A mycobacterial effector promotes ferroptosis-dependent pathogen pathogenicity and dissemination
Published in Microbiology

Cell death modalities, which are common outcomes during pathogen infection, have been reported to be exploited by pathogens for their pathogenicity and dissemination1. Emerging evidence suggests that ferroptosis, a recently defined form of programmed cell death driven by iron-dependent lipid peroxidation, plays a critical role in infectious diseases and is usually manipulated by pathogens to enhance their pathogenicity and dissemination2. However, the specific pathogen effectors and the underlying molecular mechanisms that regulate ferroptosis remain poorly understood.
Tuberculosis (TB) is a major chronic infectious disease caused by Mycobacterium tuberculosis (Mtb). In 2021, it is estimated that 10.6 million people fell ill with TB, a 4.5% increase from the previous year, and 1.6 million people died from TB3. Moreover, the continuing spread of drug-resistant TB has become one of the most urgent and difficult challenges confronting global TB control. Against this backdrop, we have been focusing on the investigation of the molecular mechanisms underlying Mtb-host interactions to explore novel targets and rational strategies for anti-TB therapy.
TB is characterized by the formation of caseous necrosis, which exhibits important hallmarks of ferroptosis including reduced levels of glutathione (GSH), glutathione peroxidase 4 (GPX4), as well as accumulation of lipid peroxides, and blocking the host ferroptotic pathway suppresses pathogen dissemination4, 5. But up to now, the specific Mtb effectors involved in host ferroptosis regulation and the underlying molecular mechanisms remain largely unexplored, which knowledge could provide new strategies and potential targets for TB treatments targeting pathogen pathogenicity and dissemination.
Mtb eukaryotic-like phosphatases and kinases usually play important roles in regulating host multiple cellular processes including apoptosis, cell proliferation, phagosome acidification, autophagy flux, and immune signaling pathways6, and thus having emerged as prominent therapeutic targets for anti-TB drugs. Therefore, through screening Mtb effectors regulating ferroptosis among Mtb eukaryotic-like phosphatases and kinases, we identified Mtb PtpA as an important effector protein involved in host ferroptosis induction. As reported previously, we have demonstrated that PtpA is a critical effector in promoting cell proliferation and inhibiting phagosome acidification as well as immune signaling pathways, depending on its tyrosine phosphatase and DNA-binding activities7, 8. In this study, we further reveal an additional function of PtpA in triggering ferroptosis by mediating the epigenetic modification in the host cell nucleus, which is independent of its tyrosine phosphatase and DNA-binding activities.
Mechanistic studies demonstrate that ferroptosis induced by Mtb PtpA requires its translocation from the cytoplasm to the nucleus. Multiple pathogens have been found to disturb various host cellular processes upon host nuclear entry9. Most bacterial effectors harbor eukaryotic nuclear localization signals (NLS) and cross the host nuclear envelope barrier depending on host importin α/β10. However, PtpA does not contain currently identifiable nuclear import motifs including nuclear localization signal (NLS) and ankyrin repeats (ARs), but enters the nucleus through the interaction between its Cys11 site and the nuclear entry transporter Ran/NTF2 complex of the host cell. It is worth mentioning that the Cys11 site in Mtb PtpA is highly conserved among pathogenic bacteria, such as Mycobacterium lepromatosis and Staphylococcus aureus, suggesting that the non-canonical RanGDP-binding Cys site may serve as a potential target against pathogenic bacteria by preventing pathogen effectors from entering host cell nucleus.
Through further exploring the mechanism by which PtpA induces ferroptosis after entering the nucleus, we found that the nuclear PtpA binds to protein arginine methyltransferase 6 (PRMT6) via its 1-50 amino acids region to increase asymmetric dimethylation of histone H3 at arginine 2 (H3R2me2a), thus inhibiting GPX4 transcription and expression, eventually inducing ferroptosis to promote Mtb pathogenicity and dissemination. Epigenetic modifications are pivotal for regulating gene expression in response to extracellular stimuli and are therefore often targeted by pathogens including Mtb to help the pathogen escape from host immune defense and survive within host cells11. Unusually, our study puts forth the viewpoint that pathogens hijack epigenetic modification to promote their pathogenicity and dissemination. It should also be mentioned that ferroptosis is usually regulated by multiple signaling pathways including cysteine-GSH-GPX4 axis, iron metabolism, reactive oxygen species (ROS) metabolism, and MAPK pathway, among which the cysteine-GSH-GPX4 axis is identified as the classical core regulator of ferroptosis12. Our findings show that PtpA triggers ferroptosis by directly disrupting the core axis of ferroptosis via targeting PRMT6, indicating that PtpA is a critical pro-ferroptotic effector in host-pathogen interaction.
In summary, our study reveals the regulatory role and detailed molecular mechanism of mycobacterial effector PtpA in promoting ferroptosis-dependent pathogen pathogenicity and dissemination (Figure 1), which provide previously unrecognized insights into pathogen-induced ferroptosis. The followings highlight the significance and breakthroughs of this study: 1) Identify an important effector protein inducing ferroptosis during Mtb infection, and reveal the molecular mechanism of Mtb regulating cell death through epigenetic modification. 2) Reveal a non-classical nuclear entry mechanism and the involved key site of Mtb PtpA. 3) Provide a new TB treatment strategy via targeting Mtb PtpA-host PRMT6 interface to inhibit Mtb-induced GPX4-dependent ferroptosis, thus reducing Mtb pathogenicity and dissemination.
