Alkali-Ion-Modified Zeolitic Imidazolate Framework Glasses
Glasses are ubiquitous in modern life. From windows and ovenware to smartphone screens, packaging, and protective coatings, they surround us. Their technological success arises from two key features: the excellent processability of the liquid state and the remarkable tunability of their chemical composition. Properties such as color, thermal parameters, resistance to thermal shock, and mechanical strength can all be adjusted through compositional design.
At first glance, this versatility seems surprising. Silicate glasses are based on a single glass former: silicon dioxide. The wide range of properties arises not from the glass former itself but from chemical modification introduced during flux melting. The introduction of modifiers such as alkali oxides lowers the connectivity of the glass network through a depolymerization mechanism, while lowering processing temperatures and facilitating glass manufacturing.
This concept has a long history. Archaeological evidence suggests that early glassmakers in Mesopotamia were already modifying glass compositions around 1500 BC. Over the following centuries, glass science has developed a detailed framework linking composition, structure, and properties in silicate glasses.
Our work asks whether these ideas can be transferred to a much younger class of glass-forming materials: zeolitic imidazolate framework (ZIF) glasses.
ZIFs belong to the broader family of metal–organic frameworks (MOFs), porous materials constructed from organic and inorganic building units. In ZIFs, typically divalent transition-metal ions such as Zn2+ or Co2+ are connected by imidazolate-type linkers, forming network structures reminiscent of those found in inorganic zeolites.
While most MOFs decompose upon heating, a small subset of ZIFs displays an unusual property: they can melt and form glasses. Rapid cooling of the melt produces a vitrous material that partially retains the microporosity of the crystalline parent framework. Materials that combine the processability of glasses with the tunable porosity of MOFs could offer new opportunities for industrial separation technologies.
Despite this promise, the chemical landscape of ZIF glasses remains limited. Only a few ZIF compositions are known to melt before decomposing, restricting the range of accessible metal ions and organic linkers. Expanding this compositional space is therefore an important goal.
Inspired by classical silicate glass chemistry, we investigated whether ZIF glasses could also be chemically modified using the respective alkali salts of their linkers.
