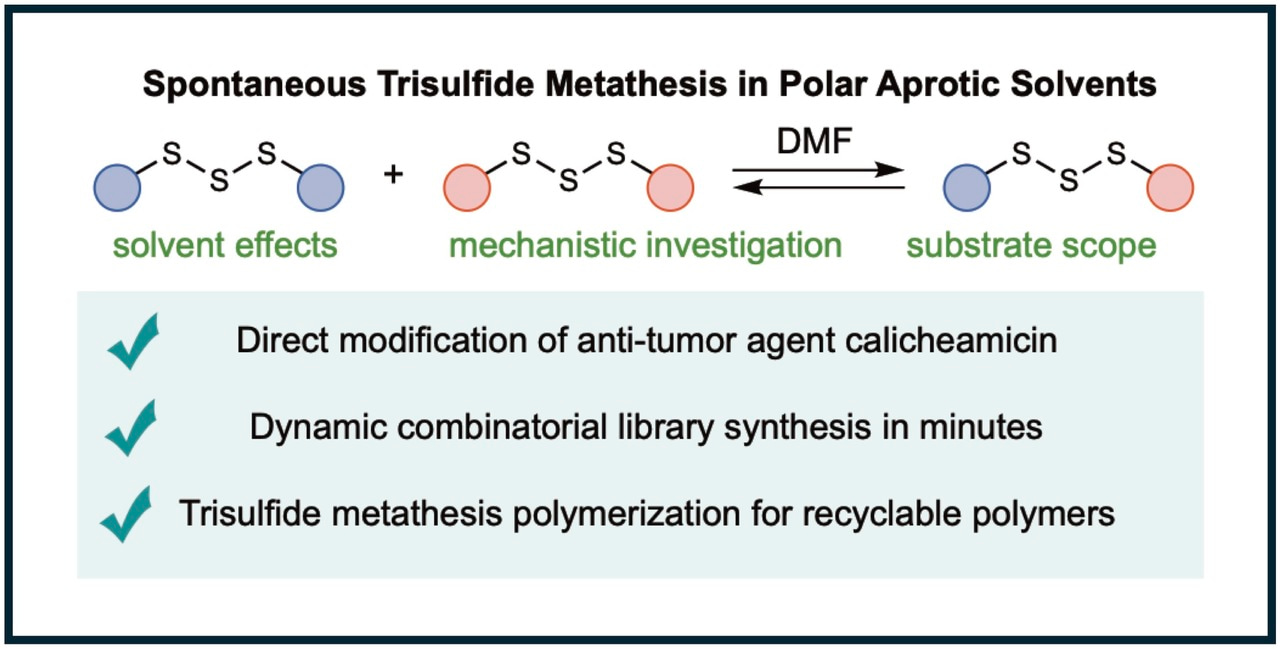
Sulfur-sulfur bonds are ubiquitous in chemical, biological and material sciences: they are found in natural products, medicine, peptides and proteins, and synthetic polymers such as vulcanized rubber. Making and breaking these S-S bonds in a controlled manner is therefore an important capability. Traditionally, S-S exchange reactions are provoked by reaction with a nucleophile or catalyst, or some energy source such as light, heat, mechanical force, or electrochemical potential. It is extremely rare for the S-S exchange to happen spontaneously, without application of these reagents or stimuli. For this reason, we were very surprised to uncover a spontaneous S-S metathesis reaction of trisulfides that requires nothing more than a polar aprotic solvent.
The discovery began with a series of unusual observations in a collaboration between our lab at Flinders University and Dr Tom Hasell’s lab at the University of Liverpool. We both had observed that solvents such as pyridine and DMF caused S-S exchange in trisulfides. We attributed the pyridine reaction to a nucleophilic catalysis mechanism, and the DMF reaction to coordination and weaking of the S-S bond—leading to homolytic cleavage and subsequent metathesis. While these were reasonable proposals at the outset, it turns out that neither of these hypotheses were correct. After years of investigation into the substrate scope, kinetics, selectivity, inhibitors, and theoretical calculations, we confidently ruled out both ionic and radical mechanisms. This was exciting and puzzling, given that nearly every S-S exchange reaction ever reported invokes such mechanisms. Ultimately, we built up a mechanistic understanding and new hypotheses that better account for the many interesting features of this reaction including its exquisite selectivity for making trisulfides (and not disulfides or tetrasulfides), extremely high rate of reaction in polar aprotic solvents, and the inhibition of the reaction by electrophiles and acids. Our working hypothesis invokes an unusual thiosulfoxide intermediate that reacts directly with another trisulfide in a concerted metathesis reaction. While such mechanisms cannot be proven, this hypothesis is so far our best account for the many interesting features of this reaction.