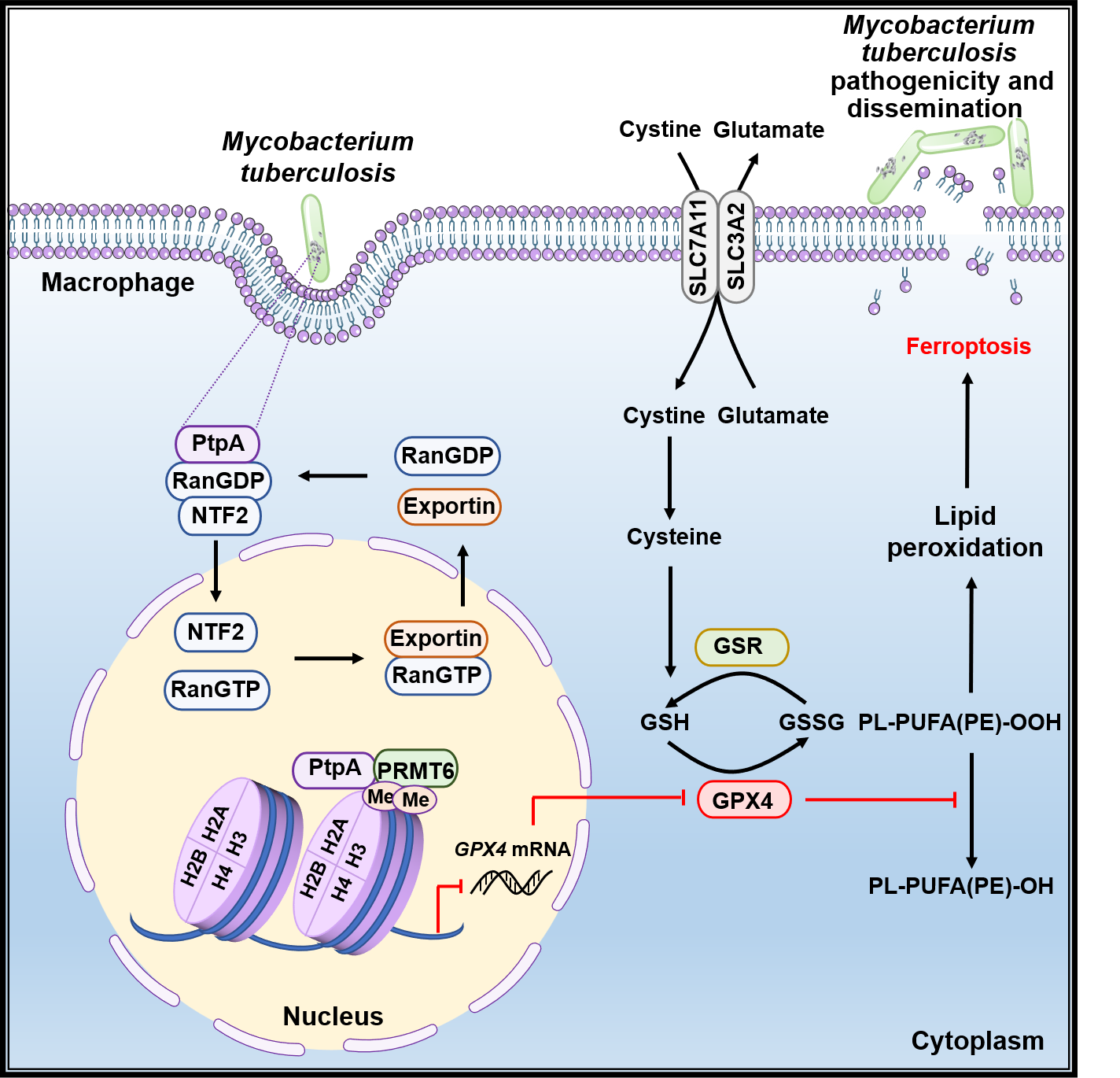
Figure 1. Schematic model showing that mycobacterial PtpA enhances PRMT6-mediated H3R2me2a to promote ferroptosis-dependent pathogen pathogenicity and dissemination.
References
- Jorgensen, I., Rayamajhi, M. & Miao, E.A. Programmed cell death as a defence against infection. Nat Rev Immunol 17, 151-164 (2017).
- Dixon, S.J. et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060-1072 (2012).
- World Health Organization. Global tuberculosis report 2022. WHO Press (2022).
- Sarathy, J.P. & Dartois, V. Caseum: a Niche for Mycobacterium tuberculosis Drug-Tolerant Persisters. Clin Microbiol Rev 33 (2020).
- Amaral, E.P. et al. A major role for ferroptosis in Mycobacterium tuberculosis-induced cell death and tissue necrosis. J Exp Med 216, 556-570 (2019).
- Wong, D., Chao, J.D. & Av-Gay, Y. Mycobacterium tuberculosis-secreted phosphatases: from pathogenesis to targets for TB drug development. Trends Microbiol 21, 100-109 (2013).
- Wang, J. et al. The mycobacterial phosphatase PtpA regulates the expression of host genes and promotes cell proliferation. Nat Commun 8, 244 (2017).
- Wang, J. et al. Mycobacterium tuberculosis suppresses innate immunity by coopting the host ubiquitin system. Nat Immunol 16, 237-245 (2015).
- Bierne, H. & Pourpre, R. Bacterial Factors Targeting the Nucleus: The Growing Family of Nucleomodulins. Toxins (Basel) 12 (2020).
- Bhat, K.H., Srivastava, S., Kotturu, S.K., Ghosh, S. & Mukhopadhyay, S. The PPE2 protein of Mycobacterium tuberculosis translocates to host nucleus and inhibits nitric oxide production. Sci Rep 7, 39706 (2017).
- Kathirvel, M. & Mahadevan, S. The role of epigenetics in tuberculosis infection. Epigenomics 8, 537-549 (2016).
- Xie, Y. et al. Ferroptosis: process and function. Cell Death Differ 23, 369-379 (2016).
Follow the Topic
-
Nature Communications

An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in