A marriage of tumor slices and microfluidics
Published in Cancer

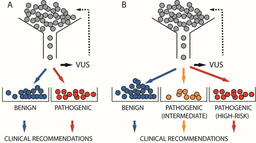
It all started with a simple question: Would it be possible to test multiple cancer drugs on a small, live, and intact tumor biopsy to help doctors decide which treatment is most efficacious? In 2012 this question occurred to the microfluidic toolmaker in our team, Albert Folch, when cancer pathologist (and previous collaborator) Ray Monnat first mentioned to him the role of the tumor microenvironment in drug efficacy. In a first meeting, they strategically decided that such an experimental procedure should target very hopeless cancers, with the help of a clinician-researcher who could provide expertise and clinical samples. Luckily we had, on the UW campus, an enthusiastic neurosurgeon, Bob Rostomily, who specializes on gliomas, brain cancers that are notoriously difficulty to treat. His lab had identified genomic instability as a potential driver of aging-related glioma malignancy (Aging Cell 2012) and intra-tumor genomic heterogeneity in high-grade gliomas (Genome Biol 2014, in collaboration with the Shendure lab). Dr. Rostomily saw that the ability to test multiple drugs on multiple tumor biopsies from the same patient could provide a practical solution to account for the confounding impact of sampling bias for informing precision therapies. (Dr. Rostomily has since moved to Houston Methodist.) Yet we had no preliminary data at the time – only a hunch that it should work, and that microfluidics was an ideal technology to tackle this important problem because clinical samples are often scarce. The Coulter Foundation came to the rescue with a pilot grant that allowed us to hire a bioengineering Ph.D. student, Tim Chang. Tim worked very hard in producing the first prototypes in PDMS, which allowed us to obtain enough preliminary data to publish a Lab on a Chip paper (2014) and launch an NIH R01 in 2014. However, we encountered a major manufacturing challenge – fabrication of each PDMS device took at least two days and required precise alignment at multiple steps that had been perfected by Tim, but never as well executed by his successors. While doable, the fabrication process was impractical. We went back to the drawing board and re-designed the platform in plastic (PMMA), so we could make many copies of the device in a reasonable amount of time. Ph.D. student Adán Rodriguez and M.S. student Kurt Castro put together a new laser-cutting process (now published in a companion Lab on a Chip 2020 paper). In collaboration with Adán, research scientist Lisa Horowitz, M.D., Ph.D. characterized and applied the platform to perform functional drug testing on live slices of xenografts and clinical samples. The platform – branded, in jest, as “Oncoslice” by Dr. Rostomily – received a patent in 2016 and formed the seed for a startup company, OncoFluidics (founded by Dr. Horowitz and Prof. Folch, also married) that seeks to harness the power of microfluidics for testing intact tissues.
Link to the paper: https://www.nature.com/article...
REFERENCE:
L. F. Horowitz, A. D. Rodriguez, Z. Dereli-Korkut, R. Lin, K. Castro, A. M. Mikheev, R. J. Monnat Jr., A. Folch & R. C. Rostomily, “Multiplexed drug testing of tumor slices using a microfluidic platform”, Nature Precision Oncology 4: 12 (2020).
Follow the Topic
-
npj Precision Oncology

An international, peer-reviewed journal committed to publishing cutting-edge scientific research in all aspects of precision oncology from basic science to translational applications to clinical medicine.
Related Collections
With Collections, you can get published faster and increase your visibility.
AI Approaches in Drug Design
Publishing Model: Open Access
Deadline: Mar 31, 2026
Genomic Instability
Publishing Model: Open Access
Deadline: Mar 24, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in