With a mechanistic framework in hand, we set out to see how it can be used in synthesis. The fast and reversible nature of the reaction prompted us to apply it to dynamic combinatorial libraries, which are useful in drug discovery. Here we showed that 8 trisulfides can be converted into 29 trisulfides in minutes, just by dissolving them in DMF.
The selective nature of the reaction motivated us to test it on complex natural products. We demonstrated that one of the most famous trisulfides (the ene-diyne anti-tumor drug, calicheamicin) can undergo trisulfide metathesis in minutes in DMF. Remarkably, the many sensitive functional groups in this complex molecule were not affected by the trisulfide metathesis reaction. The ability to selectively modify this molecule in short order may have potential use in future biomedical research.
Finally, we used monomers with trisulfide end-groups to demonstrate the first trisulfide metathesis polymerization—a new method to make poly(trisulfides). The beautiful thing about this polymerization is that it is very fast and reversible. In the synthesis, the co-product dimethyl trisulfide is removed by distillation as the polymer is formed. The polymer can be molded and used in a number of applications, and then depolymerized when recycling is required. The depolymerization is triggered by re-subjecting the polymer to dimethyl trisulfide and a polar aprotic solvent such as DMF. The depolymerization regenerates the monomer very cleanly and in high yield. We are currently exploring this chemistry as a platform for generally recyclable polymers—especially those that are not easy to recycle with current technologies.
This paper is a testament to the value of curiosity driven research, and the power of interdisciplinary collaboration.
Collaborator and senior author Dr Tom Hasell, recounting the early stages of the project: “A few years ago, my student Peiyao Yan and I noticed that a crosslinked polymer she made become soluble in the presence of a chemical solvent, DMF. A crosslinked polymer is one where all the polymer chains are bound together like a tangled net, which is why they shouldn’t dissolve. In the presence of DMF the polymer was behaving like it had broken back apart. We reached out to Justin’s group for help as we knew they had experienced similar behaviour in other systems, and I’m so glad we did. Without them we wouldn’t have realised how unusual and potentially significant this behaviour was and would certainly never have got to the bottom of what was behind it. It’s been a privilege to be part of this collaborative process, and a great reminder of the benefits of working together across groups to share expertise to achieve what we can’t alone. As for what’s next, I think the examples we’ve shown of what can be done with this chemistry are only the tip of the iceberg. The trisulfide metathesis gives a really versatile way to make reversible changes in both molecular and materials chemistry and I can’t wait to see what ideas other groups come up with on how to use it in the future.”
I whole-heartedly agree.
What began as a curiosity has now unfolded into a rich story of scientific discovery and innovation. It is a lesson to budding researchers to not ignore unusual observations—they may reveal something entirely new and valuable. This project also demonstrates the benefit of long-term, interdisciplinary collaboration that brings together diverse experts that elevate the scope and impact of the scientific endeavour. We uncovered a useful reaction, never before reported, despite the study of these molecules for more than half a century. Then, through the dedication and ingenuity of our collaborative team, demonstrated several ways in which the reaction can be used in chemistry, biomedical chemistry, and recyclable polymers.
I’m grateful to our collaborative team of dedicated researchers that helped chip away at the mechanism over many years and realize the potential of this useful reaction. There is still much to do and understand with this chemistry, and I’m excited to see where this research takes us next.
The study Spontaneous Trisulfide Metathesis in Polar Aprotic Solvents was published in Nature Chemistry (DOI: 10.1038/s41557-026-02091-z).
Professor Justin M. Chalker is a Matthew Flinders Professor of Chemistry at Flinders University in Adelaide, South Australia
Follow the Topic
-
Nature Chemistry

A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.
Ask the Editor – Polymers
Got a question for the editor about Functional polymers? Ask it here!
Continue reading announcement

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in