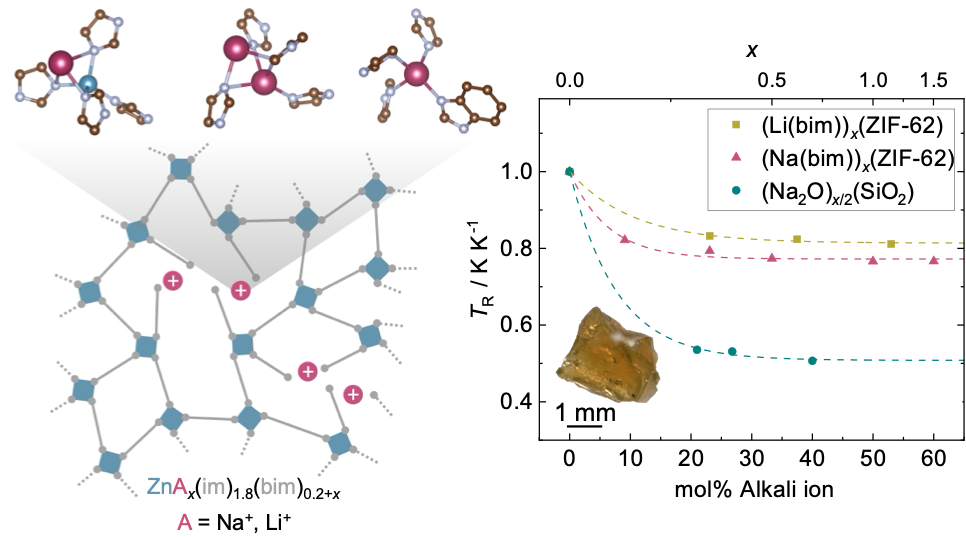
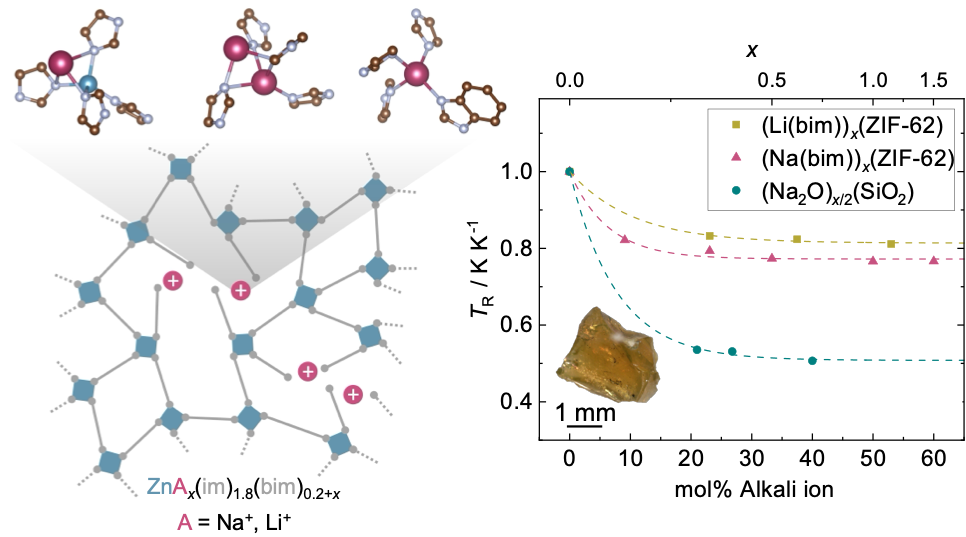
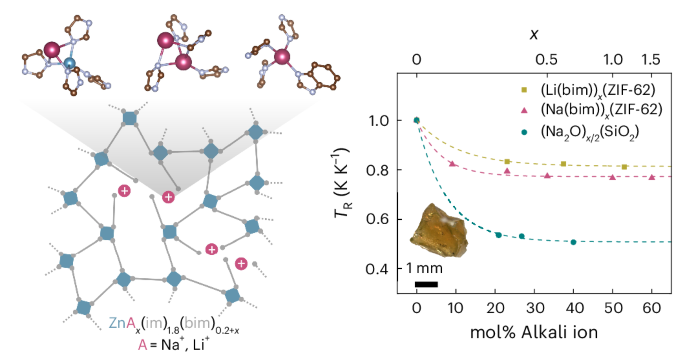
To explore this idea, we focused on ZIF-62, one of the best-known ZIF glass formers, with the composition Zn(im)1.8(bim)0.2 (im– = imidazolate, bim– = benzimidazolate). Introducing lithium or sodium benzimidazolate allowed us to mimic the role of alkali modifiers in conventional silicate glasses.
Melt-quenching mixtures of ZIF-62 with increasing amounts of sodium benzimidazolate produced a series of alkali-ion-modified ZIF glasses. The trends in thermal parameters, such as a decrease in glass transition temperature and increased liquid fragility upon growing modifier content, resemble those observed in alkali-modified silicate glasses. Structural characterisation confirmed that sodium benzimidazolate is homogeneously incorporated into the ZIF glass network. Analogously, growing modifier content leads to reduced network connectivity, increased structural disorder, and a slight densification of the glass while maintaining the tetrahedral coordination environment of the zinc centers.
To gain further insight into sodium-ion incorporation, our team at TU Dortmund University joined forces with Dominik Kubicki and Benjamin M. Gallant (University of Birmingham), who conducted 23Na solid-state NMR spectroscopy measurements, alongside computational modelling by Andrew J. Morris and Mario Antonio T. Ongkiko (also at the University of Birmingham). Together, these approaches revealed three distinct sodium-ion environments. Most sodium ions occupy tetrahedral network sites, while a smaller fraction resides inside pores in lower-coordination environments. These findings highlight substantial differences between ZIF and silicate glasses, arising from the directional bonding and steric constraints of the imidazolate linkers.
Finally, we explored whether the incorporated modifiers could be removed to further tailor porosity. Simple extraction with water increased the mesopore volume of the material, analogous to the well-known VYCOR process used to produce porous borosilicate glasses through acid-leaching.
The ability to engineer pore structures through glass modification followed by water-leaching offers a promising strategy for designing future ZIF-glass separation membranes with tunable separation performance.
More broadly, our work demonstrates that principles established in traditional glass chemistry can also guide the design of future MOF glasses, providing a conceptual framework for tuning their structure, porosity, and material properties.
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in